Enfuvirtide
Synonym(s):T-20 acetate salt
- CAS NO.:159519-65-0
- Empirical Formula: C204H301N51O64
- Molecular Weight: 4491.92
- MDL number: MFCD08062320
- EINECS: 641-385-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-11-05 11:59:05
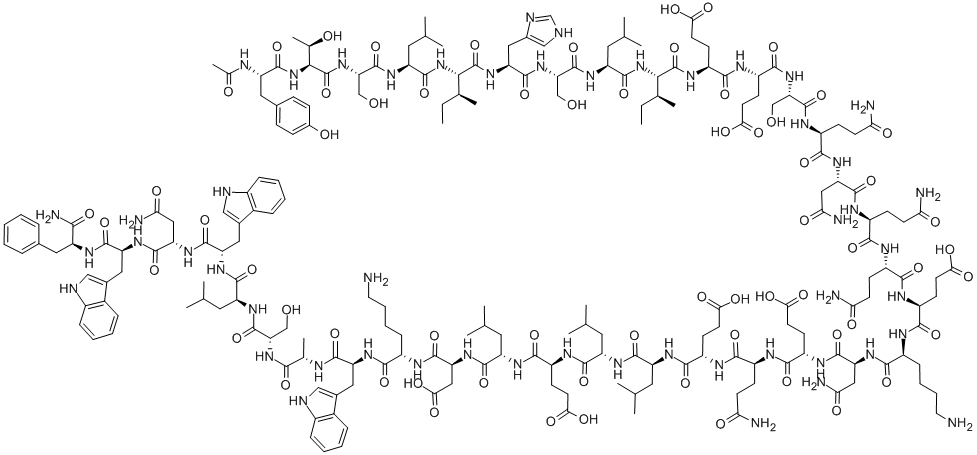
What is Enfuvirtide?
Description
Enfuvirtide is the first of a new class of HIV therapeutics that interferes with the entry of HIV-1 by inhibiting fusion of viral and cellular membranes. It binds to the first heptad-repeat (HR1) in ghe gp41 subunit of the viral envelope glycoprotein and prevents the conformational changes required for the fusion with cell membranes, thus representing a new mechanism. It is a potent inhibitor of this binding with an IC50 in laboratory and primary isolates (HIV-1 clades A–G) ranging from 4 to 280 nM (18–1260 ng/mL), but has virtually no activity against HIV-2. Due to its unique mechanism, it does not show cross-resistance to NRTI, NNRTI and PIs. It is prepared by chemical synthesis, using a combination of solution and solid phase steps. The solid phase steps utilize fmoc protection and the acid sensitive 2- chlorotrityl chloride resin to produce peptides of lengths ranging from nine to sixteen amino acids. These amino acids are subsequently coupled to form the full-length peptide followed by side-chain deprotection and column chromatography. In the HuPBMC-SCID mouse model of HIV-1 infection enfuvirtide showed a dosedependent decrease in viral load. At doses of 200 mg/kg/day, it decreased the RNA levels to 8.2 copies/1 million cells compared to 17 million copies/1 million cells in the saline-treated group. In a clinical study involving 78 heavily pretreated patients (plasma HIV RNA>5000 copies/mL), enfuvirtide dosing of 12.5–200 mg/day by b.i.d. subcutaneous injection resulted in a dose-dependent decrease in viral load (plasma HIV RNA 0.3–1.6 log 10 copies/mL). In a combination drug study where all patients received an optimized retroviral regimen with or without enfuvirtide, it was found that, at week 24, the enfuvirtide group had a 4.5 fold-relative drop in HIV RNA copies/mL relative to the standard treatment group. Subjects received 3–5 antiretroviral agents with or without enfuvirtide. In clinical trials, HIV-1 isolates from 185 patients using enfuvirtide in a cocktail with other antiretroviral agents showed 4 to 422-fold decrease in sensitivity to this drug. These more resistant types had changes in the gp41 amino acids 36–45. Enfuvirtide is dosed twice per day (90 mg) by subcutaneous injection and has an elimination half-life of about 3.8 h. It is highly plasma protein bound (92%) and has a Vdss is 5.5 L. Local injection site irritation (98% had at least one reaction), diarrhea (26.8%), nausea (20.1%), and fatigue (16.1%) were the most common adverse events.
Description
Enfuvirtide is a biomimetic peptide inhibitor of HIV-1 fusion with CD4+ cells. It inhibits HIV-1 infectivity of HeLa cells stably expressing CD4 by the HXB2 strain (IC50 = 692 pM) and by clinical isolates (IC90s = 6.1-61 nM) in a single-cycle infectivity assay. It also inhibits genomic integration of HIV-1 into human intraepithelial vaginal cells and peripheral blood mononuclear cells (PBMCs; IC50s = 51.2 and 13.58 μM, respectively). Enfuvirtide binds to a recombinant molecular mimic of HIV-1 glycoprotein gp41 that contains three N-terminal heptad and two C-terminal heptad repeat regions (Kd = 32 nM). It also binds to recombinant formyl peptide receptors (FPR) expressed in rat basophilic leukemia cells (IC50 = 5 nM) and attracts and activates human peripheral blood phagocytes, but not T lymphocytes, in vitro when used at a concentration of 100 nM. Formulations containing enfuvirtide have been used in combination therapy for the treatment of HIV-1/AIDS.
