Cortisone acetate
Synonym(s):17α,21-Dihydroxy-4-pregnene-3,11,20-trione 21-acetate;21-Acetoxy-4-pregnen-17α-ol-3,11,20-trione;4-Pregnene-17α,21-diol-3,11,20-trione 21-acetate;Cortisone 21-acetate;Cortisone acetate
- CAS NO.:50-04-4
- Empirical Formula: C23H30O6
- Molecular Weight: 402.48
- MDL number: MFCD00003609
- EINECS: 200-006-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-01-27 09:38:02
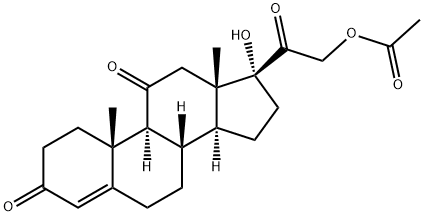
What is Cortisone acetate?
Toxicity
Data regarding acute overdoses of glucocorticoids are rare. Chronic high doses of glucocorticoids can lead to the development of cataract, glaucoma, hypertension, water retention, hyperlipidemia, peptic ulcer, pancreatitis, myopathy, osteoporosis, mood changes, psychosis, dermal atrophy, allergy, acne, hypertrichosis, immune suppression, decreased resistance to infection, moon face, hyperglycemia, hypocalcemia, hypophosphatemia, metabolic acidosis, growth suppression, and secondary adrenal insufficiency. Overdose may be treated by adjusting the dose or stopping the corticosteroid as well as initiating symptomatic and supportive treatment.
Description
Cortisone acetate is a synthetic glucocorticoid with anti-inflammatory properties. It decreases the size of Bacillus Calmette-Guérin (BCG) vaccine-induced dermal lesions and tuberculin reactions in rabbits when administered at 2 mg/kg on alternate days over the course of 46 days. It also reduces the number and percentage of activated lesion-infiltrating mononuclear cells and decreases the amount of caseous necrosis and ulceration. Cortisone acetate (2.5 mg/kg per day, s.c.) slows tissue regeneration in a rabbit model of wound healing. It also decreases the number of dexamethasone binding sites on isolated human lymphocytes by 30%. Formulations containing cortisone acetate have been used to relieve inflammation, pruritic manifestations of corticosteroid-responsive dermatoses, and in the treatment of immune and allergic disorders.
Chemical properties
White needle crystal or crystalline powder (acetone), odorless. Stable in the air. Mp239- 241°C, specific rotation [α]20D+164°(0.5%, acetone), [α]23D+169°(chloroform), [α]25D+208°-+217°(dioxane), The ethanol solution of this product has a maximum absorption at a wavelength of 240nm. Soluble in chloroform (1:4), soluble in acetone (1:75), slightly soluble in ethanol and ether, and hardly soluble in water. This product dissolves in sulfuric acid and turns yellow without fluorescence (different from hydrocortisone acetate).
Originator
Cortone Acetate,MSD,US,1950
The Uses of Cortisone acetate
Cortisone Acetate is a glucocorticoid. Cortisone Acetate is an antiinflammatory agent. Cortisone Acetate is bioavailable and readily converted to the therapeutically active form, Hydrocotisone (H71461 5).
The Uses of Cortisone acetate
Cortisone Acetate (Hydrocortisone Acetate EP Impurity D) is a glucocorticoid. Cortisone Acetate is an antiinflammatory agent. Cortisone Acetate is bioavailable and readily converted to the therapeutically active form, Hydrocotisone.
What are the applications of Application
Cortisone 21-acetate is a glucocorticoid and an antiinflammatory agent
Background
Cortisone acetate was first isolate in 1935 and became more widely researched in 1949. Since then, glucocorticoids such as cortisone acetate have been used to treat a number of inflammatory conditions such as endocrine, rheumatic, collagen, dermatologic, allergic, ophthalmic, respiratory, hematologic, neoplastic, edematous, and gastrointestinal diseases and disorders.
Cortisone acetate was granted FDA approval on 13 June 1950.
Indications
Cortisone acetate is indicated to treat a wide variety of endocrine, rheumatic, collagen, dermatologic, allergic, ophthalmic, respiratory, hematologic, neoplastic, edematous, and gastrointestinal diseases and disorders.
Definition
ChEBI: Cortisone acetate is a corticosteroid hormone.
Preparation
Cortisone acetate is prepared from dehydropregnenolone by epoxidation, Wolff's oxidation, mycoxidation, chromic acid oxidation, hydrogen bromide ring-opening, Raney's nickel-catalyzed debromination, iodination, and acetyloxy substitution.
Therapeutic Function
Glucocorticoid
General Description
Cortisone acetate, 21-(acetyloxy)-17-hydroxypregn-4-ene-3,11,20-trione, is the 21-acetate of naturally occurring cortisone with good systemicanti-inflammatory activity and low-to-moderate salt-retentionactivity after its in vivo conversion to hydrocortisoneacetate. This conversion is mediated by 11β-hydroxysteroiddehydrogenase. It is used for the entire spectrum of uses discussedpreviously under the heading, “Therapeutic Uses ofAdrenal Cortex Hormones”—collagen diseases, Addisondisease, severe shock, allergic conditions, chronic lymphocyticleukemia, and many other indications. Cortisone acetateis relatively ineffective topically, mainly because itmust be reduced in vivo to hydrocortisone. Its plasma halflifeis only about 30 minutes, compared with 90 minutes to3 hours for hydrocortisone.
