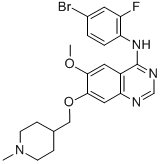
Vandetanib
- CAS NO.:443913-73-3
- Empirical Formula: C22H24BrFN4O2
- Molecular Weight: 475.35
- MDL number: MFCD07772346
- EINECS: 669-841-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-23 21:30:31
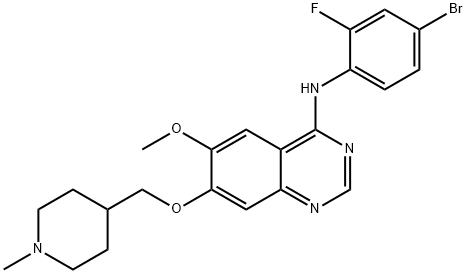
What is Vandetanib ?
Absorption
Slow- peak plasma concentrations reached at a median 6 hours. On multiple dosing, Vandetanib accumulates about 8 fold with steady state reached after around 3 months.
Description
In April 2011, the U.S. FDA approved vandetanib (ZD6474) for the treatment of symptomatic or progressive medullary thyroid cancer (MTC) in adult patients with inoperable advanced ormetastatic disease. Vandetanib inhibits KDR/VEGFR2, VEGFR3, EGFR, and RET kinases with IC50's of 40, 110, 500, and <100 nM, respectively. In athymic mice bearing MTC tumors, a 14.5-fold reduction of tumor volume was observed after 45 days of treatment with vandetanib at 50 mg/kg/day. The decrease in tumor volume was accompanied by decreases in mitotic index (Ki67) and tumor angiogenesis in treated xenografts. Key steps in the synthesis of vandetanib include the displacement of the chlorine atom from 7-benzyloxy-4-chloro-6-methoxyquinazoline with 4-bromo-2- fluoroaniline under acidic conditions in a protic solvent and a Mitsunobu reaction of a N-protected piperidine alcohol with a phenol.
Chemical properties
Yellow Solid
Originator
Astra Zeneca (United Kingdom)
The Uses of Vandetanib
Vandetanib (Zactima, ZD6474) is a potent inhibitor of VEGFR2 with IC50 of 40 nM.
The Uses of Vandetanib
Vandetanib (ZD6474) is a potent inhibitor of VEGFR2 with IC50 of 40 nM
The Uses of Vandetanib
Vandetanib is a once-daily oral inhibitor of vascular endothelial growth factor receptor-2 and epidermal growth factor receptor kinase activity. The activity of Vandetanib plus Docetaxel was assessed in patients with previously treated non-small-cell lung cancer (NSCLC).
The Uses of Vandetanib
Vandetanib is a broad spectrum, orally available kinase inhibitor that targets primarily tyrosine kinases, including vascular endothelial growth factor receptor (VEGFR) and epidermal growth factor receptor (EGFR), with IC50 values in the nanomolar range. It also potently blocks non-receptor tyrosine kinases, including ABL, RET, and SRC, as well as several serine/threonine kinases. Primarily because of its effects on receptor tyrosine kinases like VEGFR and EGFR, vandetanib inhibits angiogenesis, cell growth, and metastasis and is effective against certain forms of cancer.[Cayman Chemical]
What are the applications of Application
Vandetanib is an antagonist of VEGFR (Flk and Flk) and EGFR families.
Background
Vandetanib is an oral once-daily kinase inhibitor of tumour angiogenesis and tumour cell proliferation with the potential for use in a broad range of tumour types.
On April 6 2011, vandetanib was approved by the FDA to treat nonresectable, locally advanced, or metastatic medullary thyroid cancer in adult patients.
Indications
Vandetanib is currently approved as an alternative to local therapies for both unresectable and disseminated disease. Because Vandetanib can prolong the Q-T interval, it is contraindicated for use in patients with serious cardiac complications such as congenital long QT syndrome and uncompensated heart failure.
Definition
ChEBI: Vandetanib is a quinazoline that is 7-[(1-methylpiperidin-4-yl)methoxy]quinazoline bearing additional methoxy and 4-bromo-2-fluorophenylamino substituents at positions 6 and 4 respectively. Used for the treatment of symptomatic or progressive medullary thyroid cancer in patients with unresectable locally advanced or metastatic disease. It has a role as a tyrosine kinase inhibitor and an antineoplastic agent. It is an aromatic ether, a secondary amine, a member of quinazolines, a member of piperidines, an organobromine compound and an organofluorine compound.
brand name
Zactima (AstraZeneca);Caprelsa.
General Description
Class: receptor tyrosine kinase; Treatment: RET-altered thyroid cancers; Other name: AZD-6474; Elimination half-life = 19 days; Protein binding = 90%
Pharmacokinetics
Mean IC50 of approximately 2.1 μg/mL.
