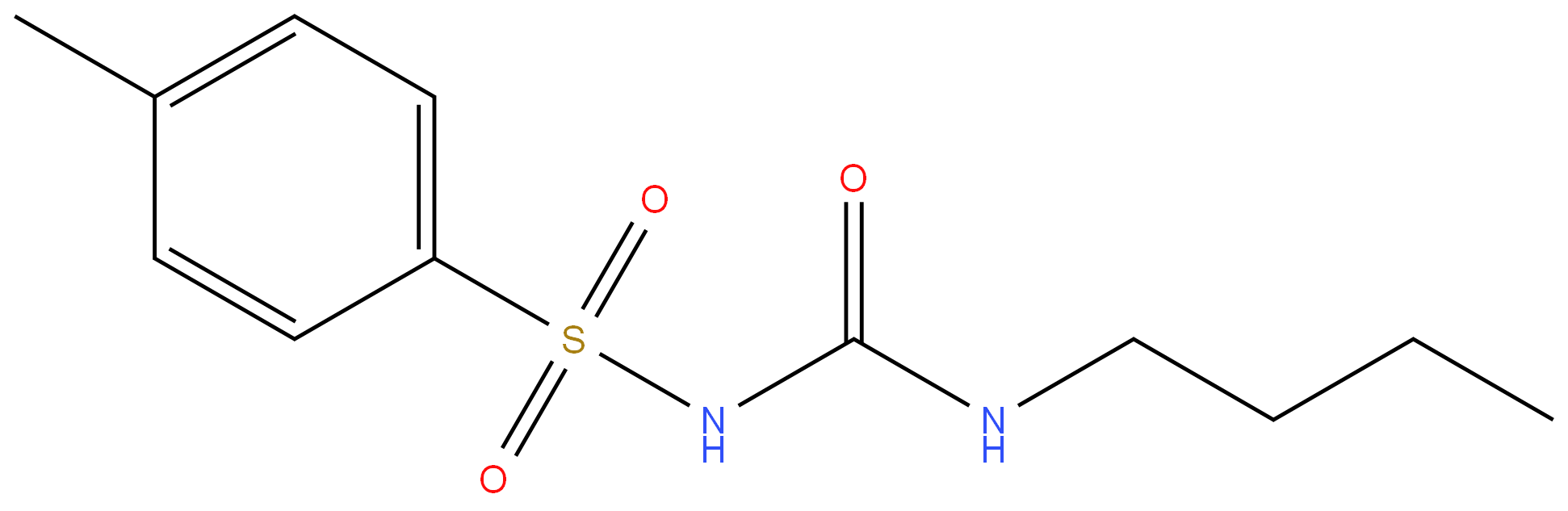
TOLBUTAMIDE
Synonym(s):N-[(Butylamino)carbonyl]-4-methylbenzenesulfonamide;1-Butyl-3-(4-methylphenylsulfonyl)urea;Tolbutamide
- CAS NO.:64-77-7
- Empirical Formula: C12H18N2O3S
- Molecular Weight: 270.35
- MDL number: MFCD00027169
- EINECS: 200-594-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:08:52
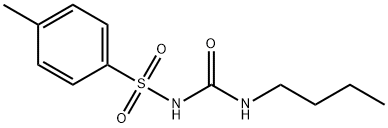
What is TOLBUTAMIDE?
Absorption
Readily absorbed following oral administration. Tolbutamide is detectable in plasma 30-60 minutes following oral administration of a single dose with peak plasma concentrations occurring within 3-5 hours.
Absorption is unaltered if taken with food but is increased with high pH.
Toxicity
Oral, mouse: LD50 = 2600 mg/kg
Chemical properties
White or almost white, crystalline powder.
Originator
Dolipol,Hoechst,France,1956
The Uses of TOLBUTAMIDE
Tolbutamide, have been used in a cDNA microarray assay to probe changes in gene expression in HepG2 cells upon their administration. It has been utilized to counteract insulin activity in a patch-clamp investigation of ATP sensitive K+ channels in mouse pancreatic β-cells. The activity of various biotransformation enzymes in cultured primary rat proximal tubular cells in the presence of tolbutamide and other compounds has been studied.
The Uses of TOLBUTAMIDE
An antidiabetic, used as a hypoglycemic agent in veterinary medicine.
The Uses of TOLBUTAMIDE
It is used for type II diabetes mellitus of medium severity with no expressed microvascular complications.
