Oxalic acid
Synonym(s):Ethanedioic acid dihydrate;Oxalic acid;Oxalic acid solution
- CAS NO.:144-62-7
- Empirical Formula: C2H2O4
- Molecular Weight: 90.03
- MDL number: MFCD00002573
- EINECS: 205-634-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-03-10 17:18:44
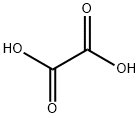
What is Oxalic acid?
Description
Oxalic acid is a colourless, odourless powder or granular solid. The anhydrous form (COOH)2 is an odourless, white solid; the solution is a colourless liquid. Molecularweight = 90.04; Specific gravity (H2O:1) = 1.90; Sublimationpoint =150-157℃; Freezing/Melting point (decomposes):101.7℃; 190℃ (anhydrous). It is one of the strongest organic acids. It is widely used in wood restorers and deck cleaners to dissolve the top layer of dried wood and expose a clean layer.
Chemical properties
Oxalic acid is a colourless, odourless powder or granular solid. The anhydrous form (COOH)2 is an odourless, white solid; the solution is a colourless liquid. Hygroscopic. Soluble in ethanol, soluble in water, slightly soluble in ether, and insoluble in benzene and chloroform.
The Uses of Oxalic acid
Oxalic acid was used in the synthesis of hemicellulose hydrolysates of yellow poplars, used in the synthesis of three-dimensionally ordered macroporous metal oxides or carbonates via templating with polystyrene spheres and as a supporting electrolyte in the electrochemical synthesis of polyaniline-polypyrrole composite coatings. It is made by the action of nitric acid on sugars, starch, or cellulose. This highly poisonous colourless crystal is soluble in water, alcohol, and ether. It was used to make ferric oxalate, as a preservative for pyrogallic acid developers, as a sensitizer for platinum papers, and to reduce the density of cyanotype prints.
Definition
ChEBI: Oxalic acid is an alpha,omega-dicarboxylic acid that is ethane substituted by carboxyl groups at positions 1 and 2. It has a role as a human metabolite, a plant metabolite and an algal metabolite. It is a conjugate acid of an oxalate(1-) and an oxalate.
Production Methods
Many industrial processes have been employed for the manufacture of oxalic acid since it was first synthesized. The following processes are in use worldwide: oxidation of carbohydrates, the ethylene glycol process, the propylene process, the dialkyl oxalate process, and the sodium formate process. Sodium formate process is no longer economical in the leading industrial countries, except for China.
Nitric acid oxidation is used where carbohydrates, ethylene glycol, and propylene are the starting materials. The dialkyl oxalate process is the newest, where dialkyl oxalate is synthesized from carbon monoxide and alcohol, then hydrolyzed to oxalic acid. This process has been developed by UBE Industries in Japan.Many attempts have been made to synthesize oxalic acid by electrochemical reduction of carbon dioxide in either aqueous or nonaqueous electrolytes.
Reactions
The reactions of oxalic acid, including the formation of normal and acid salts and esters, are typical of the dicarboxylic acids class. Oxalic acid, however, does not form an anhydride.
On rapid heating, oxalic acid decomposes to formic acid, carbon monoxide, carbon dioxide, and water. In aqueous solution, it is decomposed by uv, x-ray, or γ -radiation with the liberation of carbon dioxide. Photodecomposition also occurs in the presence of uranyl salts.
Oxalic acid is a mild reducing agent, and is oxidized by potassium permanganate in acid solution to give carbon dioxide and water. Oxalic acid is catalytically reduced by hydrogen in the presence of ruthenium catalyst to ethylene glycol, and electronically reduced to glyoxylic acid.
Oxalic acid reacts with various metals to form metal salts, which are quite important as the derivatives of oxalic acid. It also reacts easily with alcohols to give esters.
General Description
Odorless white solid. Sinks and mixes with water.
Air & Water Reactions
Water soluble. Hygroscopic
Reactivity Profile
Oxalic acid is hygroscopic and sensitive to heat. Oxalic acid may react violently with furfuryl alcohol, silver, sodium, perchlorate, sodium hypochlorite, strong oxidizers, sodium chlorite, acid chlorides, metals and alkali metals. . The heating of mixtures of Oxalic acid and urea has lead to explosions. This is due to the rapid generation of the gases, CO2, CO, and NH3, [Praxis Naturwiss. Chem., 1987, 36(8), 41-42]. Oxalic acid and urea react at high temperatures to form toxic and flammable ammonia and carbon monoxide gasses, and inert CO2 gas [Von Bentzinger, R. et al., Praxis Naturwiss. Chem., 1987, 36(8), 41-42].
Health Hazard
As dust or as a solution, can cause severe burns of eyes, skin, or mucous membranes. Ingestion of 5 grams has caused death with symptoms of nausea, shock, collapse, and convulsions coming on rapidly. Repeated or prolonged skin exposure can cause dermatitis and slow-healing ulcers.
