Citric acid
Synonym(s):2-Hydroxy-propane-1,2,3-tricarboxylic acid;2-Hydroxypropane-1,2,3-tricarboxylic acid, Hydroxytricarballylic acid;Citric acid
- CAS NO.:77-92-9
- Empirical Formula: C6H8O7
- Molecular Weight: 192.12
- MDL number: MFCD00011669
- EINECS: 201-069-1
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-23 21:30:31
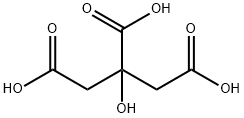
What is Citric acid?
Toxicity
ORAL (LD50): Acute: 5040 mg/kg [Mouse]. 3000 mg/kg [Rat].
Definition
Citric acid is an important natural compound known since the late 18th century. The pioneering Swedish–German chemist Carl Wilhelm Scheele isolated it from lemon juice in 1784. It has since been found in other citrus fruits, pineapples, and even animal tissues. With its 3 carboxyl groups and one hydroxyl group, Citric acid can serve as a crosslinker between polymer molecules and as a linker to link cyclodextrins and a polymer, such as HPMC.
Description
Citric acid is a weak organic acid containing three carboxylic acid functional groups. In organic chemistry it is generally used as an aqueous solution during aqueous work-ups.
Chemical properties
Citric acid is soluble 66 % in water, 33% in alcohol,
3% in ether. Soluble about 20% in Propylene
glycol. Virtually odorless. The aqueous solution
has a clean acid taste, pleasant in the concentration of 0.02 to 0.08%. Citric acid is a weak organic acid with the formula C6H8O7. It is a natural preservative / conservative and is also used to add an acidic, or sour, taste to foods and soft drinks. In biochemistry, the conjugate base of citric acid, citrate, is important as an intermediate in the citric acid cycle, which occurs in the metabolism of all aerobic organisms.
Citric acid is a commodity chemical, and more than a million tonnes are produced every year by fermentation. It is used mainly as an acidifier, as a flavoring, and as a chelating agent.
Physical properties
CITRIC ACID, white crystalline solid, decomposes at higher temperatures, sp gr 1.542. Citric acid is soluble in H2O or alcohol and slightly soluble in ether. pKa values are 5.21, 4.28 and 2.92 at 25 °C (extrapolated to zero ionic strength).
In biological systems around pH 7, the two species present are the citrate ion and mono-hydrogen citrate ion. the pH of a 1 mM solution of citric acid will be about 3.2.
History
The discovery of citric acid is credited to Jabir ibn Hayyan (Latin name Geber, 721–815).
Citric acid was first isolated in 1784 by the Swedish chemist Carl Wilhelm Scheele (1742–1786), who crystallized it from lemon juice.
The crystalline structure of anhydrous citric acid, obtained by cooling hot concentrated solution of the monohydrate form, was first elucidated by Yuill and Bennett in 1934 by X-ray diffraction.
In 1960 Nordman and co-workers further suggested that in the anhydrous form two molecules of the acid are linked through hydrogen bonds between two –COOH groups of each monomer.
The Uses of Citric acid
citric acid has astringent and anti-oxidant properties. It can also be used as a product stabilizer, pH adjuster, and preservative with a low sensitizing potential. It is not usually irritating to normal skin, but it can cause burning and redness when applied to chapped, cracked, or otherwise inflamed skin. It is derived from citrus fruits.
The Uses of Citric acid

A mixture of the SM (36.8 g, 0.12 mol), aq 2N NaOH (850 mL) and dioxane (170 mL) were stirred at 50 C for 24 h. The reaction mixture was cooled to RT, diluted with EtOAc (1.0 L), and acidified to pH 1 using aq 4N HCl. The resulting precipitate was filtered, washed with H2O, and dried in vacuo to provide the product as an off-white solid. [25.4 g]
The Uses of Citric acid
Citric Acid is an acidulant and antioxidant produced by mold fermentation of sugar solutions and by extraction from lemon juice, lime juice, and pineapple canning residue. it is the predominant acid in oranges, lemons, and limes. it exists in anhydrous and monohydrate forms. the anhydrous form is crystallized in hot solutions and the monohydrate form is crystallized from cold (below 36.5°c) solutions. anhydrous citric acid has a solubility of 146 g and monohydrate citric acid has a solubility of 175 g/100 ml of distilled water at 20°c. a 1% solution has a ph of 2.3 at 25°c. it is a hygroscopic, strong acid of tart flavor. it is used as an acidulant in fruit drinks and carbonated beverages at 0.25-0.40%, in cheese at 3-4%, and in jellies. it is used as an antioxidant in instant potatoes, wheat chips, and potato sticks, where it prevents spoilage by trapping the metal ions. it is used in combination with antioxidants in the processing of fresh frozen fruits to prevent discoloration.
