Trimellitic Anhydride
Synonym(s):1,2,4-Benzenetricarboxylic 1,2-anhydride;Benzene-1,2,4-tricarboxylic anhydride;Trimellitic acid anhydride;Trimellitic anhydride
- CAS NO.:552-30-7
- Empirical Formula: C9H4O5
- Molecular Weight: 192.13
- MDL number: MFCD00005925
- EINECS: 209-008-0
- Update Date: 2025-09-18 19:10:02
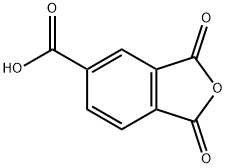
What is Trimellitic Anhydride?
Chemical properties
Trimellitic anhydride is a white powder or flakes. It is the anhydride of trimellitic acid (1,2,4-benzenetricarboxylic acid). It is soluble in water, ethanol, acetone and other general organic solvents.
The Uses of Trimellitic Anhydride
Trimellitic Anhydride is mainly used as raw material for polyester resin,polyimide resin,plasticizer TOTM ,and also be used for producing heatproof & insulating,adhesive,surfactant, paint,synthesize dye materials etc. Trimellitic anhydride is used as an embossing agent for vinyl flooring and as a curing agent for epoxy resins. It is also used as an intermediate for the synthesis of surface coatings chemicals, adhesives, polymers, dyes printing inks, pharmaceuticals .
The Uses of Trimellitic Anhydride
Trimellitic anhydride is mainly applied to produce good heat-proof, weather-proof and solvent-proof trimellitate plasticizers, among which the most popular product is tri(2-ethylhexyl)trimellitate. It is also used to synthesize polyester resins.TMA can also be applied to curing agent of epoxy resins.
Definition
ChEBI: Trimellitic anhydride is a 2-benzofuran compound having oxo groups at the 1- and 3-positions and a carboxy substituent at the 5-position; the cyclic anhydride formed from the carboxy groups at the 1- and 2-positions of trimellitic acid. It has a role as an epitope, an allergen and a hapten. It is a cyclic dicarboxylic anhydride, a dioxo monocarboxylic acid and a member of 2-benzofurans. It is functionally related to a phthalic anhydride and a trimellitic acid.
General Description
1,2,4-Benzenetricarboxylic anhydride(TA) is a hydrophilic monomer with multipurpose usage; as a curing agent for epoxy based resins and as a plasticizer for polyvinyl chloride(PVC).
Air & Water Reactions
Sensitive to moisture. Hydrolyzes slowly. Solutions in water or alcohol may be unstable. Insoluble in water.
Reactivity Profile
1,2,4-Benzenetricarboxylic anhydride reacts exothermically with water. This reaction is expected to be slow, but can become vigorous if local heating accelerates it. Reaction with water is accelerated by acids. Incompatible with acids, strong oxidizing agents, alcohols, amines, and bases. Incompatible with strong oxidizing agents, strong acids or strong bases. .
Hazard
Toxic by inhalation. Respiratory sensitization.
Health Hazard
Trimellitic anhydride (TMAN) causes both respiratory irritation and immunologic respiratory disease.
Safety Profile
Moderately toxic by ingestion. Has caused pulmonary edema from inhalation. Irritant to lungs and air passages. May be a powerful allergen. Typical attack consists of breathlessness, wheezing, cough, running nose, immunologcal sensitization, and asthma symptoms. When heated to decomposition it emits acrid smoke and irritating fumes. See also ANHYDRIDES.
Synthesis
3-Xylene is carbonylated with carbon monoxide in the presence of boron trifluoride and hydrogen fluoride to form 2,4-dimethylbenzaldehyde. 2,4-Dimethylbenzaldehyde is decomplexed from the acids, purified, and oxidized to trimellitic acid. Trimellitic acid is subjected to normal dehydration and purification steps to obtain high quality trimellitic anhydride.
