Naled
Synonym(s):(1,2-Dibromo-2,2-dichloroethyl) dimethyl phosphate;Bromchlophos;Naled
- CAS NO.:300-76-5
- Empirical Formula: C4H7Br2Cl2O4P
- Molecular Weight: 380.78
- MDL number: MFCD00053227
- EINECS: 206-098-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-06-08 09:02:56
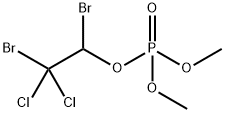
What is Naled?
Description
Sensitization to Naled seems to be rare.
Description
Naled is an organophosphate insecticide and acaricide that inhibits acetylcholinesterase (AChE) and butyrylcholinesterase (BChE). It reduces the number of A. sollicitans mosquitos by 97% after one hour when aerially applied at a concentration of 0.1 pounds per acre. Naled (125 ppm AI) induces 100 and 64% mortality of T. telarius adults and immature mites, respectively, in an immediate contact toxicity test but does not induce mortality in mite eggs. It is toxic to rats with an LD50 value of 250 mg/kg. Formulations containing naled have been used in the control of mosquitoes in public areas and of crop-damaging insects in agriculture.
Chemical properties
Clear Colorless Liquid
Chemical properties
Naled is a white crystalline solid (when pure) or light straw-colored liquid (above 26.7℃) with a slightly pungent insecticide odor.
Physical properties
Colorless to pale yellow liquid or solid with a pungent odor
The Uses of Naled
Naled is used to control mites, sucking pests and some other insects in a wide variety of crops. It is also used for the control of public and animal health pests including mosquitoes.
The Uses of Naled
Insecticide; acaricide.
The Uses of Naled
Insecticide used for control of spider mites, sucking and chewing insects in fruits, vegetables and ornamentals. Its use may be restricted.
The Uses of Naled
A cholinesterase inhibitor. Insecticide; acaricide.
What are the applications of Application
Naled is a cholinesterase inhibitor
Definition
ChEBI: An dialkyl phosphate resulting from the formal condensation of the acidic hydroxy group of dimethyl hydrogen phosphate with the alcoholic hydroxy group of 1,2-dibromo-2,2-dichloroethanol. An organophosphate insecticide, it is no longer approved for use wit in the European Union.
General Description
DIBROM is a white solid that may be dissolved in a liquid organic carrier with a pungent odor. DIBROM is a water emulsifiable liquid. DIBROM is insoluble in water and sinks in water. DIBROM can cause illness by inhalation, skin absorption and/or ingestion. The primary hazard is the threat to the environment. Immediate steps should be taken to limit its spread to the environment. If DIBROM is in liquid form, DIBROM can easily penetrate the soil and contaminate groundwater and nearby streams. DIBROM is used as a pesticide.
Air & Water Reactions
Practically insoluble in water [Farm Chemicals Handbook]. Hydrolyzed slowly in presence of water.
Reactivity Profile
DIBROM is incompatible with the following: Strong oxidizers, acids, sunlight, water [Note: Corrosive to metals. Hydrolyzed in presence of water.] . Unstable in presence of Iron [USCG, 1999]. Organophosphates are susceptible to formation of highly toxic and flammable phosphine gas in the presence of strong reducing agents such as hydrides. Partial oxidation by oxidizing agents may result in the release of toxic phosphorus oxides.
Hazard
Technical compound is a moderately volatile liquid. Bp 110C (0.5 mm Hg). Insoluble in water; slightly soluble in aliphatic solvents; very soluble in aromatic solvents; hydrolyzes in water.
Health Hazard
INHALATION OR INGESTION: Symptoms secondary to cholinesterase inhibition are: headache, giddiness, nervousness, blurred vision, weakness, nausea, cramps, diarrhea, chest discomfort, sweating, miosis, tearing, salivation, and other excessive respiratory tract secretion, vomiting, cyanosis, muscle twitching, and convulsions. EYES: Irritating. SKIN: Irritating-can cause dermatitis.
Fire Hazard
May be combustible. (NOAA, 2007)
Agricultural Uses
Insecticide, Fungicide, Bactericide, Acaricide: Naled is a fast-acting, nonsystemic contact and stomach insecticide used to control aphids, mites, mosquitoes,and flies on crops and in greenhouses, mushroom houses, animal and poultry houses, kennels, food-processing plants, and aquaria and in outdoor mosquito control. Liquid formulations can be applied to greenhouse heating pipes to kill insects by vapor action. It has been used by veterinarians to kill parasitic worms (other than tapeworms) in dogs. Naled may no longer be used in and around the home by residents or professional applicators. Naled is available in dust, emulsion concentrate, liquid, and ULV formulations. Also used in cooling towers, veterinary medicine, pulp and paper mill systems, hospitals, swimming pools, and bathrooms. A U.S. EPA restricted Use Pesticide (RUP). Not approved for use in EU countries. Registered for use in the U.S.
Trade name
AI3-24988®; ARTHODIBROM®; BROMCHLOPHOS®; BROMEX®; DIBROM®; FLYKILLER®; LUCANAL®; HIBROM®; ORTHO® 4355; ORTHODIBROM®; ORTHODIBROMO®; PROKIL® Naled; TRUMPET®
Contact allergens
Naled is an organophosphate cholinesterase inhibitor that is used as an insecticide and acaricide. Sensitization seems to be very rare.
Potential Exposure
A potential danger to those involved in the manufacture, formulation, and application of this insecticide, fungicide, bactericide, acaricide. Also used in cooling towers, veterinary medicine, pulp and paper mill systems; hospitals, swimming pools; and bathrooms.