Background
A key intermediate in metabolism. It is an acid compound found in citrus fruits. The salts of citric acid (citrates) can be used as anticoagulants due to their calcium-chelating ability.
Citric acid is one of the active ingredients in Phexxi, a non-hormonal contraceptive agent that was approved by the FDA on May 2020. It is also used in combination with magnesium oxide to form magnesium citrate, an osmotic laxative.
What are the applications of Application
Citric Acid, Anhydrous is used in the preparation of citrate buffer for antigen and epitope unmasking in IHC
Definition
ChEBI: Citric acid is a tricarboxylic acid that is propane-1,2,3-tricarboxylic acid bearing a hydroxy substituent at position 2. It is an important metabolite in the pathway of all aerobic organisms. It has a role as a food acidity regulator, a chelator, an antimicrobial agent and a fundamental metabolite. It is a conjugate acid of a citrate(1-) and a citrate anion.
Preparation
By mycological fermentation using molasses and strains of Aspergillus niger; from citrus juices and pineapple wastes
What are the applications of Application
Citric acid is a weak organic acid that is known as a commodity chemical, as more than a million tonnes are produced every year by mycological fermentation on an industrial scale using crude sugar sol utions, such as molasses and strains of Aspergillus niger. Citric acid is widely distributed in plants and in animal tissues and fluids and exist in greater than grace amounts in variety of fruits and vegetables, most notably in citrus fruits such as lemon and limes. Citric acid is mainly used as an acidifier, flavoring agent and chelating agent. It was also used as a chemical restrainer particularly in developers for the collodion process and in silver nitrate solutions used for sensitizing salted and albumen papers.
Biotechnological Production
Fermentation is the technology of choice for citric acid synthesis. Different bacteria (e.g. Arthrobacter paraffinens and Bacillus licheniformis), filamentous fungi (e.g. Aspergilus niger and Penicillium citrinum) and yeasts (e.g. Candida tropicalis and Yarrowia lipolytica) are able to produce citric acid. Due to high productivity and easy handling, citric acid is usually produced by fermentation with A. niger.
Aroma threshold values
By mycological fermentation using molasses and strains of Aspergillus niger; from citrus juices and pineapple wastes
Benefits
Citric acid is not a vitamin or mineral and is not required in the diet. However, citric acid, not to be confused with ascorbic acid (vitamin C), is beneficial for people with kidney stones. It inhibits stone formation and breaks up small stones that are beginning to form. Citric acid is protective; the more citric acid in your urine, the more protected you are against forming new kidney stones. Citrate, used in calcium citrate supplements and in some medications (such as potassium citrate), is closely related to citric acid and also has stone prevention benefits. These medications may be prescribed to alkalinize your urine.
General Description
Citric acid appears as colorless, odorless crystals with an acid taste. Denser than water. (USCG, 1999)
Air & Water Reactions
The pure material is moisture sensitive (undergoes slow hydrolysis) Water soluble.
Reactivity Profile
Citric acid reacts with oxidizing agents, bases, reducing agents and metal nitrates . Reactions with metal nitrates are potentially explosive. Heating to the point of decomposition causes emission of acrid smoke and fumes [Lewis].
Health Hazard
Inhalation of dust irritates nose and throat. Contact with eyes causes irritation.
Biochem/physiol Actions
Citric acid in dietary form can augments absorption of aluminium in antacids. It also facilitates the phytoremediation of heavy metal contaminated soil and can transform cadmium into more transportable forms.
