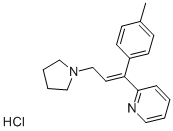
Triprolidine
Synonym(s):Triprolidine hydrochloride monohydrate
- CAS NO.:486-12-4
- Empirical Formula: C19H22N2
- Molecular Weight: 278.39
- MDL number: MFCD00038040
- EINECS: 207-627-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-11-19 23:02:33
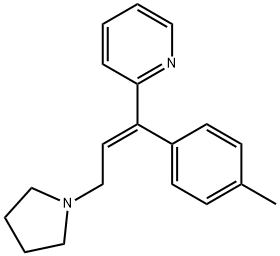
What is Triprolidine?
Absorption
Rapidly absorbed in the intestinal tract.
Toxicity
Symptoms of overdose include drowsiness, weakness, inco-ordination, difficulty with micturition, respiratory depression, hypotension, agitation, irritability, convulsions, hypertension, palpitation and tachycardia.
Originator
Actidil,Burroughs-Wellcome,US,1958
The Uses of Triprolidine
Antihistaminic.
Indications
For the symptomatic relief of seasonal or perennial allergic rhinitis or nonallergic rhinitis; allergic conjunctivitis; and mild, uncomplicated allergic skin manifestations of urticaria and angioedema. Also used in combination with other agents for the symptomatic relief of symptoms associated with the common cold.
Background
First generation histamine H1 antagonist used in allergic rhinitis; asthma; and urticaria. It is a component of cough and cold medicines. It may cause drowsiness.
Definition
ChEBI: An N-alkylpyrrolidine that is acrivastine in which the pyridine ring is lacking the propenoic acid substituent. It is a sedating antihistamine that is used (generally as the monohydrochloride monohydrate) for the relief of the symptoms o uticaria, rhinitis, and various pruritic skin disorders.
Manufacturing Process
4-Methylacetophenone is first reacted with paraformaldehyde and then with
pyrrolidine to give p-methyl-ω-pyrralidinopropiophenone.
Atomized lithium (26 g, 3.75 mols) and sodium-dried ether (200 cc) are
placed in a 3-liter, 3-necked flask fitted with a Herschberg stirrer,
thermometer pocket and a water condenser closed by a calcium chloride tube.
A slow stream of dry nitrogen is blown through the flask, which is cooled to -
10°C and n-butyl chloride (138 g, 156 cc, 1.5 mols) is run in with rapid
stirring; the mixture is stirred for a further 30 minutes, and then cooled to -
60°C
2-Bromopyridine (193 g, 1.22 mols) is then added dropwise over 20 minutes,
the temperature of the reaction mixture being maintained at -50°C. The
mixture is stirred for 10 minutes at -50°C and p-methyl-ω-
pyrrolidinopropiophenone (112.5 g, 0.5 mol) in dry benzene is then added
dropwise over ca 30 minutes, at a temperature of -50°C. The mixture is
stirred for a further 2 hours, the temperature being allowed to rise to -30°C
but no higher.
The mixture is poured onto excess ice, acidified with concentrated
hydrochloric acid, the ether layer separated and extracted with water (1 x 200
cc). The combined aqueous extracts are washed with ether (1 x 200 cc)
basified with 0.880 ammonia and extracted with chloroform (3 x 350 cc); the
extract is washed with water (2 x 100 cc), dried over sodium sulfate,
evaporated, and the residue extracted with boiling light petroleum (BP 60° to
80°C; 10 volumes), filtered hot and evaporated to dryness. The residue is
recrystallized from alcohol to give a cream solid (119 g, 80%), MP 117° to
118°C. Recrystallization gives 1-(4-methylphenyl)-1-(2-pyridyl)-3-
pyrrolidonopropan-1-ol, MP 119° to 120°C.
1-(4-Methylphenyl)-1-(2-pyridyl)-3-pyrrolidinopropan-1-ol (10.0 g) is heated
in a steam bath for 30 minutes with 85% aqueous sulfuric acid (30 cc). The
solution is then poured onto crushed ice, excess of ammonia solution added
and the liberated oil extracted with light petroleum (BP 60° to 80°C). The
extract is dried over anhydrous sodium sulfate and the solvent evaporated to
leave an amber syrup (8.8 g) consisting of the cis and trans isomers of 1-(4-
methylphenyl)-1-(2-pyridyl)-3-pyrrolidinoprop-1-ene as described in US Patent
2,712,023. The isomers may be separated by base exchange chromatography.
The 4-methyl-ω-pyrrolidinopropiophenone required as the starting product for the preparation of the carbinol is prepared by the Mannich reaction (Blicke,
Organic Reactions, 1942, vol 1, p 303; Adamson & Billinghurst, Journal of the
Chemical Society, 1950,1039) from 4-methylacetophenone and pyrrolidine.
The hydrochloride has a MP of 170°C with decomposition.
brand name
Actidil (GlaxoSmithKline); Myidyl (USl).
Therapeutic Function
Antihistaminic
Pharmacokinetics
In allergic reactions an allergen interacts with and cross-links surface IgE antibodies on mast cells and basophils. Once the mast cell-antibody-antigen complex is formed, a complex series of events occurs that eventually leads to cell-degranulation and the release of histamine (and other chemical mediators) from the mast cell or basophil. Once released, histamine can react with local or widespread tissues through histamine receptors. Histamine, acting on H1-receptors, produces pruritis, vasodilatation, hypotension, flushing, headache, tachycardia, and bronchoconstriction. Histamine also increases vascular permeability and potentiates pain. Triprolidine, is a histamine H1 antagonist that competes with histamine for the normal H1-receptor sites on effector cells of the gastrointestinal tract, blood vessels and respiratory tract. It provides effective, temporary relief of sneezing, watery and itchy eyes, and runny nose due to hay fever and other upper respiratory allergies. Triprolidine has anticholinergic and sedative effects.
Metabolism
Not Available
Properties of Triprolidine
| Melting point: | 59-61° |
| Boiling point: | 435.4±34.0 °C(Predicted) |
| Density | 1.061±0.06 g/cm3(Predicted) |
| pka | pKa 4.01(H2O
t = 23.0) (Uncertain);9.69(H2O
t = 23.0) (Uncertain) |
| CAS DataBase Reference | 486-12-4(CAS DataBase Reference) |
| NIST Chemistry Reference | Triprolidine(486-12-4) |
Safety information for Triprolidine
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Triprolidine
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid 1-Bromo-3,5-Di-Tert-Butylbenzene S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran


You may like
-
 Triprolidine CAS 486-12-4View Details
Triprolidine CAS 486-12-4View Details
486-12-4 -
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1