Fire Hazard
Special Hazards of Combustion Products: Generates poisonous gases
Flammability and Explosibility
Non flammable
Agricultural Uses
Oxalic acid, (COOH)2, also called ethanedioic acid, is a
white, crystalline solid, slightly soluble in water. It is a
naturally occurring highly oxidized organic compound with significant chelating activity. It is strongly acidic
and poisonous, produced by many plants like sorrel
(sourwood), the leaf blades of rhubarb, bark of
eucalyptus and many plant roots. In plant cells and
tissues, oxalic acid gets accumulated as either sodium,
potassium or calcium oxalate, of which the latter occurs
as crystals. In turn, salts of oxalic acids enter the bodies
of animals and human beings, causing pathological
disorders, depending upon the amount consumed. Many
species of fungi like Aspergillus, Penicillium, Mucor, as
well as some lichens and slime moulds produce calcium
oxalate crystals. Upon the death of these microorganisms,
plants and animals, the salts get released into
the soil, causing some amount of toxicity. However,
oxalate-degrading microbes, called Oxalobacter
formigenes, decrease oxalate absorption in animals and
humans.
Oxalic acid is the first of a series of dicarboxylic
acids. It is used (a) as a bleaching agent for stains like rust
or ink, (b) in textile and leather production, and (c) as
monoglyceryl oxalate in the production of ally1 alcohol
and formic acid.
Safety Profile
Poison by subcutaneous route. Moderately toxic by ingestion. A skin and severe eye irritant. Acute oxalic poisoning results from ingestion of a solution of the acid. There is marked corrosion of the mouth, esophagus, and stomach, with symptoms of vomiting, burning abdominal pain, collapse, and sometimes convulsions. Death may follow quickly. The systemic effects are attributed to the removal by the oxalic acid of the calcium in the blood. The renal tubules become obstructed by the insoluble calcium oxalate, and there is profound hdney dlsturbance. The chief effects of inhalation of the dusts or vapor are severe irritation of the eyes and upper respiratory tract, gastrointestinal disturbances, albuminuria, gradual loss of weight, increasing weakness and nervous system complaints, ulceration of the mucous membranes of the nose and throat, epistaxis, headache, irritation, and nervousness. Oxalic acid has a caustic action on the skin and may cause dermatitis; a case of early gangrene of the fingers resembling that caused by phenol has been described. More severe cases may show albuminuria, chronic cough, vomiting, pain in the back, and gradual emaciation and weakness. The skin lesions are characterized by crachng and fissuring of the skin and the development of slow-healing ulcers. The skin may be bluish in color, and the nails brittle and yellow. Violent reaction with furfuryl alcohol, Ag, NaClO3, NaOCl. When heated to decomposition it emits acrid smoke and irritating fumes. See also OXALATES
Potential Exposure
Oxalic acid is used in textile finishing, paint stripping; metal and equipment cleaning; as an intermediate; as an analytic reagent and in the manufacture of dyes, inks, bleaches, and paint removers; varnishes, wood, and metal cleansers; dextrin, cream of tartar, celluloid, oxalates, tartaric acid, purified methyl alcohol, glycerol, and stable hydrogen cyanide. It is also used in the photographic, ceramic, metallurgic, rubber, leather, engraving, pharmaceutical, paper, and lithographic industries.
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. If victim is conscious, administer water ormilk. Do not induce vomiting. Medical observation isrecommended for 24-48 h after breathing overexposure, aspulmonary edema may be delayed. As first aid for pulmonary edema, a doctor or authorized paramedic may consideradministering a corticosteroid spray.
Source
Oxalic acid occurs naturally in many plants including buckwheat leaves (111,000 ppm), lambs quarter (140,000 to 300,000 ppm), black pepper (4,000 to 34,000 ppm), star fruit (50,000 to 95,800 ppm), purslane (1,679 to 16,790 ppm), nance bark (27,300 ppm), rhubarb 4,400 to 13,360 ppm), tea leaves (2,192 to 10,000 ppm), bitter lettuce (10,000 ppm), spinach (6,580 ppm), cacao (1,520 to 5,000 ppm), bananas (22 to 5,240 ppm), ginger (5,000 ppm in rhizome), cashews (3,184 ppm), almonds (4,073 ppm), taro roots (1,334 ppm), tamarind (1,960 ppm), garden sorrel (3,000 ppm), mustard green leaves (1,287 ppm), peppers (257 to 1,171 ppm), sweet potato roots (1,000 ppm), pumpkins, oats (400 ppm), tomatillo (109 to 536 ppm), various cabbage leaves (59 to 350 ppm), and horseradish.
Oxalic acid was identified as a constituent in various composted organic wastes. Detectable concentrations were reported in all 21 composts extracted with water. Concentrations ranged from 0.60 mmol/kg in straw + dairy cattle manure to 21.89 mmol/kg in straw + wood bark + dairy cattle manure. The overall average concentration was 9.67 mmol/kg.
Environmental Fate
Biological. Heukelekian and Rand (1955) reported a 5-d BOD value of 0.12 g/g which is 66.7%
of the ThOD value of 0.18 g/g.
Chemical/Physical. At temperatures greater than 189.5 °C, decomposes to carbon dioxide,
carbon monoxide, formic acid, and water (Windholz et al., 1983). Ozonolysis of oxalic acid in
distilled water at 25 °C under acidic conditions (pH 6.3) yielded carbon dioxide (Kuo et al., 1977).
