Sodium acetate
Synonym(s):Acetic acid sodium salt;Sodium acetate;Sodium acetate buffer 3M;Sodium Acetate, Anhydrous, Molecular Biology Grade - CAS 127-09-3 - Calbiochem
- CAS NO.:127-09-3
- Empirical Formula: C2H3NaO2
- Molecular Weight: 82.03379
- MDL number: MFCD00012459
- EINECS: 204-823-8
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-26 18:10:20
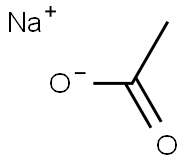
What is Sodium acetate?
Absorption
It is readily available in the circulation after IV administration.
Toxicity
LD50: 25956 mg/kg (Rat.)
Description
Sodium acetate (CH?COONa), the sodium salt of acetic acid, is a colorless deliquescent solid with broad applications. Industrially, it is used to neutralize sulfuric acid waste streams in textiles and as a photoresist for aniline dyes, serves as a concrete sealant to prevent water damage, acts as a seasoning in food, and functions as a buffer solution in laboratories. It is also employed in heating pads, hand warmers, and "hot ice" experiments. Laboratory synthesis involves reactions between acetates and sodium carbonate, bicarbonate, or hydroxide, while industrial production utilizes glacial acetic acid and sodium hydroxide.
Chemical properties
Sodium acetate (CH?COONa, often abbreviated as NaOAc), the sodium salt of acetic acid, is commonly produced by reacting acetic acid with sodium carbonate. This colorless salt is highly soluble in water but less so in alcohol, and finds broad applications, such as a pH modifier in toning baths.
Physical properties
Anhydrous sodium acetate is a colorless crystalline solid with a density of 1.528 g/cm3 and a melting point of 324°C. It is highly soluble in water and moderately soluble in ethanol. The trihydrate form is also a colorless crystal, with a density of 1.45 g/cm3; it decomposes at 58°C, is extremely soluble in water, has a pH of 8.9 for a 0.1M aqueous solution, and is moderately soluble in ethanol at 5.3 g/100mL.
The Uses of Sodium acetate
Sodium acetate is used as a mordant in dyeing and finds applications in photography, food additives, glucose purification, meat preservation, tanning, and as a dehydrating agent. In analytical chemistry, it is employed to prepare buffer solutions. In the food industry, it serves as a preservative for processed meats, often combined with other acid-based preservatives such as lactates and propionates at typical inclusion levels of 0.2–0.5%, and is also used in salad dressings and ready-to-eat meals.
What are the applications of Application
Sodium acetate (NaOAc) is the sodium salt of acetic acid and is generally used in organic chemistry as a weak base. Potassium acetate (KOAc) is commonly used than sodium acetate, usually as a base in palladium-catalyzed reactions. Sodium Acetate, Anhydrous is for precipitation of nucleic acids and preparation of gel stains for protein gel electrophoresis. In addition, Sodium acetate buffer solution is 3 M, non-sterile with pH 5.2±0.1; 0.2 μm filtered, and may be used in various molecular biology applications.
Background
Sodium Acetate is chemically designated CH3COONa, a hygroscopic powder soluble in water. Sodium acetate could be used as an additive in food, industry, concrete manufacturing, heating pads, and buffer solutions. Sodium is the principal cation of extracellular fluid. It comprises more than 90% of total cations at its normal plasma concentration of approximately 140 mEq/liter. The sodium ion exerts a primary role in controlling total body water and its distribution. Acetate ions act as hydrogen ion acceptors, an alternative to bicarbonate. Medically, sodium acetate is an important component as an electrolyte replenisher when given intravenously. It is mainly indicated to correct sodium levels in hyponatremic patients. It can also be used in metabolic acidosis and urine alkalinization.
Indications
Injection, USP 40 mEq is indicated as a source of sodium, for addition to large volume intravenous fluids to prevent or correct hyponatremia in patients with restricted or no oral intake. It is also useful as an additive for preparing specific intravenous fluid formulas when the needs of the patient cannot be met by standard electrolyte or nutrient solutions. Sodium acetate and other bicarbonate precursors are alkalinising agents, and can be used to correct metabolic acidosis, or for alkalinisation of the urine.
Preparation
Sodium acetate is produced by reacting sodium hydroxide or sodium carbonate with acetic acid in an aqueous solution, followed by evaporation to obtain hydrated crystals.
The chemical equations are as follows:
NaOH + CH?COOH → CH?COONa + H?O
Na?CO? + CH?COOH → 2CH?COONa + CO? + H?O
Metabolism
In liver, sodium acetate is being metabolized into bicarbonate. To form bicarbonate, acetate is slowly hydrolyzed to carbon dioxide and water, which are then converted to bicarbonate by the addition of a hydrogen ion.
Properties of Sodium acetate
| Melting point: | >300 °C (dec.)(lit.) |
| Density | 1.01 g/mL at 20 °C |
| refractive index | 1.4640 |
| FEMA | 3024 | SODIUM ACETATE |
| Flash point: | >250 °C |
| storage temp. | Store at +5°C to +30°C. |
| solubility | H2O: 3 M at 20 °C, clear, colorless |
| form | powder |
| appearance | White powder |
| pka | 4.756[at 20 ℃] |
| Specific Gravity | 1.45 |
| color | white |
| Odor | Slight acetic acid |
| PH | 7.87(1 mM solution);8.33(10 mM solution);8.75(100 mM solution);9.04(1000 mM solution) |
| PH Range | 8.5 - 9.9 at 246 g/l at 25 °C |
| Water Solubility | 500 g/L (20 ºC) |
| Sensitive | Hygroscopic |
| Hydrolytic Sensitivity | 0: forms stable aqueous solutions |
| λmax | λ: 260 nm Amax: 0.03 λ: 280 nm Amax: 0.02 |
| Merck | 14,8571 |
| Boiling point: | >400°C(decomposition) |
| BRN | 3595639 |
| Stability: | Stable. Incompatible with strong oxidizing agents, halogens. Moisture sensitive. |
| CAS DataBase Reference | 127-09-3(CAS DataBase Reference) |
| NIST Chemistry Reference | Sodium ethanoate(127-09-3) |
| EPA Substance Registry System | Sodium acetate (127-09-3) |
Safety information for Sodium acetate
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H320:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P337+P313:IF eye irritation persists: Get medical advice/attention. |
Computed Descriptors for Sodium acetate
| InChIKey | VMHLLURERBWHNL-UHFFFAOYSA-M |
Sodium acetate manufacturer
JSK Chemicals
CEFA CILINAS BIOTICS PVT LTD
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran


You may like
-
 SODIUM ACETATE 99%View Details
SODIUM ACETATE 99%View Details -
 Sodium Acetate 99%View Details
Sodium Acetate 99%View Details -
 Sodium Acetate Anhydrous 99%View Details
Sodium Acetate Anhydrous 99%View Details -
 Sodium acetate anhydrous 99%View Details
Sodium acetate anhydrous 99%View Details -
 Sodium acetate 99%View Details
Sodium acetate 99%View Details -
 Sodium acetate 98%View Details
Sodium acetate 98%View Details -
 Sodium acetate, anhydrous, 98% 99%View Details
Sodium acetate, anhydrous, 98% 99%View Details -
 sodium acetate CASView Details
sodium acetate CASView Details