Potential Exposure
TMA is used to produce trimellitate plasticizers, poly (amide-imide) polymers; in paints, enamels, and coatings; polymers, polyesters; as a curing agent for epoxy and other resins; in vinyl plasticizers; agricultural chemicals; dyes and pigments; pharmaceuticals, surface active agents; modifiers, intermediates, and specialty chemicals.
Carcinogenicity
There are no reports of carcinogenicity associated with TMAN exposure. It was not mutagenic in bacterial assays with or without metabolic activation. No teratogenic effects or developmental toxicity was seen in rats or guinea pigs exposed to 500mg/m3 for 6 hours/day during their period of major organogenesis.
Incompatibilities
Dust can cause an explosion. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. Reacts slowly with water, forming trimellitic acid. Compounds of the carboxyl group react with all bases, both inorganic and organic (i.e., amines) releasing substantial heat, water and a salt that may be harmful.
Waste Disposal
Dissolve or mix the material with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. All federal, state, and local environmental regulations must be observed.
Properties of Trimellitic Anhydride
| Melting point: | 163-166 °C (lit.) |
| Boiling point: | 390 °C |
| Density | 1.54 |
| vapor density | 6.6 (vs air) |
| vapor pressure | <0.01 mm Hg ( 20 °C) |
| refractive index | 1.4717 (estimate) |
| Flash point: | 227 °C |
| storage temp. | Store below +30°C. |
| solubility | 24.4g/l (decomposition) |
| form | Flakes |
| pka | 3.11±0.20(Predicted) |
| color | White to off-white |
| PH | 2 (21g/l, H2O, 20℃) |
| explosive limit | 1-7%(V) |
| Water Solubility | DECOMPOSES |
| Sensitive | Moisture Sensitive |
| Merck | 14,9703 |
| BRN | 9394 |
| Exposure limits | ACGIH: TWA 0.0005 mg/m3; STEL 0.002 mg/m3 (Skin) NIOSH: TWA 0.005 ppm(0.04 mg/m3) |
| Stability: | Stable. Moisture sensitive. |
| CAS DataBase Reference | 552-30-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Trimellitic anhydride(552-30-7) |
| EPA Substance Registry System | Trimellitic anhydride (552-30-7) |
Safety information for Trimellitic Anhydride
| Signal word | Danger |
| Pictogram(s) |
 Corrosion Corrosives GHS05  Exclamation Mark Irritant GHS07  Health Hazard GHS08 |
| GHS Hazard Statements |
H317:Sensitisation, Skin H318:Serious eye damage/eye irritation H334:Sensitisation, respiratory H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Trimellitic Anhydride
| InChIKey | SRPWOOOHEPICQU-UHFFFAOYSA-N |
Trimellitic Anhydride manufacturer
Triveni Interchem Private Limited (Group Of Triveni Chemicals)
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran


You may like
-
 Trimellitic anhydride 97% CAS 552-30-7View Details
Trimellitic anhydride 97% CAS 552-30-7View Details
552-30-7 -
 Trimellitic Anhydride CAS 552-30-7View Details
Trimellitic Anhydride CAS 552-30-7View Details
552-30-7 -
 1,2,4-Benzenetricarboxylic anhydride, 97% CAS 552-30-7View Details
1,2,4-Benzenetricarboxylic anhydride, 97% CAS 552-30-7View Details
552-30-7 -
 Trimellitic Anhydride CAS 552-30-7View Details
Trimellitic Anhydride CAS 552-30-7View Details
552-30-7 -
 Powder Trimellitic Anhydride Cas: 552-30-7, Packaging Type: BagView Details
Powder Trimellitic Anhydride Cas: 552-30-7, Packaging Type: BagView Details
552-30-7 -
 Tri Mellitic Anhydride (TMA), 50 kg BagView Details
Tri Mellitic Anhydride (TMA), 50 kg BagView Details
552-30-7 -
 Trimellitic AnhydrideView Details
Trimellitic AnhydrideView Details
552-30-7 -
 Trimellitic Anhydride PowderView Details
Trimellitic Anhydride PowderView Details
552-30-7
