Trimethylphosphine
Synonym(s):PMe3;Trimethylphosphane;Trimethylphosphorus
- CAS NO.:594-09-2
- Empirical Formula: C3H9P
- Molecular Weight: 76.08
- MDL number: MFCD00008510
- EINECS: 209-823-1
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-09-25 17:15:13
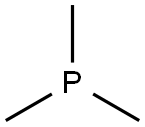
What is Trimethylphosphine?
Chemical properties
Clear colorless liquid
The Uses of Trimethylphosphine
Trimethylphosphine (PMe3) is an electron-rich phosphine ligand used as a reagent in Mitsunobu reaction.
It can be used:
- In transformation of azides into carbamates; aziridines to azidoalcohols; and ketoximes to ketones with 2,2′-dipyridyl diselenide.
- In Aza-Wittig reaction;, C-H bond activation of imines; cross-coupling reactions.
- In the preparation of (cyanomethylene) trimethylphosphorane (CMMP) which is used as a reagent in Mitsunobu type reaction.
- As a reagent in the synthesis of ruthenium trimethylphosphine complexes, which in turn are used as catalysts for hydrogenation of CO2 to formic acid.
The Uses of Trimethylphosphine
Trimethylphosphine (PMe3) solution is the suitable reagent used in the synthesis of Fe/Te cluster type complex, Fe6Te8(PMe3)6. It may be employed as a probe to investigate the acid sites in Y-zeolite. It may be used for the synthesis of hexakis(trimethylphosphine)tris-μ-methylene-diruthenium(III).
The Uses of Trimethylphosphine
Trimethylphosphine is an electron-rich phosphine ligand used in the Mitsunobu reaction. It participates in the transformation of azides into carbamates, aziridines from azidoalcohols, iminophosphoranes and aza-Wittig reaction. This product is offered as a solution in 2-methyltetrahydrofuran for more convenient handling.
Definition
ChEBI: Trimethylphosphine is a tertiary phosphine.
General Description
Trimethylphosphine is the reagent used in Mitsunobu reaction. It participates in the transformation of azides into carbamates, aziridines from azidoalcohols, iminophosphoranes and aza-Wittig reaction. Electron diffraction study of trimethylphosphine has been reported.
Purification Methods
All operations should be carried out in an efficient fume cupboard because it is flammable, toxic and has a foul odor. Distil trimethylphosphine at atmospheric pressure in a stream of dry N2 (apparatus should be held together with springs to avoid loss of gas from increased pressure in the system) and the distillate run into a solution of AgI in aqueous KI whereby the silver complex [Me3PAgI]4 separates steadily. Filter off the complex, wash it with saturated aqueous KI solution, then H2O and dry it in a vacuum desiccator over P2O5. The dry complex is heated in a flask (in a stream of dry N2) in an oil bath at 140o, when pure Me3P distils off (bath temperature can be raised up to 260o). The vapour pressure of Me3P at 20o is 466mm and the b is 37.8o [Thomas & Eriks Inorg Synth IX 59 1967]. Alternatively, freshly distilled Me3P (6g) is shaken with a solution of AgI (13.2g, 1.1mol) in saturated aqueous KI solution (50mL) for 2hours. A white solid, not wetted with H2O, separates rapidly. It is collected, washed with the KI solution, H2O, and dried [Mann et al. J Chem Soc 1829 1937]. The silver complex is stable if kept dry in the dark, in which state it can be kept indefinitely. Me3P can be generated from the complex when required. Store it under N2 in a sealed container. It has been distilled in a vacuum line at -78o in vacuo and condensed at -96o [IR and NMR: Crosbie & Sheldrick J Inorg Nucl Chem 31 3684 1969]. The pK2 2 by NMR was 8.80 [Silver & Lutz J Am Chem Soc 83 786 1961, pK2 5 8.65: Henderson & Strueuli J Am Chem Soc 82 5791 1960].
Properties of Trimethylphosphine
| Melting point: | -86 °C (lit.) |
| Boiling point: | 38-40 °C (lit.) |
| Density | 0.738 g/mL at 20 °C (lit.) |
| vapor pressure | 7.24 psi ( 20 °C) |
| refractive index | n |
| Flash point: | −4 °F |
| storage temp. | 2-8°C |
| form | liquid |
| Specific Gravity | 0.748 |
| color | colorless |
| Water Solubility | Soluble in water. |
| Sensitive | Air & Moisture Sensitive |
| BRN | 969138 |
| CAS DataBase Reference | 594-09-2(CAS DataBase Reference) |
| NIST Chemistry Reference | Phosphine, trimethyl-(594-09-2) |
Safety information for Trimethylphosphine
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H225:Flammable liquids H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Trimethylphosphine
| InChIKey | YWWDBCBWQNCYNR-UHFFFAOYSA-N |
Trimethylphosphine manufacturer
Sainor Laboratories Pvt Ltd Unit III
New Products
Indole Methyl Resin tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Boc-His(Boc)-OH 2-CTC Resin 4-Chloro-7-tosy1-7Hpyrrolo[2,3-d]pyrimidine 5,7-Dibromo-1H-indole 2,5-dichloro-N-hydroxy-4,6-dimethylpyridine-3-carboximidamide 2,2-Dimethoxy-7-azaspiro[3.5]nonane hydrochloride 4-chloromethyl-5-methyl-1,3-dioxol-2-one (DMDO-Cl) R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDIIMIDAZOLE 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) N-METHYL INDAZOLE-3-CARBOXYLIC ACID 4-((2-hydroxyethyl)thio)benzoic acid 1-(TERT-BUTOXYCARBONYL)-2-PYRROLIDINONE Methyl 6-methylnicotinate 3-Pyridineacrylic acid tert-Butyl carbazate TETRAHYDRO-2H-PYRAN-3-OL 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile 2,4-dihydroxybenzaldehyde 1,3-Diethyl-1,3-Diphenylurea Methyl 2-methylquinoline-6-carboxylateRelated products of tetrahydrofuran


You may like
-
 Trimethyl phosphine 98%View Details
Trimethyl phosphine 98%View Details -
 Trimethylphosphine, 99% CAS 594-09-2View Details
Trimethylphosphine, 99% CAS 594-09-2View Details
594-09-2 -
 Tri methyl phosphine 1.0M in THF CAS 594-09-2View Details
Tri methyl phosphine 1.0M in THF CAS 594-09-2View Details
594-09-2 -
 Trimethylphosphine CAS 594-09-2View Details
Trimethylphosphine CAS 594-09-2View Details
594-09-2 -
 Trimethylphosphine solution CAS 594-09-2View Details
Trimethylphosphine solution CAS 594-09-2View Details
594-09-2 -
 Trimethylphosphine solution CAS 594-09-2View Details
Trimethylphosphine solution CAS 594-09-2View Details
594-09-2 -
 Trimethylphosphine solution CAS 594-09-2View Details
Trimethylphosphine solution CAS 594-09-2View Details
594-09-2 -
 TRIMETHYL PHOSPHINE (594-09-2)View Details
TRIMETHYL PHOSPHINE (594-09-2)View Details
594-09-2