Clinical Use
Vandetanib, an oral VEGF, EGF, and RET receptor tyrosine kinase inhibitor, was developed by AstraZeneca for the treatment of symptomatic or aggressive medullary thyroid cancer (MTC) in patients with advanced or metastatic disease. This is the first drug approved for the treatment of MTC. Trials for other cancer indications such as small-cell lung cancer (SCLC), breast cancer, head and neck cancer, colorectal cancer, hormone-resistant prostate cancer, and papillary thyroid cancer are currently being explored. While AstraZeneca had previously developed ZD-4190 which displays similar efficacy and pharmacokinetic profile to vandetanib, vandetanib exhibited significantly improved solubility.
Synthesis
Vandetanib contains a 4-anilinoquinazoline scaffold similar to other EGFR inhibitors, and the synthesis described below is based on a recent patent (Scheme).
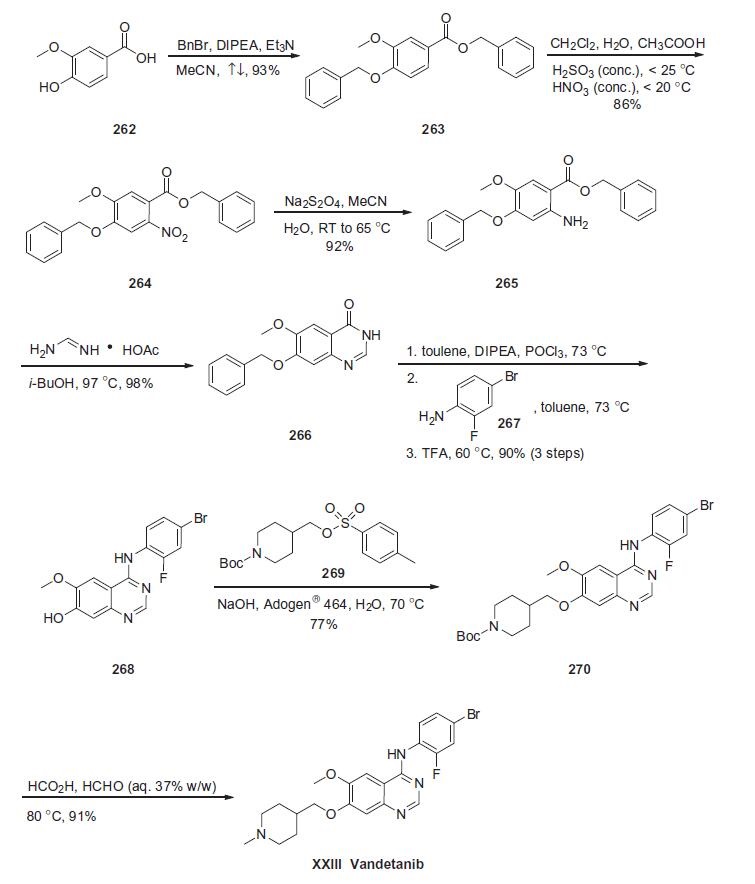
Commercially available vanillic acid (262) was treated with benzyl bromide, DIPEA and Et3N to give ethereal ester 263 in 93% yield. Arene 263 was then subjected to nitration conditions to provide nitroarene 264 in 86% yield, which underwent immediate reduction with sodium dithionite in acetonitrile and water to give aniline 265 in 92% yield. Aniline 265 was then treated with foramidine acetate in isobutanol which affected an intramolecular cyclization reaction, giving rise to dihydroquinazolin-4-one 266 in 98% yield. Heterocycle 266 was treated with phosphorous oxychloride and the resulting quinazoline chloride was subsequently reacted with 4-bromo-2-fluoroaniline 267 and trifluoroacetic acid to give hydroxyaniline 268 in 90% for the three-step sequence. Phenolic azacycle 268 was then alkylated with sulfonate 269 to furnish piperidine 270 in 77% yield. Subsequent treatment with formic acid and aqueous formaldehyde under elevated temperatures gave vandetanib (XXIII) in 91% yield.
Drug interactions
Potentially hazardous interactions with other drugs
Analgesics: possibly increased risk of ventricular
arrhythmias with methadone - avoid.
Anti-arrhythmics: possibly increased risk of
ventricular arrhythmias with amiodarone or
disopyramide - avoid.
Antibacterials: possibly increased risk of ventricular
arrhythmias with parenteral erythromycin and
moxifloxacin - avoid; concentration reduced by
rifampicin - avoid.
Antihistamines: possibly increased risk of ventricular
arrhythmias with mizolastine - avoid.
