Esomeprazole
- CAS NO.:119141-88-7
- Empirical Formula: C17H19N3O3S
- Molecular Weight: 345.42
- MDL number: MFCD09907604
- EINECS: 643-098-6
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-07-16 09:52:44
What is Esomeprazole?
Absorption
After oral administration, peak plasma levels (Cmax) occur at approximately 1.5 hours (Tmax). The Cmax increases proportionally when the dose is increased, and there is a three-fold increase in the area under the plasma concentration-time curve (AUC) from 20 to 40 mg. At repeated once-daily dosing with 40 mg, the systemic bioavailability is approximately 90% compared to 64% after a single dose of 40 mg. The mean exposure (AUC) to esomeprazole increases from 4.32 μmolhr/L on Day 1 to 11.2 μmolhr/L on Day 5 after 40 mg once daily dosing. The AUC after administration of a single 40 mg dose of Esomeprazole is decreased by 43% to 53% after food intake compared to fasting conditions. Esomeprazole should be taken at least one hour before meals.
Combination Therapy with Antimicrobials:
Esomeprazole magnesium 40 mg once daily was given in combination with Clarithromycin 500 mg twice daily and Amoxicillin 1000 mg twice daily for 7 days to 17 healthy male and female subjects. The mean steady state AUC and Cmax of esomeprazole increased by 70% and 18%, respectively during triple combination therapy compared to treatment with esomeprazole alone. The observed increase in esomeprazole exposure during co-administration with clarithromycin and amoxicillin is not expected to produce significant safety concerns.
Toxicity
Blurred vision, confusion, drowsiness, dry mouth, flushing headache, nausea, rapid heartbeat, sweating
Description
Esomeprazole, formulated as a magnesium salt, reached the market as a treatment for acid-related diseases such as gastro-esophageal reflux (GERD) disease including peptic ulcer disease and reflux esophagitis. Esomeprazole (formerly perprazole) is the active (S)- enantiomer of omeprazole (1988) and the first proton pump inhibitor developed as an optical isomer. It can be obtained by several routes such as asymmetric oxidation of the pro-chiral pyridylmethyl benzimidazole sulfide, separation from the racemic sulfoxide by chiral chromatography or separation of a diastereomeric mixture obtained from the racemic compound and a chiral acid, followed by hydrolysis. Biochemical studies have shown that esomeprazole irreversibly inhibits the gastric H+/K+-adenosine triphosphatase (ATPase), an enzyme system involved at the secretory surface of the stomach’s parietal cells responsible for the secretion of gastric acid. Compared with racemic omeprazole in healthy subjects, esomeprazole has higher bioavailability, is absorbed more rapidly and exhibits a more uniform and predictable dose-response with higher plasma levels, leading to less inter-individual variability between slow and rapid metabolizers. In extensive clinical trials in patients suffering from GERD symptoms, esomeprazole provided superior acid control and significantly reduced the healing time compared to omeprazole.
Originator
AstraZeneca (UK)
The Uses of Esomeprazole
Esomeprazole is used to treat certain stomach and esophagus problems (such as acid reflux, ulcers). It works by decreasing the amount of acid your stomach makes. It relieves symptoms such as heartburn, difficulty swallowing, and persistent cough.
The Uses of Esomeprazole
(-)-Omeprazole can be used to treat migraine.
Background
Esomeprazole, sold under the brand name Nexium, is a proton pump inhibitor (PPI) medication used for the management of gastroesophageal reflux disease (GERD), for gastric protection to prevent recurrence of stomach ulcers or gastric damage from chronic use of NSAIDs, and for the treatment of pathological hypersecretory conditions including Zollinger-Ellison (ZE) Syndrome. It can also be found in quadruple regimens for the treatment of H. pylori infections along with other antibiotics including Amoxicillin, Clarithromycin, and Metronidazole, for example. Its efficacy is considered similar to other medications within the PPI class including Omeprazole, Pantoprazole, Lansoprazole, Dexlansoprazole, and Rabeprazole. Esomeprazole is the s-isomer of Omeprazole, which is a racemate of the S- and R-enantiomer. Esomeprazole has been shown to inhibit acid secretion to a similar extent as Omeprazole, without any significant differences between the two compounds in vitro.
Esomeprazole exerts its stomach acid-suppressing effects by preventing the final step in gastric acid production by covalently binding to sulfhydryl groups of cysteines found on the (H+, K+)-ATPase enzyme at the secretory surface of gastric parietal cells. This effect leads to inhibition of both basal and stimulated gastric acid secretion, irrespective of the stimulus. As the binding of esomeprazole to the (H+, K+)-ATPase enzyme is irreversible and new enzyme needs to be expressed in order to resume acid secretion, esomeprazole's duration of antisecretory effect persists longer than 24 hours.