Pharmacokinetics
Corticosteroids bind to the glucocorticoid receptor, inhibiting pro-inflammatory signals, and promoting anti-inflammatory signals. The duration of action is moderate as it is generally given once daily. Corticosteroids have a wide therapeutic window as patients may require doses that are multiples of what the body naturally produces. Patients taking corticosteroids should be counselled regarding the risk of hypothalamic-pituitary-adrenal axis suppression and increased susceptibility to infections.
Metabolism
Not Available
Purification Methods
Crystallise -1cortisone-21-acetate from acetone or CHCl3. The UV has 15,800 M-1cm at 238nm in dioxane. [Sarett J Biol Chem 162 601 1946, Beilstein 8 III 4058, 5 IV 3481.]
References
1. Cortisone acetate in skin disease; local effect in the skin from topical application and local injection. DOI:10.1001/ARCHDERM.1952.01530210056007
2. McCue, R.E., Dannenberg, A.M., Jr., Huguchi, S., et al. The effect of cortisone on the accumulation, activation, and necrosis of macrophages in tuberculous lesions. Inflammation 3(2), 159-176 (1978). DOI:10.1007/BF00910737
3. Joseph, J., and Tydd, M. The effects of cortisone acetate on tissue regeneration in the rabbit’s ear. J. Anat. 115(Pt. 3), 445-460 (1973).
4. Schlechte, J.A., Ginsberg, B.H., and Sherman, B.M. Regulation of the glucocorticoid receptor in human lymphocytes. J. Steroid Biochem. 16(1), 69-74 (1982). DOI:10.1016/0022-4731(82)90145-5
5. Sittig's Pharmaceutical Manufacturing Encyclopedia
6. Textbook of organic medicinal and pharmaceutical chemistry. DOI:10.1002/jps.3030451120
7. Purification of Laboratory Chemicals DOI:10.5860/choice.50-6768
Properties of Cortisone acetate
| Melting point: | 237-240 °C(lit.) |
| Boiling point: | 577.3±50.0 °C(Predicted) |
| alpha | D25 +164° (c = 0.5 in acetone); D25 +208 to +217° (dioxane) |
| Density | 1.26±0.1 g/cm3(Predicted) |
| refractive index | 212 ° (C=1, MeOH) |
| storage temp. | 2-8°C |
| solubility | Practically insoluble in water, freely soluble in methylene chloride, soluble in dioxan, sparingly soluble in acetone, slightly soluble in ethanol (96 per cent) and in methanol. |
| form | neat |
| pka | 12.32±0.60(Predicted) |
| form | Solid |
| color | White to Off-White |
| Water Solubility | 19mg/L(25 ºC) |
| Merck | 14,2539 |
| BRN | 2067543 |
| Stability: | Stable. Incompatible with strong oxidizing agents. |
| CAS DataBase Reference | 50-04-4(CAS DataBase Reference) |
| EPA Substance Registry System | Cortisone acetate (50-04-4) |
Safety information for Cortisone acetate
Computed Descriptors for Cortisone acetate
Cortisone acetate manufacturer
Allmpus Laboratories Pvt Ltd
New Products
1-Boc-4-cyanopiperidine tert-Butyl carbazate 1-(TERT-BUTOXYCARBONYL)-2-PYRROLIDINONE TETRABUTYLAMMONIUM CYANIDE TETRAHYDRO-2H-PYRAN-3-OL 3-Pyridineacrylic acid Nickel(II) perchlorate hexahydrate, 98% 4-Bromophenylacetonitrile, 95% 3-Bromo-4-fluoroaniline, 97% Sodium tetraborate decahydrate, 98% Palladium(II) acetate, trimer, Pd 99% 4-Bromo-2-chlorotoluene, 97% Tadalafil Clopidogrel bisulfate Sitagliptin Phosphate Monohydrate Cabergoline Fexofinadine HCl Etoricoxib 4-Amino Acetophenone 2-Chloro Acetophenone Amlodipine Base 2,3,5-Triiodobenzoic Acid Pyrrolidine Diiodo PentoxideRelated products of tetrahydrofuran




You may like
-
 Hydrocortisone Aceate EP Impurity D 50-04-4 95.8View Details
Hydrocortisone Aceate EP Impurity D 50-04-4 95.8View Details
50-04-4 -
 HYDROCORTISONE ACETATE EP IMPURITY D 97.62View Details
HYDROCORTISONE ACETATE EP IMPURITY D 97.62View Details
50-04-4 -
 50-04-4 HYDROCORTISONE ACETATE EP IMPURITY D 98View Details
50-04-4 HYDROCORTISONE ACETATE EP IMPURITY D 98View Details
50-04-4 -
 Cortisone acetate CAS 50-04-4View Details
Cortisone acetate CAS 50-04-4View Details
50-04-4 -
 Cortisone acetate 99% (HPLC) CAS 50-04-4View Details
Cortisone acetate 99% (HPLC) CAS 50-04-4View Details
50-04-4 -
 366789-02-8 Riveroxaban 98%View Details
366789-02-8 Riveroxaban 98%View Details
366789-02-8 -
 Carvedilol 98%View Details
Carvedilol 98%View Details
72956-09-3 -
 73590-58-6 Omeprazole 98%View Details
73590-58-6 Omeprazole 98%View Details
73590-58-6
