Lithium borohydride
Synonym(s):Lithium boron hydride, Lithium boranate;Lithium tetrahydroborate
- CAS NO.:16949-15-8
- Empirical Formula: BH4Li
- Molecular Weight: 21.78
- MDL number: MFCD00011088
- EINECS: 241-021-7
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-01-27 09:38:02
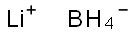
What is Lithium borohydride?
Chemical properties
WHITE POWDER
Physical properties
White orthorhombic crystals; density 0.67 g/cm3; decomposes in moist air; melts at 268°C; decomposes at 380°C; reacts with water; dissolves in ether, tetrahydrofuran, and diethylamine; solubility in ether, 25g/L at 25°C.
The Uses of Lithium borohydride
Strong reducing agent. Used to reduce compounds containing ketones, aldehydes, ester carbonyls, acid chlorides, lactones, and epoxidesLithium borohydride is a versatile reducing agent for aldehydes, ketones, nitriles, primary amides, acid chlorides, lactones, epoxides, and esters. It is involved in the preparation of intercalation compounds and nanocomposites. It is used as a precursor for other borohydrides and acts as a catalyst in hydroboration reactions. It is known to be good reversible storage compound for anhydrous ammonia and hydrogen.
The Uses of Lithium borohydride

To a solution of the SM (300 mg, 0.9 mmol) in THF (15 mL) at -78 C was added dropwise LiBH4 (1.8 mL, 1.8 mmol). The reaction mixture was stirred at -78 C for 5 min, after which time it was quenched with sat aq NH4Cl. The mixture was filtered and the filtrate was extracted with EtOAc (3 x 30 mL). The combined organics were dried and concentrated to provide the product as a yellow solid. [110 mg, 48%]
The Uses of Lithium borohydride
Strong reducing agent. Used in the reduction of Compounds contg ketonic, aldehydic, or ester carbonyls and a nitrile group, where reduction of the carbonyl, but not of the nitrile group, is wanted. In the determination of free carboxyl groups in peptides and proteins; after esterification and acetylation, only the ester groups, and none of the peptide bonds are reduced.
The Uses of Lithium borohydride
Lithium borohydride (LiBH4) is a complex hydride with a high hydrogen density. It is a strong reducing agent and an electrode material. It has a high gravimetric (18.4 wt%) and volumetric (121 kg/m3) hydrogen storage capacities. It can also be used in lithium-ion batteries.
What are the applications of Application
Lithium borohydride is a reducing agent for esters
General Description
A white to grayish crystalline powder.
Air & Water Reactions
Likely to ignite when moistened with water [Lab. Gov. Chemist 1965].
Reactivity Profile
Lithium borohydride is a strong reducing agent. Is easily ignited and burns vigorously once ignited. Reacts on contact with water or acids to form hydrogen gas and corrosive products. Reaction with limited amounts of water or moisture may cause ignition after a delay [Gaylord, 1965, p. 22].
Health Hazard
Inhalation or contact with vapors, substance or decomposition products may cause severe injury or death. May produce corrosive solutions on contact with water. Fire will produce irritating, corrosive and/or toxic gases. Runoff from fire control may cause pollution.
Fire Hazard
Produce flammable gases on contact with water. May ignite on contact with water or moist air. Some react vigorously or explosively on contact with water. May be ignited by heat, sparks or flames. May re-ignite after fire is extinguished. Some are transported in highly flammable liquids. Runoff may create fire or explosion hazard.
Safety Profile
Poison by ingestion, inhalation, and skin contact. Flammable; can liberate H2. Incompatible with H20 as moisture on fibers of cellulose or as liquid. See also LITHIUM, BORON COMPOUNDS, and HYDRIDES.
Purification Methods
It is crystallised from Et2O, and pumped free of ether at 90-100o during 2hours [Schaeffer et al. J Am Chem Soc 78 729 1956]. Store it dry as it decomposes slowly in moist air. [Becher in Handbook of Preparative Inorganic Chemistry (Ed. Brauer) Academic Press Vol I p 775 1963.]