PPIs such as esomeprazole have also been shown to inhibit the activity of dimethylarginine dimethylaminohydrolase (DDAH), an enzyme necessary for cardiovascular health. DDAH inhibition causes a consequent accumulation of the nitric oxide synthase inhibitor asymmetric dimethylarginie (ADMA), which is thought to cause the association of PPIs with increased risk of cardiovascular events in patients with unstable coronary syndromes.
Due to their good safety profile and as several PPIs are available over the counter without a prescription, their current use in North America is widespread. Long term use of PPIs such as esomeprazole has been associated with possible adverse effects, however, including increased susceptibility to bacterial infections (including gastrointestinal C. difficile), reduced absorption of micronutrients such as iron and B12, and an increased risk of developing hypomagnesemia and hypocalcemia which may contribute to osteoporosis and bone fractures later in life.
Rapid discontinuation of PPIs such as esomeprazole may cause a rebound effect and a short term increase in hypersecretion. Esomeprazole doses should be slowly lowered, or tapered, before discontinuing to prevent this rebound effect.
Indications
Esomeprazole is indicated for the treatment of acid-reflux disorders including healing and maintenance of erosive esophagitis, and symptomatic gastroesophageal reflux disease (GERD), peptic ulcer disease, H. pylori eradication, prevention of gastrointestinal bleeds with NSAID use, and for the long-term treatment of pathological hypersecretory conditions including Zollinger-Ellison Syndrome.
Definition
ChEBI: A 5-methoxy-2-{[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]sulfinyl}-1H-benzimidazole that has S configuration at the sulfur atom. An inhibitor of gastric acid secretion, it is used (generally as its sodium or magnesium alt) for the treatment of gastro-oesophageal reflux disease, dyspepsia, peptic ulcer disease, and Zollinger-Ellison syndrome.
brand name
Nexium
Pharmacokinetics
Esomeprazole is a compound that inhibits gastric acid secretion and is indicated in the treatment of gastroesophageal reflux disease (GERD), the healing of erosive esophagitis, and H. pylori eradication to reduce the risk of duodenal ulcer recurrence. Esomeprazole belongs to a new class of antisecretory compounds, the substituted benzimidazoles, that do not exhibit anticholinergic or H2 histamine antagonistic properties, but that suppress gastric acid secretion by specific inhibition of the H+/K+ ATPase at the secretory surface of the gastric parietal cell. By doing so, it inhibits acid secretion into the gsatric lumen. This effect is dose-related and leads to inhibition of both basal and stimulated acid secretion irrespective of the stimulus.
Esomeprazole is the s-isomer of Omeprazole, which is a racemate of the S- and R-enantiomer. Esomeprazole has been shown to inhibit acid secretion to a similar extent as Omeprazole, without any significant differences between the two compounds in vitro.
PPIs such as esomeprazole have also been shown to inhibit the activity of dimethylarginine dimethylaminohydrolase (DDAH), an enzyme necessary for cardiovascular health. DDAH inhibition causes a consequent accumulation of the nitric oxide synthase inhibitor asymmetric dimethylarginie (ADMA), which is thought to cause the association of PPIs with increased risk of cardiovascular events in patients with unstable coronary syndromes.
Due to their good safety profile and as several PPIs are available over the counter without a prescription, their current use in North America is widespread. Long term use of PPIs such as esomeprazole has been associated with possible adverse effects, however, including increased susceptibility to bacterial infections (including gastrointestinal C. difficile), reduced absorption of micronutrients including iron and B12, and an increased risk of developing hypomagnesemia and hypocalcemia which may contribute to osteoporosis and bone fractures later in life.
Drug interactions
Potentially hazardous interactions with other drugsAnticoagulants: effect of coumarins possibly enhanced.Antiepileptics: effects of fosphenytoin and phenytoin enhanced.Antifungals: absorption of itraconazole and ketoconazole reduced; avoid with posaconazole; concentration possibly increased by voriconazole.Antivirals: concentration of atazanavir and rilpivirine reduced - avoid concomitant use; concentration of raltegravir and saquinavir possibly increased - avoid; concentration of esomeprazole reduced by tipranavir.Clopidogrel: reduced antiplatelet effect.Cytotoxics: possibly reduced excretion of methotrexate; avoid with dasatinib, erlotinib and vandetanib; possibly reduced lapatinib absorption; possibly reduced absorption of pazopanib.Ulipristal: reduced contraceptive effect, avoid with high dose ulipristal
Metabolism
Esomeprazole is extensively metabolized in the liver by the cytochrome P450 (CYP) enzyme system. The metabolites of esomeprazole lack antisecretory activity. The major part of esomeprazole’s metabolism is dependent upon the CYP2C19 isoenzyme, which forms the hydroxy and desmethyl metabolites. The remaining amount is dependent on CYP3A4 which forms the sulphone metabolite. CYP2C19 isoenzyme exhibits polymorphism in the metabolism of esomeprazole, since some 3% of Caucasians and 15 to 20% of Asians lack CYP2C19 and are termed Poor Metabolizers. However, the influence of CYP 2C19 polymorphism is less pronounced for esomeprazole than for omeprazole. At steady state, the ratio of AUC in Poor Metabolizers to AUC in the rest of the population (Extensive Metabolizers) is approximately 2.