Biotechnological Applications
Citrate, the conjugate base of citric acid is one of a series of compounds involved in the physiological oxidation of fats, proteins, and carbohydrates to carbon dioxide and water.This series of chemical reactions is central to nearly all metabolic reactions, and is the source of two-thirds of the foodderived energy in higher organisms. The series of reactions is known by various names, including the "citric acid cycle", the "Krebs cycle" or "Szent-Gyergyi — Krebs cycle", and the "tricarboxylic acid (TCA) cycle".
Besides, citrate is a critical component of bone, helping to regulate the size of calcium crystals.
Safety Profile
Poison by intravenous route. Moderately toxic by subcutaneous and intraperitoneal routes. Mildly toxic byingestion. A severe eye and moderate skin irritant. An irritating organic acid, some allergenic properties. Combustible liquid. Potentially explosive reaction with metal nitrates. When heated to decomposition it emits acrid smoke and fumes.
Natural Source
Citric acid is a white, crystalline, weak organic acid present in most plants and many animals as an intermediate in cellular respiration. Lemons and limes have high citric acid content, which may account for up to 8% of the fruit's dry weight.
Citrus fruits such as lemons and limes have the highest quantities of naturally occurring citric acid, according to research from 2014. Other natural sources include tangerines, oranges, grapefruits, pineapples, tomatoes, broccoli, carrots, and berries.
Properties of Citric acid
| Melting point: | 153-159 °C (lit.) |
| Boiling point: | 248.08°C (rough estimate) |
| Density | 1.67 g/cm3 at 20 °C |
| vapor density | 7.26 (vs air) |
| vapor pressure | <0.1 hPa (20 °C) |
| FEMA | 2306 | CITRIC ACID |
| refractive index | 1.493~1.509 |
| Flash point: | 100 °C |
| storage temp. | 2-8°C |
| solubility | Citric acid also dissolves in absolute (anhydrous) ethanol (76 parts of citric acid per 100 parts of ethanol) at 15 °C. |
| appearance | colorless or white crystals or powder |
| appearance | White crystalline solid |
| form | grit |
| pka | 3.14(at 20℃) |
| color | White |
| PH | 3.24(1 mM solution);2.62(10 mM solution);2.08(100 mM solution); |
| Odor | Odorless |
| explosive limit | 8%, 65°F |
| Water Solubility | soluble in Water (1174g/L at 10°C, 1809g/L at 30°C, 3825g/L at 80°C). |
| Sensitive | Hygroscopic |
| λmax | λ: 260 nm Amax: 0.20 λ: 280 nm Amax: 0.10 |
| Merck | 14,2326 |
| JECFA Number | 218 |
| BRN | 782061 |
| Stability: | Stable. Incompatible with bases, strong oxidizing agents, reducing agents, metal nitrates. |
| CAS DataBase Reference | 77-92-9(CAS DataBase Reference) |
| NIST Chemistry Reference | 1,2,3-Propanetricarboxylic acid, 2-hydroxy-(77-92-9) |
| EPA Substance Registry System | Citric acid (77-92-9) |
Safety information for Citric acid
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Citric acid
| InChIKey | KRKNYBCHXYNGOX-UHFFFAOYSA-N |
Citric acid manufacturer
JSK Chemicals
Bisan Chemicals And Engineers Private Limited
Wang Pharmaceuticals & Chemicals
Oxford Lab Fine Chem LLP
Vasa Pharmachem Pvt Ltd. (VPPL)
R P Chemicals
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran

You may like
-
 Citric Acid 98%View Details
Citric Acid 98%View Details -
 CITRIC ACID ANHYDROUS 99%View Details
CITRIC ACID ANHYDROUS 99%View Details -
 CITRIC ACID ANHYDROUS 99%View Details
CITRIC ACID ANHYDROUS 99%View Details -
 Citric acid 99%View Details
Citric acid 99%View Details -
 Citric acid 99%View Details
Citric acid 99%View Details -
 Citric Acid 99%View Details
Citric Acid 99%View Details -
 Citric Acid CASView Details
Citric Acid CASView Details -
 Buffer solution pH 7.0 (Phosphate) CASView Details
Buffer solution pH 7.0 (Phosphate) CASView Details