Background
Tolbutamide is an oral antihyperglycemic agent used for the treatment of non-insulin-dependent diabetes mellitus (NIDDM). It is structurally similar to acetohexamide, chlorpropamide and tolazamide and belongs to the sulfonylurea class of insulin secretagogues, which act by stimulating β cells of the pancreas to release insulin. Sulfonylureas increase both basal insulin secretion and meal-stimulated insulin release. Medications in this class differ in their dose, rate of absorption, duration of action, route of elimination and binding site on their target pancreatic β cell receptor. Sulfonylureas also increase peripheral glucose utilization, decrease hepatic gluconeogenesis and may increase the number and sensitivity of insulin receptors. Sulfonylureas are associated with weight gain, though less so than insulin. Due to their mechanism of action, sulfonylureas may cause hypoglycemia and require consistent food intake to decrease this risk. The risk of hypoglycemia is increased in elderly, debilitated and malnourished individuals. Tolbutamide appears to be metabolized in the liver. Tolbutamide and its metabolites are excreted in urine (75-85%) and feces.
Indications
For treatment of NIDDM (non-insulin-dependent diabetes mellitus) in conjunction with diet and exercise.
What are the applications of Application
Tolbutamide is a hypoglycemic agonist for secretion of islet hormones
Definition
ChEBI: An N-sulfonylurea that consists of 1-butylurea having a tosyl group attached at the 3-position.
Manufacturing Process
50 grams of n-butyl isocyanate are stirred at room temperature into a
suspension of 96 grams of sodium 4-methyl-benzenesulfonamide in 120 cc of
dry nitrobenzene and the whole is then heated for 7 hours at 100°C. After
being cooled, the reaction mixture, which is a thick magma, is diluted with
methylene chloride or ethyl acetate and the sodium salt of the sulfonylurea
formed is separated by centrifuging. The centrifuged crystalline residue freed
from organic solvents is dissolved in 500 to 600 cc of water heated at 50°C
and decolorized with animal charcoal.
The precipitate obtained by acidification with dilute hydrochloric acid is
dissolved in an equivalent quantity of dilute ammonia solution (about 1:20),
again treated with animal charcoal and reprecipitated with dilute hydrochloric
acid. In this manner N-4-methylbenzenesulfonyl-N'-n-butyl-urea is obtained in
analytically pure form in a yield of 70 to 80% of theory. It melts at 125° to
127°C (with decomposition).
Therapeutic Function
Oral hypoglycemic
General Description
White crystals.
General Description
Tolbutamide is N-[(butylamino)carbonyl]-4-methylbenzenesulfonamide;or 1-butyl-3-(p-tolylsulfonyl)urea (Orinase,generic). Orinase Diagnostic was the sodium salt, which isfreely soluble in water for injection, but this product was discontinuedc. 2000.
General Description
Tolbutamide, 1-butyl-3-(p-tolylsulfonyl)urea (Orinase), occurs as a white, crystalline powderthat is insoluble in water and soluble in alcohol or aqueousalkali. It is stable in air.
Tolbutamide is absorbed rapidly in responsive diabetic patients.The blood sugar level reaches a minimum after 5 to8 hours. It is oxidized rapidly in vivo to 1-butyl-3-(p-carboxyphenyl)sulfonylurea, which is inactive. The metabolite isfreely soluble at urinary pH; if the urine is strongly acidified,however, as in the use of sulfosalicylic acid as a protein precipitant,a white precipitate of the free acid may be formed.
Air & Water Reactions
Insoluble in water.
Reactivity Profile
TOLBUTAMIDE is an amide. Amides/imides react with azo and diazo compounds to generate toxic gases. Flammable gases are formed by the reaction of organic amides/imides with strong reducing agents. Amides are very weak bases (weaker than water). Imides are less basic yet and in fact react with strong bases to form salts. That is, they can react as acids. Mixing amides with dehydrating agents such as P2O5 or SOCl2 generates the corresponding nitrile. The combustion of these compounds generates mixed oxides of nitrogen (NOx). TOLBUTAMIDE is incompatible with acids. .
Fire Hazard
Flash point data for TOLBUTAMIDE are not available. TOLBUTAMIDE is probably combustible.
Biological Activity
tolbutamide is a potent inhibitor of camp with an ic50 value of 4mm [1].tolbutamide has been reported to inhibit both the basal and the cyclic amp-stimulated protein kinase activites with an ic50 value of 4mm for cyclic amp-dependent kinase activity. in addition, tolbutamide has been revealed to inhibit both soluble and membrane-bound protein kinase from canine heart. moreover, the tolbutamide inhibition of adipose tissue cyclic amp- dependent protein kinase is explanation for antilipolytic effects [1]. besides, tolbutamide and dbcamp has been exhibited to increase about four-fold levels of cx43 mrna and decrease about 80% the expression of ki-67 [2].
Mechanism of action
Tolbutamide is one of the most widely used antidiabetic agents. Its action is preferably connected with stimulatory action of β-cells in the pancreas, which results in intensive insulin secretion.
Pharmacokinetics
Tolbutamide, a first-generation sulfonylurea antidiabetic agent, is used with diet to lower blood glucose levels in patients with diabetes mellitus type II. Tolbutamide is twice as potent as the related second-generation agent glipizide. Tolbutamide lowers blood sugar by stimulating the pancreas to secrete insulin and helping the body use insulin efficiently. The pancreas must be able to produce insulin for this drug to work.
