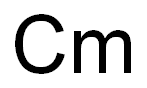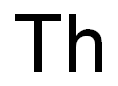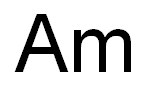actinium
- Empirical Formula: C2H4O
- Molecular Weight: 44.05256

What is actinium?
Physical properties
Actinium is an extremely radioactive, silvery-white, heavy metal that glows in the dark withan eerie bluish light. It decays rapidly which makes it difficult to study, given that it changesinto thorium and francium through electron capture and alpha decay. Its melting point is1,051°C, its boiling point is 3,198°C, and its density is 10.07g/cm3.
Isotopes
There are a total of 35 isotopes of actinium, none of which are stable. All areradioactive, and none exist in the Earth’s crust in any large amounts, although a few canbe extracted from large quantities of pitchblende and other minerals. All are extremelyscarce. Those produced artificially in nuclear reactors, cyclotrons, or linear acceleratorshave relatively short half-lives, ranging from 69 nanoseconds to 21 years.
Origin of Name
The name “actinium” is derived from the Greek word(s) aktis or aktinos, meaning “beam” or “ray.”
Occurrence
Actinium is a rare element that is found in very small amounts in uranium ore (pitchblende),making it difficult and expensive to extract even a small quantity. It is less expensiveand easier to produce small amounts by bombarding the element radium with neutrons in anuclear reactor. Actinium has few commercial uses.
History
Its presence in air was suspected by Cavendish in 1785, discovered by Lord Rayleigh and Sir William Ramsay in 1894. The gas is prepared by fractionation of liquid air, the atmosphere containing 0.94% argon. The atmosphere of Mars contains 1.6% of 40Ar and 5 p.p.m. of 36Ar. Argon is two and one half times as soluble in water as nitrogen, having about the same solubility as oxygen. It is recognized by the characteristic lines in the red end of the spectrum. It is used in electric light bulbs and in fluorescent tubes at a pressure of about 400 Pa, and in filling photo tubes, glow tubes, etc. Argon is also used as an inert gas shield for arc welding and cutting, as a blanket for the production of titanium and other reactive elements, and as a protective atmosphere for growing silicon and germanium crystals. Argon is colorless and odorless, both as a gas and liquid. It is available in high-purity form. Commercial argon is available at a cost of about 3¢ per cubic foot. Argon is considered to be a very inert gas and is not known to form true chemical compounds, as do krypton, xenon, and radon. However, it does form a hydrate having a dissociation pressure of 105 atm at 0°C. Ion molecules such as (ArKr)+, (ArXe)+, (NeAr)+ have been observed spectroscopically. Argon also forms a clathrate with β-hydroquinone. This clathrate is stable and can be stored for a considerable time, but a true chemical bond does not exist. Van der Waals’ forces act to hold the argon. In August 2000, researchers at the University of Helsinki, Finland reported they made a new argon compound HArF 4-4 The Elements by shining UV light on frozen argon that contained a small amount of HF. Naturally occurring argon is a mixture of three isotopes. Seventeen other radioactive isotopes are now known to exist. Commercial argon is priced at about $70/300 cu. ft. or 8.5 cu. meters.
Characteristics
Actinium is the last (bottom) member of group 3 (IIIB) of elements in the periodic tableand the first of the actinide series of metallic elements that share similar chemical and physicalcharacteristics. Actinium is also closely related in its characteristics to the element lanthanum,which is located just above it in group 3. The elements in this series range from atomic number89 (actinium) through 103 (lawrencium). Actinium’s most stable isotope is actinium-227,with a half-life of about 22 years. It decays into Fr-223 by alpha decay and Th-227 throughbeta decay, and both of these isotopes are decay products from uranium-235.
The Uses of actinium
There are no significant uses for actinium because of its scarcity and the expense of producingit. The only practical use for small amounts of actinium is as a tracer in medicine andindustry. It is too difficult to produce in substantial quantities to make it useful. Actiniumcan be used as a source of neutrons to bombard other elements to produce isotopes of thoseelements, but other neutron sources are less expensive.
Definition
A soft silverywhite
radioactive metallic element that is
the first member of the actinoid series. It
occurs in minute quantities in uranium ores and the metal can be obtained by reducing
the trifluoride with lithium. It can
be produced by neutron bombardment of
radium and is used as a source of alpha
particles. The metal glows in the dark; it reacts
with water to produce hydrogen.
Symbol: Ac; m.p. 1050±50°C; b.p.
3200±300°C; r.d. 10.06 (20°C); p.n. 89;
most stable isotope 227Ac (half-life 21.77
years).
Definition
actinium: Symbol Ac. A silveryradioactive metallic element belongingto group 3 (formerly IIIA) of theperiodic table; a.n. 89; mass numberof most stable isotope 227 (half-life21.7 years); m.p. 1050 ± 50°C; b.p.3200°C (estimated). Actinium–227 occursin natural uranium to an extentof about 0.715%. Actinium-228 (halflife6.13 hours) also occurs in nature.There are 22 other artificial isotopes,all radioactive and all with very shorthalf-lives. Its chemistry is similar tothat of lanthanum. Its main use is asa source of alpha particles. The elementwas discovered by A. Debiernein 1899.
Hazard
Most of the radioactive isotopes of actinium pose an extreme radiation hazard. They arebone-seeking radioactive poisons.
Safety information for actinium
New Products
Tert-butyl bis(2-chloroethyl)carbamate 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid DIETHYL AMINOMALONATE HYDROCHLORIDE 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) N-METHYL INDAZOLE-3-CARBOXYLIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran
You may like
-
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1 -
 733039-20-8 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 98+View Details
733039-20-8 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 98+View Details
733039-20-8