Prostaglandin F2a
Synonym(s):Prostaglandin F 2α - CAS 551-11-1 - Calbiochem
- CAS NO.:551-11-1
- Empirical Formula: C20H34O5
- Molecular Weight: 354.49
- MDL number: MFCD00135231
- EINECS: 215-355-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-11-19 20:33:22
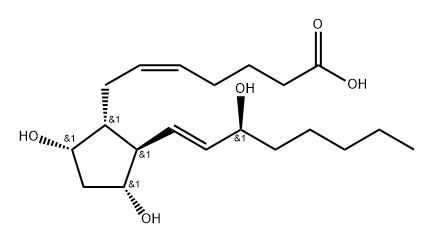
What is Prostaglandin F2a?
Description
Prostaglandin F2α (PGF2α) is a widely distributed PG occurring in many species. It causes contraction of vascular, bronchial, intestinal, and myometrial smooth muscle, and also exhibits potent luteolytic activity. PGF2α exerts its receptor mediated physiological activity at 50-
History
Prostaglandins (PGs) are a class of important endogenous products with a wide range of physiological activities. PGs were first discovered and named by American scholar Von Eluer in 1930. In 1962, Bergstorm extracted two pure PGs (PGF1 and PGF2) and determined their chemical structures. In 1969, Willis first proposed that PGs are an inflammatory mediator in the body. Subsequently, various physiological and pharmacological activities of PGs have been intensively studied.
The Uses of Prostaglandin F2a
Prostaglandin F2α is one of the most biologically studied of the primary prostaglandins. Closely related to Prostaglandin E2 (PGE2) in that both prostaglandins are biosynthesized from the same precursors and that PGF2 is the synthetic reduction product of PGE2.
Background
Dinoprost has been investigated in Headache.
What are the applications of Application
Prostaglandin F2α is a potent smooth muscle contricting prostaglandin shown to inhibit adipocyte differentiation
Definition
ChEBI: Prostaglandin F2α is a prostaglandins Falpha that is prosta-5,13-dien-1-oic acid substituted by hydroxy groups at positions 9, 11 and 15. It is a naturally occurring prostaglandin used to induce labor.
Pharmacokinetics
Dinoprost is a natural prostaglandin F2α (PGF2α), which can directly act on the myometrium, stimulate the pregnant uterus to contract the uterine muscle, and can soften and dilate the cervix, so it can be used for induced abortion and late labor induction. However, due to the instability of dinoprost at room temperature, inconvenient storage and transportation, complex synthesis process and high cost, the application of dinoprost is difficult to popularize.
Safety Profile
Poison by subcutaneous, intravenous, and intramuscular routes. Moderately toxic by ingestion. Human and experimental teratogenic and experimental reproductive effects. Human reproductive effects by subcutaneous, intravenous, intramuscular, intraperitoneal, intravaginal, and intraplacental routes: postpartum depression and other maternal effects, abortion, and changes in measures of ferulity. Human teratogenic effects by intraplacental route: extra embryonic structures. Human systemic effects by intravenous route: hypermoulity, diarrhea, nausea or vomiting. Human mutation data reported. When heated to decomposition it emits acrid smoke and fumes
Metabolism
Not Available
Mode of action
Prostaglandin F2α (PGF2α) stimulates myometrial activity, relaxes the cervix, inhibits corpus luteal steroidogenesis, and induces luteolysis by direct action on the corpus luteum. In pregnancy, PGF2α is medically used to sustain contracture and provoke myometrial ischemia to accelerate labor and prevent significant blood loss in labor.
Properties of Prostaglandin F2a
| Melting point: | 25-35° |
| Boiling point: | 407.69°C (rough estimate) |
| alpha | D25 +23.5° (c = 1 in tetrahydrofuran) |
| Density | 1.0458 (rough estimate) |
| refractive index | 1.6120 (estimate) |
| storage temp. | Store at -20°C |
| solubility | DMSO:100.0(Max Conc. mg/mL);282.1(Max Conc. mM) Water:100.0(Max Conc. mg/mL);282.1(Max Conc. mM) |
| pka | pKa 4.90(H2O,t=25±2,I=0.0,c<0.01,N2) (Uncertain) |
| form | Low melting white solid or colorless oil. |
| color | White to light brown |
| CAS DataBase Reference | 551-11-1(CAS DataBase Reference) |
Safety information for Prostaglandin F2a
| Signal word | Danger |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07  Health Hazard GHS08 |
| GHS Hazard Statements |
H302:Acute toxicity,oral |
| Precautionary Statement Codes |
P201:Obtain special instructions before use. P202:Do not handle until all safety precautions have been read and understood. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P270:Do not eat, drink or smoke when using this product. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P308+P313:IF exposed or concerned: Get medical advice/attention. |
Computed Descriptors for Prostaglandin F2a
| InChIKey | PXGPLTODNUVGFL-JPYXJAOSNA-N |
| SMILES | C([C@H]1[C@H](C[C@@H](O)[C@@H]1/C=C/[C@@H](O)CCCCC)O)/C=C\CCCC(=O)O |&1:1,2,4,6,9,r| |
Prostaglandin F2a manufacturer
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid 1-Bromo-3,5-Di-Tert-Butylbenzene S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran

You may like
-
 551-11-1 Dinoprost 98%View Details
551-11-1 Dinoprost 98%View Details
551-11-1 -
 Prostaglandin F2α CAS 551-11-1View Details
Prostaglandin F2α CAS 551-11-1View Details
551-11-1 -
 Prostaglandin F 2α CAS 551-11-1View Details
Prostaglandin F 2α CAS 551-11-1View Details
551-11-1 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1