Following administration of equimolar doses, the S- and R-isomers are metabolized differently by the liver, resulting in higher plasma levels of the S- than of the R-isomer.
Nine major urinary metabolites have been detected. The two main metabolites have been identified as hydroxyesomeprazole and the corresponding carboxylic acid. Three major metabolites have been identified in plasma: the 5-O-desmethyl- and sulphone derivatives and hydroxyesomeprazole. The major metabolites of esomeprazole have no effect on gastric acid secretion.
Metabolism
Esomeprazole is completely metabolised by the cytochrome P450 system (CYP). The major part of the metabolism of esomeprazole is dependent on the polymorphic CYP2C19, responsible for the formation of the hydroxy- and desmethyl metabolites of esomeprazole. The remaining part is dependent on another specific isoform, CYP3A4, responsible for the formation of esomeprazole sulphone, the main metabolite in plasma. The major metabolites of esomeprazole have no effect on gastric acid secretion.Almost 80% of an oral dose of esomeprazole is excreted as metabolites in the urine, the remainder in the faeces. Less than 1% of the parent drug is found in urine.
Properties of Esomeprazole
| Boiling point: | 600.0±60.0 °C(Predicted) |
| alpha | D20 -155° (c = 0.5 in chloroform) |
| Density | 1.37±0.1 g/cm3(Predicted) |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | DMF: 30 mg/ml,DMSO: 20 mg/ml,Ethanol: 10 mg/ml,PBS (pH 7.2): 10 mg/ml |
| pka | 8.50±0.10(Predicted) |
| form | A solid |
| CAS DataBase Reference | 119141-88-7(CAS DataBase Reference) |
Safety information for Esomeprazole
Computed Descriptors for Esomeprazole
Esomeprazole manufacturer
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran



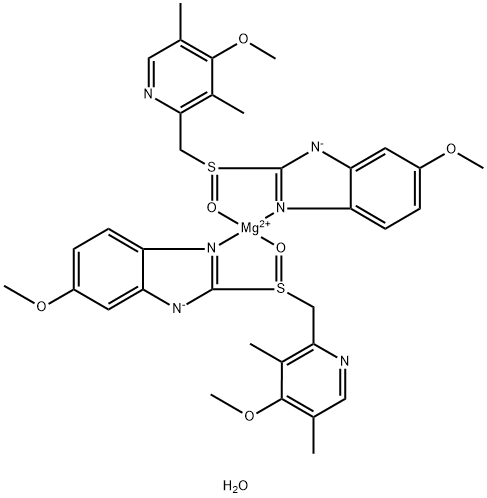
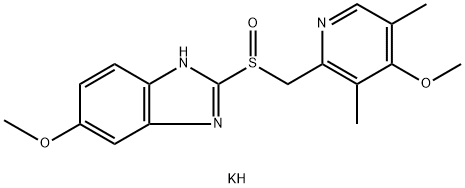
You may like
-
 119141-88-7 ESOMEPRAZOLE 8.5%, 15.0%, 17%, 22%, 22.5%, 27.10% EC PELLETS 98%View Details
119141-88-7 ESOMEPRAZOLE 8.5%, 15.0%, 17%, 22%, 22.5%, 27.10% EC PELLETS 98%View Details
119141-88-7 -
 119141-88-7 98%View Details
119141-88-7 98%View Details
119141-88-7 -
 Esomeprazole 98%View Details
Esomeprazole 98%View Details -
 119141-88-7 98%View Details
119141-88-7 98%View Details
119141-88-7 -
 Esomeprazole 119141-88-7 98%View Details
Esomeprazole 119141-88-7 98%View Details
119141-88-7 -
 119141-88-7 Esomeprazole 98%View Details
119141-88-7 Esomeprazole 98%View Details
119141-88-7 -
 119141-88-7 98%View Details
119141-88-7 98%View Details
119141-88-7 -
 Esomeprazole 99%View Details
Esomeprazole 99%View Details
119141-88-7