Antimalarials: possibly increased risk of ventricular
arrhythmias with artemether with lumefantrine -
avoid.
Antipsychotics: possibly increased risk of ventricular
arrhythmias with amisulpiride, chlorpromazine,
haloperidol, pimozide, sulpiride and zuclopenthixol
- avoid; avoid concomitant use with clozapine, risk
of agranulocytosis.
Beta-blockers: possibly increased risk of ventricular
arrhythmias with sotalol - avoid.
Cytotoxics: possibly increased risk of ventricular
arrhythmias with arsenic trioxide - avoid.
Hormone antagonist: possibly increased risk of
ventricular arrhythmias with toremifene - avoid.
5HT3
-receptor antagonists: possibly increased risk of
ventricular arrhythmias with ondansetron - avoid.
Pentamidine: possibly increased risk of ventricular
arrhythmias - avoid.
Metabolism
N-desmethyl-vandetanib is primarily produced by
CYP3A4, and vandetanib-N-oxide is primarily produced
by flavin-containing monooxygenase enzymes FMO1
and FMO3.
Unchanged vandentanib and metabolites vandetanib
N-oxide and N-desmethyl vandetanib were detected in
plasma, urine (25
%) and faeces (44
%).
Metabolism
Unchanged vandentanib and metabolites vandetanib N-oxide and N-desmethyl vandetanib were detected in plasma, urine and feces. N-desmethyl-vandetanib is primarily produced by CYP3A4, and vandetanib-N-oxide is primarily produced by flavin–containing monooxygenase enzymes FMO1 and FMO3.
Storage
Store at -20°C
References
Wedge et al. (2002), ZD6474 inhibits vascular endothelial growth factor signaling, angiogenesis and tumor growth following oral administration; Cancer Res., 62 4645 Ciardiello et al. (2003), Antitumor effects of ZD6474, a small molecule vascular endothelial growth factor receptor tyrosine kinase inhibitor, with additional activity against epidermal growth factor receptor tyrosine kinase; Clin. Cancer Res., 9 1546 Herbst et al. (2007), Vandetanib (ZD6474): an orally available receptor tyrosine kinase inhibitor that selectively targets pathways critical for tumor growth and angiogenesis; Expert Opin. Investig. Drugs, 16 239
Properties of Vandetanib
| Melting point: | 240-2430C |
| Boiling point: | 538.2±50.0 °C(Predicted) |
| Density | 1.406 |
| Flash point: | 279.3℃ |
| storage temp. | Keep in dark place,Inert atmosphere,Store in freezer, under -20°C |
| solubility | Soluble in DMSO (30 mg/ml); Ethanol (10 mg/ml with warming) |
| form | solid |
| pka | 8.92±0.10(Predicted) |
| color | White to off-white |
| Stability: | Stable for 2 years from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20°C for up to 1 month. |
| CAS DataBase Reference | 443913-73-3 |
Safety information for Vandetanib
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05  Exclamation Mark Irritant GHS07  Environment GHS09 |
| GHS Hazard Statements |
H227:Flammable liquids H302:Acute toxicity,oral H312:Acute toxicity,dermal H315:Skin corrosion/irritation H318:Serious eye damage/eye irritation H332:Acute toxicity,inhalation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation H410:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P270:Do not eat, drink or smoke when using this product. P271:Use only outdoors or in a well-ventilated area. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P310:Immediately call a POISON CENTER or doctor/physician. P330:Rinse mouth. P362:Take off contaminated clothing and wash before reuse. P302+P352:IF ON SKIN: wash with plenty of soap and water. P304+P340:IF INHALED: Remove victim to fresh air and Keep at rest in a position comfortable for breathing. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P332+P313:IF SKIN irritation occurs: Get medical advice/attention. P370+P378:In case of fire: Use … for extinction. P405:Store locked up. P403+P233:Store in a well-ventilated place. Keep container tightly closed. P403+P235:Store in a well-ventilated place. Keep cool. P501:Dispose of contents/container to..… |
Computed Descriptors for Vandetanib
Vandetanib manufacturer
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran
![[2-(4-chlorophenyl)quinolin-4-yl]-(2-piperidyl)methanol](https://img.chemicalbook.in/CAS/GIF/5428-80-8.gif)
You may like
-
 443913-73-3 Vandetanib 98%View Details
443913-73-3 Vandetanib 98%View Details
443913-73-3 -
 Vandetanib 98% CAS 443913-73-3View Details
Vandetanib 98% CAS 443913-73-3View Details
443913-73-3 -
 Vandetanib CAS 443913-73-3View Details
Vandetanib CAS 443913-73-3View Details
443913-73-3 -
 3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details
3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details -
 20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 -
 3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details
3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details -
 57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 -
 4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
5305-40-8