Properties of Lithium borohydride
| Melting point: | 280 °C |
| Boiling point: | 66°C/760mmHg |
| Density | 0.896 g/mL at 25 °C |
| Flash point: | −1 °F |
| storage temp. | water-free area |
| solubility | Soluble in ether, THF, and aliphatic aminesSoluble in ether, tetrahydrofuran, aliphatic amines and ethanol. |
| form | Powder |
| appearance | White solid |
| Specific Gravity | 0.66 |
| color | White |
| explosive limit | 4.00-75.60%(V) |
| Water Solubility | soluble H2O above pH 7, ether, tetrahydrofuran, aliphatic amines [MER06] |
| Sensitive | Air & Moisture Sensitive |
| Merck | 14,5525 |
| CAS DataBase Reference | 16949-15-8(CAS DataBase Reference) |
| NIST Chemistry Reference | Lithium tetrahydroborate(16949-15-8) |
| EPA Substance Registry System | Borate(1-), tetrahydro-, lithium (16949-15-8) |
Safety information for Lithium borohydride
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Corrosion Corrosives GHS05  Skull and Crossbones Acute Toxicity GHS06 |
| GHS Hazard Statements |
H260:Substances And Mixtures Which, In Contact With Water,Emit Flammable Gases H301:Acute toxicity,oral H314:Skin corrosion/irritation |
| Precautionary Statement Codes |
P260:Do not breathe dust/fume/gas/mist/vapours/spray. P280:Wear protective gloves/protective clothing/eye protection/face protection. P231+P232:Handle under inert gas. Protect from moisture. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Lithium borohydride
Lithium borohydride manufacturer
ASM Organics
New Products
3-Iodophenylacetic acid 3-Pyridineacetonitrile, α-hydroxy- 2-Propanamine, 1-chloro-, hydrochloride (9CI) 3-(hexyloxy)-4-(pyridin-3-yl)-1,2,5-thiadiazole 2-Hexyn-1-ol Dibenzo-18-crown-6 Nickel(II) perchlorate hexahydrate, 98% 4-Bromophenylacetonitrile, 95% 3-Bromo-4-fluoroaniline, 97% Sodium tetraborate decahydrate, 98% Palladium(II) acetate, trimer, Pd 99% 4-Bromo-2-chlorotoluene, 97% N N Dimethylformamide Dimethyl Acetal (Dmf Dma) 2,3-Dichloro Benzoyl Cyanide [Side Chain] Bis(2-Chloroethyl) Amine Hydrochloride L-Glutamic Acid Diethyl Ester Hydrochloride 5-(Difluoromethoxy)-2-Mercaptobenzimidazole 1-Ethyl-3-(3-Dimethylaminopropyl)-Carbodiimide Hydrochloride [EDC Hcl] 1,4-Napthoquinone Bromoiodomethane Sodium Bicarbonate Methylene Dichloride (MDC) Ethyl Acetate Indole-3-Carbinol (I3C)Related products of tetrahydrofuran


You may like
-
 Lithium borohydride CAS 16949-15-8View Details
Lithium borohydride CAS 16949-15-8View Details
16949-15-8 -
 Lithium borohydride CAS 16949-15-8View Details
Lithium borohydride CAS 16949-15-8View Details
16949-15-8 -
 Lithium borohydride CAS 16949-15-8View Details
Lithium borohydride CAS 16949-15-8View Details
16949-15-8 -
 Lithium borohydride CAS 16949-15-8View Details
Lithium borohydride CAS 16949-15-8View Details
16949-15-8 -
 Lithium borohydride, ≥95% CAS 16949-15-8View Details
Lithium borohydride, ≥95% CAS 16949-15-8View Details
16949-15-8 -
 Lithium borohydride CAS 16949-15-8View Details
Lithium borohydride CAS 16949-15-8View Details
16949-15-8 -
 Lithium borohydride solution CAS 16949-15-8View Details
Lithium borohydride solution CAS 16949-15-8View Details
16949-15-8 -
 Lithium borohydride solution CAS 16949-15-8View Details
Lithium borohydride solution CAS 16949-15-8View Details
16949-15-8