Clinical Use
Tolbutamide should be used only when the diabetic patientis an adult or shows adult-onset diabetes, and the patientshould adhere to dietary restrictions.
Safety Profile
Moderately toxic by ingestion and several other routes. A human teratogen. Human reproductive effects by ingestion and possibly other routes: stillbirth, developmental abnormalities of the cardlovascular (circulatory) system and urogenital system, and unspecified neonatal effects. Human systemic effects by ingestion: nausea or vomiting, hypoglycemia. Other experimental teratogenic and reproductive effects. Mutation data reported. Implicated in aplastic anemia. When heated to decomposition it emits very toxic fumes of NO, and SOx.
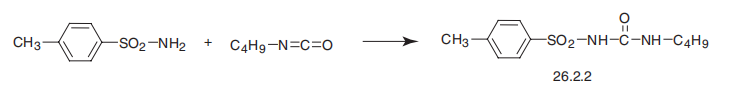
Synthesis
Tolbutamide, 1-butyl-3-p-toluenesulfonylurea (26.2.2), is made in a single step reaction by interaction of p-toluenesulfonylamide (in the form of sodium salt) with butylisocyanate.
Drug interactions
Potentially hazardous interactions with other drugs
Analgesics: effects enhanced by NSAIDs - avoid
with azapropazone.
Antibacterials: effects enhanced by chloramphenicol,
sulphonamides, tetracyclines and trimethoprim;
effect reduced by rifamycins.
Anticoagulants: effect possibly enhanced by
coumarins; also possibly changes to INR.
Antifungals: concentration increased by fluconazole
and miconazole, and possibly voriconazole.
Lipid-regulating drugs: possibly additive
hypoglycaemic effect with fibrates.
Sulfinpyrazone: enhanced effect of sulphonylureas.
Metabolism
Metabolized in the liver principally via oxidation of the p-methyl group producing the carboxyl metabolite, 1-butyl-3-p-carboxyphenylsulfonylurea. May also be metabolized to hydroxytolbutamide. Tolbutamide does not undergo acetylation like antibacterial sulfonamides as it does not have a p-amino group.
Metabolism
Tolbutamide is metabolised in the liver by hydroxylation mediated by the cytochrome P450 isoenzyme CYP2C9. It is excreted in the urine chiefly as inactive metabolites.
References
[1] wray hl, harris aw. adenosine 3', 5'-monophosphate-dependent protein kinase in adipose tissue: inhibition by tolbutamide. biochem biophys res commun. 1973 jul 2;53(1):291-4.
[2] sánchez-alvarez r1, paíno t, herrero-gonzález s, medina jm, tabernero a. tolbutamide reduces glioma cell proliferation by increasing connexin43, which promotes the up-regulation of p21 and p27 and subsequent changes in retinoblastoma phosphorylation. glia. 2006 aug 1;54(2):125-34.
Properties of TOLBUTAMIDE
| Melting point: | 128-130°C |
| Density | 1.2450 |
| refractive index | 1.6360 (estimate) |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | Soluble in chloroform. |
| form | neat |
| pka | pKa 5.32(H2O
t = 37) (Uncertain) |
| form | Solid |
| color | White to Off-White |
| Water Solubility | 0.14g/L(25 ºC) |
| Merck | 14,9507 |
| BRN | 1984428 |
| Specific Activity | 50-60 mCi/mmol |
| Concentration | 0.1 mCi/ml |
| Solvent | Ethanol |
| Stability: | Stable. Combustible. |
| CAS DataBase Reference | 64-77-7(CAS DataBase Reference) |
| EPA Substance Registry System | Tolbutamide (64-77-7) |
Safety information for TOLBUTAMIDE
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H317:Sensitisation, Skin |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P321:Specific treatment (see … on this label). P333+P313:IF SKIN irritation or rash occurs: Get medical advice/attention. |
Computed Descriptors for TOLBUTAMIDE
TOLBUTAMIDE manufacturer
Kothari Phytochemicals and Industries Ltd.
Bihani Chemical Industries Private Limited
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate N-octanoyl benzotriazole 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5,6-Dimethoxyindanone 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran
You may like
-
 64-77-7 Tolbutamide 98%View Details
64-77-7 Tolbutamide 98%View Details
64-77-7 -
 64-77-7 99%View Details
64-77-7 99%View Details
64-77-7 -
 Tolbutamide 98%View Details
Tolbutamide 98%View Details
64-77-7 -
 Tolbutamide CAS 64-77-7View Details
Tolbutamide CAS 64-77-7View Details
64-77-7 -
 Tolbutamide 95% CAS 64-77-7View Details
Tolbutamide 95% CAS 64-77-7View Details
64-77-7 -
 Tolbutamide CAS 64-77-7View Details
Tolbutamide CAS 64-77-7View Details
64-77-7 -
 Tolbutamide CAS 64-77-7View Details
Tolbutamide CAS 64-77-7View Details
64-77-7 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1