Carcinogenicity
When dogs were given 0.2, 2.0, or 20 mg/kg/day naled by gavage for 1 year, cholinergic signs (soft stools/diarrhea, salivation, and emesis), increases in mineralization of spinal cord, and mild testicular degeneration in males occurred at 2 and 20 mg/kg/day dose levels . Erythrocyte and brain cholinesterase activities were depressed at the same dose levels. Anemia also occurred at 2 and 20 mg/kg/day, and erythrocyte count, hemoglobin, and hematocrit were reduced. At 20 mg/kg/ day dose level, liver and kidney weights increased but these were not accompanied by histopathological changes. There was no evidence of carcinogenicity.
Environmental Fate
Chemical/Physical. Completely hydrolyzed in water within 2 days (Windholz et al.,
1983). In the presence of metals or reducing agents, naled loses bromine, forming dichlorvos
(Hartley and Kidd, 1987)
Naled emits toxic fumes of bromines, chlorides and phosphorus oxides when heated
to decomposition (Lewis, 1990).
Metabolic pathway
Naled is produced by the photochemical bromination of the dichlorovinyl moiety of dichlorvos. The main route of naled metabolism and transformation in the environment is through debromination to dichlorvos which is probably the active cholinesterase inhibitor in vivo. Naled is also rapidly hydrolysed to bromodichloroacetaldehyde in aqueous environments: consequently much of its detoxification is likely to be via a nonenzymatic hydrolytic route.
Solubility in water
Freely soluble in ketone, alcohols, aromatic and chlorinated hydrocarbons but sparingly soluble in petroleum solvents and mineral oils (Windholz et al., 1983)
Shipping
UN2811 Toxic solids, organic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required. UN2783 Organophosphorus pesticides, solid, toxic, Hazard Class: 6.1; Labels: 6.1-Poisonous materials. UN3018 Organophosphorus pesticides, liquid, toxic, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.
Degradation
Naled is stable when dry but it is rapidly hydrolysed in aqueous media and more rapidly in alkaline and acidic media. It is degraded by sunlight. In the presence of metals and reducing agents, bromine is lost and dichlorvos (2) is formed (PM). In unbuffered aqueous solution naled was rapidly hydrolysed via two routes, one leading to the formation of dimethyl phosphate (3) and bromodichloroacetaldehyde (4) and the second via desmethyhaled (5), which further decomposed to yield bromodichloroacetaldehyde (4) and monomethyl phosphate (6) as shown in Scheme 1. Naled was found to be much less hydrolytically stable than dichlorvos and exhibited a maximum stability at pH 4 (Jentzen and Fischer, 1978).
Incompatibilities
Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. Hydrolyzed in presence of water. Degraded by sunlight. Decomposes when heated; on contact with acids, acid fumes; bases, producing fumes of hydrogen chloride, hydrogen bromide, phosphorous oxides. Reacts with acids, strong oxidizers in sunlight. Slowly reacts with water; hydrolysis is; corrosive to metals. Attacks some plastics, rubber and coatings.
Waste Disposal
This pesticide is more stable to hydrolysis than dichlorvos (50% hydrolysis at pH 9 @ 37.5℃ in 301 minutes). It is unstable in alkaline conditions, in presence of iron; and is degraded by sunlight. About 10% hydrolysis per day is obtained in ambient water. Incineration is recommended for large amounts. In accordance with 40CFR165, follow recommendations for the disposal of pesticides and pesticide containers. Must be disposed properly by following package label directions or by contacting your local or federal environmental control agency, or by contacting your regional EPA office.
Properties of Naled
| Melting point: | 212℃ (decomposition) |
| Boiling point: | 110℃ (0.5 Torr) |
| Density | 1.96 g/cm3 |
| vapor pressure | 2 (quoted, Verschueren, 1983) |
| refractive index | 1.5108 (28℃) |
| storage temp. | 0-6°C
|
| solubility | Freely soluble in ketone, alcohols, aromatic and chlorinated hydrocarbons but sparingly soluble in
petroleum solvents and mineral oils (Windholz et al., 1983) |
| Water Solubility | 2000mg l-1(20 °C) |
| form | solid |
| Specific Gravity | 1.96 (20℃) |
| Merck | 13,6384 |
| BRN | 2049930 |
| Exposure limits | NIOSH REL: TWA 3 mg/m3, IDLH 200 mg/m3; OSHA PEL: TWA 3
mg/m3; ACGIH TLV: TWA 3 mg/m3. |
| Stability: | Light Sensitive, Moisture Sensitive |
| EPA Substance Registry System | Naled (300-76-5) |
Safety information for Naled
| Signal word | Danger |
| Pictogram(s) |
 Skull and Crossbones Acute Toxicity GHS06  Environment GHS09 |
| GHS Hazard Statements |
H301:Acute toxicity,oral H312:Acute toxicity,dermal H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H400:Hazardous to the aquatic environment, acute hazard |
| Precautionary Statement Codes |
P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P310:IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Naled
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium THOMAIND PAPER PH 2.0 TO 4.5 1 BOX BUFFER CAPSULE PH 9.2 - 10 CAP SODIUM CHLORIDE 0.1N CVS ALLOXAN MONOHYDRATE 98% PLATINUM 0.5% ON 3 MM ALUMINA PELLETS (TYPE 73) LITHIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran





You may like
-
 Dibrom® CAS 300-76-5View Details
Dibrom® CAS 300-76-5View Details
300-76-5 -
 1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
99903-60-3 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
1805639-70-6 -
 1784294-80-9 98%View Details
1784294-80-9 98%View Details
1784294-80-9 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4