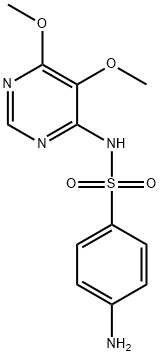
Sulfameter
Synonym(s):N1-(5-Methoxypyrimidin-2-yl)sulfanilamide;5-Methoxysulfadiazine;Sulfamethoxydiazine
- CAS NO.:651-06-9
- Empirical Formula: C11H12N4O3S
- Molecular Weight: 280.3
- MDL number: MFCD00006067
- EINECS: 211-480-8
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-06-08 09:02:58
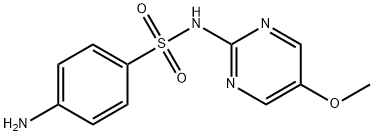
What is Sulfameter?
Chemical properties
Crystalline Solid
Originator
Sulla,Robins,US,1968
The Uses of Sulfameter
Sulfameter is an antibacterial.
The Uses of Sulfameter
Sulfameter is a sulfonamide antibiotic used as a leprostatic agent and to treat urinary tract infections. It has been investigated as a potential therapy for chloroquine-resistant malaria during pregnancy.
Background
Long acting sulfonamide used in leprosy, urinary, and respiratory tract infections.
Definition
ChEBI: A sulfonamide consisting of pyrimidine having a methoxy substituent at the 5-position and a 4-aminobenzenesulfonamido group at the 2-position.
What are the applications of Application
Sulfameter is An antibacterial
Manufacturing Process
2-Amino-5-methoxy pyrimidine is obtained having a melting point of about
300°C by condensation of methoxymalonic acid ester with guanidine
carbonate in the presence of sodium ethylate. The resultant reaction product
is then converted to 2-amino-5-methoxy-4,6-dichloropyrimidine (melting point
216°C to 217°C) by heating this reaction product with phosphorus
oxychloride. The dichloro compound is then suspended in water with zinc dust
and is tested in the presence of caustic alkaline or carbonates to produce the
2-aminod-methoxy pyrimidine compound, melting point 80°C to 82°C,
(benzene).
12.6 g of 2-amino-5-methoxy pyrimidine, 26.4 g of carbethoxy-sulfanilic acid
chloride and 50 cc of dry pyridine are heated for 30 minutes with frequent
shaking to a temperature of 80°C. The reaction product is then mixed with 200
cc of water and with dilute hydrochloric acid (0.1 N) until the reaction is acid to
Congo Red indicator. A precipitate is formed which is then filtered under
suction, washed with distilled water, and dried at 150°C. A practically
quantitative yield is recovered of 2-(p-carbethoxyaminobenzene-sulfonamido)-
5-methoxypyrimidine, melting point 248°C to 250°C.To hydrolyze the sulfapyrimidine compound, the same is heated at 90°C with
200 cc of 2 N potassium hydroxide solution for about one hour until complete
solution is obtained. The resultant solution is then cooled to room temperature
(25°C) and acidified with acetic acid to precipitate the hydrolyzed product,
which is then recrystallized from dilute acetone admixed with animal charcoal.
Therapeutic Function
Antibacterial
General Description
Chemical structure: sulfonamide
Pharmaceutical Applications
2-Sulfanilamido-5-methoxypyrimidine. A long-acting compound with activity similar to that of sulfadiazine. Binding to plasma proteins is about 75%.
Biochem/physiol Actions
Sulfameter is a sulfonamide antibiotic. Sulfonamides block the synthesis of dihydrofolic acid by inhibiting the enzyme dihydropteroate synthase. Sulfonamides are competitive inhibitors of bacterial para-aminobenzoic acid (PABA), which is required for bacterial synthesis of folic acid. Sulfameter is a dihydrofolate reductase (DHFR) inhibitor. Sulfonamides are active against Gram positive bacteria and Gram negative bacteria. Mode of resistance is via the alteration of dihydropteroate synthase or alternative pathway for folic acid synthesis.
Metabolism
Not Available
Properties of Sulfameter
| Melting point: | 214-216°C |
| Boiling point: | 539.4±56.0 °C(Predicted) |
| Density | 1.3936 (rough estimate) |
| refractive index | 1.6200 (estimate) |
| storage temp. | 2-8°C |
| solubility | DMSO (Slightly), Methanol (Sparingly) |
| form | powder |
| pka | pKa 7.02 (Uncertain) |
| color | white with faint yellow cast |
| Water Solubility | 0.47g/L(30 ºC) |
| Merck | 13,8999 |
| BRN | 621130 |
| CAS DataBase Reference | 651-06-9(CAS DataBase Reference) |
| NIST Chemistry Reference | Sulfameter(651-06-9) |
Safety information for Sulfameter
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Sulfameter
| InChIKey | GPTONYMQFTZPKC-UHFFFAOYSA-N |
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE 4-Bromopyrazole 5,6-Dimethoxyindanone Tert-butyl bis(2-chloroethyl)carbamate (2-Hydroxyphenyl)acetonitrile 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran

You may like
-
 Sulfameter 98% CAS 651-06-9View Details
Sulfameter 98% CAS 651-06-9View Details
651-06-9 -
 Sulfameter CAS 651-06-9View Details
Sulfameter CAS 651-06-9View Details
651-06-9 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1