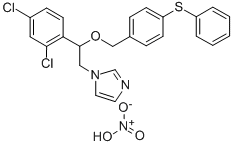
Butoconazole nitrate
Synonym(s):Butoconazole nitrate;Gynazole-1;Gynomyk;Exelgyn;Femstat One
- CAS NO.:64872-77-1
- Empirical Formula: C19H18Cl3N3O3S
- Molecular Weight: 474.79
- MDL number: MFCD00058159
- EINECS: 613-721-6
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-11-19 23:02:33
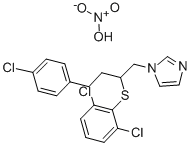
What is Butoconazole nitrate?
Description
Butoconazole Nitrate is an antifungal imidazole antimycotics. It is useful in the topical treatment of vulvovaginal candidiasis, being similar in effectiveness to miconazole and clotrimazole.
Description
Butoconazole is an imidazole antifungal agent that completely inhibits growth of a variety of fungi in vitro when used at concentrations of 0.05-30 μg/ml. It is active against vaginal infections of pure and mixed strains of C. albicans in mice when used vaginally at concentrations of 0.25-2% and 0.1-1%, respectively. Formulations containing butoconazole have been used in the treatment of vaginal yeast infections.
Chemical properties
White Solid
Originator
Syntex (USA)
The Uses of Butoconazole nitrate
Imidazole derivative with antifungal properties
What are the applications of Application
Butoconazole nitrate is an antifungal imidazole compound
Definition
ChEBI: An organic nitrate salt obtained by reaction of equimolar amounts of butaconazole and nitric acid. An antifungal agent, it is used in gynaecology for treatment of vulvovaginal infections caused by Candida species, particularly Candida a bicans.
Manufacturing Process
1H-Imidazole, 1-(4-(4-chlorophenyl)-2-((2,6-dichlorophenyl)thio)butyl)-, (+/-
)-, mononitrate may be prepared by the same way as described below for
enantiomers.
To a solution of Li2CuCl4 (0.10 M, 8.8 ml, 0.88 mmol) in dry THF (75 ml) was
added dropwise a solution of 4-chloromagnesium chloride (17.5 mmol) in
ether (15 ml) at -35°C. After stirring for 45 min, a pre-cooled (-35°C) solution
of (S)-(+)-glycydil tosylate (2.0 g, 8.8 mmol) in THF (5 ml) was added via
syringe. After 2 h at -35°C, the mixture was quenched with saturated NH4Cl
and extracted with ether. The organic layer was dried (Na2SO4), evaporated to
dryness and purificated by flash chromatography affording (2S)-1-(p-toluenesulphonyloxy)-4-(4-chlorophenyl)butan-2-ol, as a white solid (1.9 g,
77%), m.p. 72.7-74°C.
(2S)-1-[2-Hydroxy-4-(4-chlorophenyl)butyl]-1H-imidazole was prepared as
follows: to a solution of imidasole (0.52 g, 7.61 mmol) in dry DMF (5 ml) at
0°C under N2 was added NaH (60% dispersion in oil, 0.3 g, 7.61 mmol). The
reaction mixture was warmed to room temperature and stirred for 30 min. A
solution of (2S)-1-(p-toluenesulphonyloxy)-4-(4chlorophenyl)butan-2-ol in
DMF (15 ml) was then added dropwise over 15 min. The reaction mixture was
heated at 75°C for 24 h, cooled poured into water and extracted with ethyl
acetate. The organic phase was dried and evaporated and was the residue
purified by flash chromatography. Gradient elution (1-5% MeOH/CH2Cl2)
afforded (2S)-1-[2-hydroxy-4-(4-chlorophenyl)butyl]-1H-imidazole which was
recrystallized from EtOAc/Et2O (236 mg, 67%), m.p. 128°-131°C; [α]D25-
23.23 (c 0.4, CHCl3).
(2S)-1-[2-Hydroxy-4-(4-chlorophenyl)butyl]-1H-imidazole nitrate was
prepared as follows. To a solution of (2S)-1-(p-toluenesulphonyloxy)-4-(4-
chlorophenyl)butan-2-ol (250 mg, 1 mmol) in THF (5 ml) at 0°C was added
triethylamine (0.28 ml, 2.0 mmol), followed by methanesulfonyl chloride (0,15
ml, 2.0 mmol). The reaction mixture was was warmed to room temperature
and stirred for 1 h. The mixture was poured into aq. NaHCO3, extracted with
EtOAC, and the organic phase dried and evaporated to dryness. The resulting
mesylate was dissolved in acetone (50 ml), then 2,6-dichlorobenzenethiol(464 mg, 2.6 mmol) and K2CO3 (568 mg, 4.1 mmol) were added. The mixture
was heated at reflux under N2, cooled to room temperature, evaporated to
dryness and partionated between water and EtOAc. The organic phase was
dried (Na2SO4), evaporated, and residue purified by flash chromatography (1-
2% MeOH/CH2Cl2 gradient elution) to give an oil which was converted to
nitrate salt. Recrystallization from EtOAc/Et2O gave 260 mg (55%) (2S)-1-[2-
hydroxy-4-(4-chlorophenyl)butyl]-1H-imidazole nitrate, m.p. 120°-124°C;
[α]D25+22.68 (c 0.4, EtOH).
The R enantiomer was prepared the same way from (-)-glycidyl tosylate.
brand name
Femstat (Roche);Femstat 3 (Bayer); Gynazole-1 (KV Pharmaceutical).
Therapeutic Function
Antifungal
Clinical Use
1-[4-(4-Chlorophenyl)-2-[(2,6-dichlorophenyl)-thio]butyl]-1H-imidazole (Femstat) is an extremely broad-spectrum antifungaldrug that is specifically effective against C. albicans.It is supplied as a vaginal cream containing 2% of thesalt. It is intended for the treatment of vaginal candidiasis.
Properties of Butoconazole nitrate
| Melting point: | 159°C (dec.) |
| storage temp. | Sealed in dry,2-8°C |
| solubility | DMSO or DMF: soluble |
| form | powder |
| color | White to Off-White |
| Merck | 14,1529 |
| CAS DataBase Reference | 64872-77-1(CAS DataBase Reference) |
Safety information for Butoconazole nitrate
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral |
Computed Descriptors for Butoconazole nitrate
| InChIKey | ZHPWRQIPPNZNML-UHFFFAOYSA-N |
Butoconazole nitrate manufacturer
BDR Pharmaceuticals International Pvt Ltd
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid 1-Bromo-3,5-Di-Tert-Butylbenzene S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran

You may like
-
 64872-77-1 Butoconazole nitrate 98%View Details
64872-77-1 Butoconazole nitrate 98%View Details
64872-77-1 -
 Butoconazole Nitrate CAS 64872-77-1View Details
Butoconazole Nitrate CAS 64872-77-1View Details
64872-77-1 -
 Butoconazole nitrate 95% CAS 64872-77-1View Details
Butoconazole nitrate 95% CAS 64872-77-1View Details
64872-77-1 -
 Butoconazole nitrate CAS 64872-77-1View Details
Butoconazole nitrate CAS 64872-77-1View Details
64872-77-1 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1