Absorbs moisture in air forming the dihydrate (Huntress and Mulliken, 1941).
Reacts with bases forming water soluble salts.
storage
Color Code—White: Corrosive or Contact Hazard;Store separately in a corrosion-resistant location. Prior toworking with this chemical you should be trained on itsproper handling and storage. Oxalic acid must be stored toavoid contact with silver or strong oxidizers (such as chlorine and bromine) because violent reactions occur. Store intightly closed containers in a cool, well-ventilated areaaway from heat. Sources of ignition, such as smoking andopen flames, are prohibited where oxalic acid is used, handled, or stored in a manner that could create a potential fireor explosion hazard.
Shipping
UN3261 Corrosive solid, acidic, organic, n.o.s., Hazard class: 8; Labels: 8-Corrosive material, Technical Name Required.
Incompatibilities
The aqueous solution is a medium-strong acid. Compounds of the carboxyl group react with all bases, both inorganic and organic (i.e., amines) releasing substantial heat, water and a salt that may be harmful. Incompatible with arsenic compounds (releases hydrogen cyanide gas), diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides (releasing heat, toxic, and possibly flammable gases), thiosulfates and dithionites (releasing hydrogen sulfate and oxides of sulfur). Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from silver compounds; strong alkalis; chlorites. Contact with some silver compounds forms explosive materials.
Waste Disposal
Pretreatment involves chemical reaction with limestone or calcium oxide forming calcium oxalate. This may then be incinerated utilizing particulate collection equipment to collect calcium oxide for recycling.
Properties of Oxalic acid
| Melting point: | 189.5 °C (dec.)(lit.) |
| Boiling point: | 365.1°C (estimate) |
| Density | 0.99 g/mL at 25 °C |
| vapor density | 4.4 (vs air) |
| vapor pressure | <0.01 mm Hg ( 20 °C) |
| refractive index | 1.4261 (estimate) |
| Flash point: | 101-157°C |
| storage temp. | Store below +30°C. |
| solubility | water: soluble108g/L at 25°C |
| form | Liquid |
| pka | 1.23(at 25℃) |
| color | White |
| PH | 3(1 mM solution);2.09(10 mM solution);1.31(100 mM solution); |
| PH Range | 6 - 8 at 25 °C |
| Odor | odorless |
| Water Solubility | 90 g/L (20 ºC) |
| Sublimation | 101-157 ºC |
| Merck | 14,6911 |
| BRN | 385686 |
| Henry's Law Constant | 1.43 at pH 4 (quoted, Gaffney et al., 1987) |
| Exposure limits | NIOSH REL: TWA 1, STEL 2, IDLH 500; OSHA PEL: TWA 1;
ACGIH TLV: TWA 1, STEL 2 (adopted). |
| Stability: | Stable, but moisture sensitive. Incompatible with metals. |
| CAS DataBase Reference | 144-62-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Oxalic acid(144-62-7) |
| EPA Substance Registry System | Oxalic acid (144-62-7) |
Safety information for Oxalic acid
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H318:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P270:Do not eat, drink or smoke when using this product. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Oxalic acid
| InChIKey | MUBZPKHOEPUJKR-UHFFFAOYSA-N |
Oxalic acid manufacturer
New Products
Methyl (R)-1-Boc-4,4-difluoropyrrolidine-2-carboxylate 2,2-Difluoropropylamine hydrochloride tert-butyl 3-bromoazetidine-1-carboxylate (R)-1-Boc-3-hydroxypyrrolidine DIFLUOROACETIC ANHYDRIDE 2,2-Difluoropropionic acid Diallylamine, 99% Calcium hydroxide, 95% Aluminum oxide, basic 2-Bromophenylacetonitrile, 97% L-tert-Leucine,97% N-Hydroxy-2-methylpropanimidamide 4-(3,4-Dichlorophenyl)-3,4-Dihydro-N-Methyl-1-(2H)-Naphthalenimine (Schiff Base) 2-AMINO-3,5-DIBROMO BENZALDEHYDE [ADBA] L-Glutamic Acid Dimethyl Ester Hcl 10-Methoxy-5H-dibenz[b,f]azepine 5-Cyanophthalide N, N-Carbonyldiimidazole (CDI) Dibenzoyl Peroxide Titanium Dioxide 2-(Methylthio) Benzonitrile Sodium Acetate Anhydrous Allopurinol 1,5-DibromopentaneRelated products of tetrahydrofuran

You may like
-
 OXALIC ACID 99%View Details
OXALIC ACID 99%View Details
114-62-7 -
 OXALIC ACID 98%View Details
OXALIC ACID 98%View Details -
 Oxalic acid 99%View Details
Oxalic acid 99%View Details -
 Oxalic Acid 99%View Details
Oxalic Acid 99%View Details -
 Oxalic acid 99%View Details
Oxalic acid 99%View Details -
 Oxalic acid 98%View Details
Oxalic acid 98%View Details -
 Oxalic acid solution CASView Details
Oxalic acid solution CASView Details -
 Oxalic Acid CASView Details
Oxalic Acid CASView Details