Originator
Duke University (US)
The Uses of Enfuvirtide
Antiviral (blockade of gp-41 mediated membrane fusion). Fuzeon (Roche) [Note—The trivial name, pentafuside, has appeared in literature.
Definition
ChEBI: A synthetic 36-amino acid peptide consisting of N-acetyltyrosyl, threonyl, seryl, leucyl, isoleucyl, histidyl, seryl, leucyl, isoleucyl, alpha-glutamyl, alpha-glutamyl, seryl, glutaminyl, asparaginyl, glutaminyl, glutamin l, alpha-glutamyl, lysyl, asparaginyl, alpha-glutamyl, alpha-glutamyl, alpha-glutamyl, leucyl, leucyl, alpha-glutamyl, leucyl, alpha-aspartyl, lysyl, tryp ophyl, alanyl, seryl, leucyl, tryptophyl, asparaginyl, tryptophyl, and phenylalaninamide residues joined in sequence. An HIV fusion inhibitor, it was the first of a novel class of antiretroviral drugs used in combination therapy for the treatment of HIV-1 nfection. It interferes with entry of HIV into cells by binding to the gp41 sub-unit of the viral envelope glycoprotein, so inhibiting fusion of viral and cellular membranes.
brand name
Fuzeon
Biological Functions
Enfuvirtide is used in combination with other antiretrovirals and works against a variety of HIV-1 variants, but it is not active against HIV-2. Resistance to enfuvirtide can develop when the virus produces changes in a 10-amino-acid domain between residues 36 to 45 in the gp41 HIV surface glycoprotein.
Acquired resistance
Resistance is mediated by amino acid substitutions within the first heptad repeat region of gp41 at amino acids 36–45. Resistance emerges fairly rapidly in patients experiencing virological failure with an enfuvirtide-containing antiretroviral regimen, and is associated with the return of the plasma HIV load toward baseline within a few weeks.
General Description
Enfuvirtide acetate salt is a linear synthetic peptide made of 36 amino acids. It has an acetylated N-terminus and a carboxamide C-terminus. Enfuvirtide is catabolized with the help of proteolytic enzymes.
Pharmaceutical Applications
A linear 36-amino acid synthetic peptide with an acetylated N-terminus and a carboxamide C-terminus. It is formulated as a lyophilized powder to be reconstituted for subcutaneous injection.
Biochem/physiol Actions
Enfuvirtide is an HIV fusion inhibitor used to patients with multi-drug resistant HIV. Enfuvirtide binds to gp41 preventing the creation of an entry pore for the HIV-1 virus.
Pharmacokinetics
Subcutaneous absorption c. 84.3%
Cmax 90 mg s/c twice daily c. 4.59 mg/L
Plasma half-life c. 3.8 h
Volume of distribution 5.5 L
Plasma protein binding c. 92%
Absorption and distribution
Absorption of the 90 mg dose is comparable when injected into the subcutaneous tissue of the abdomen, thigh or arm. It does not penetrate the CSF or semen. Distribution into breast milk has not been described.
Metabolism and excretion
It probably undergoes catabolism to its constituent amino acids, with subsequent recycling of the amino acids in the body pool.
Clinical Use
Treatment of HIV infection (in combination with other antiretroviral drugs) in adults and children older than 6 years who show evidence of HIV-1 replication despite ongoing antiretroviral therapy
Side Effects
It does not seem to have any long-term toxicities (including the HIV lipodystrophy syndrome) associated with other commonly used antiretrovirals. Reaction at the injection site, variously characterized by local pain, erythema, pruritus, induration, ecchymosis, nodules or cysts, is experienced by more than 90% of patients and may lead to treatment fatigue.
Drug interactions
Potentially hazardous interactions with other drugs Orlistat: absorption possibly reduced by orlistat.
Metabolism
As a peptide, enfuvirtide is expected to undergo catabolism to its constituent amino acids, with subsequent recycling of the amino acids in the body pool. In vitro human microsomal studies and in vivo studies indicate that enfuvirtide is not an inhibitor of CYP450 enzymes. In in vitro human microsomal and hepatocyte studies, hydrolysis of the amide group of the C-terminus amino acid, phenylalanine results in a deamidated metabolite. Mass balance studies to determine elimination pathway(s) of enfuvirtide have not been performed in humans.
Properties of Enfuvirtide
| storage temp. | -20°C |
| solubility | DMF: 5 mg/ml; DMSO: 5 mg/ml; PBS (pH 7.2): 2 mg/ml |
| form | powder |
| color | white to off-white |
Safety information for Enfuvirtide
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. |
Computed Descriptors for Enfuvirtide
New Products
Tert-butyl bis(2-chloroethyl)carbamate 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid DIETHYL AMINOMALONATE HYDROCHLORIDE 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) N-METHYL INDAZOLE-3-CARBOXYLIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran





You may like
-
 Enfuvirtide acetate salt CAS 159519-65-0View Details
Enfuvirtide acetate salt CAS 159519-65-0View Details
159519-65-0 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1 -
 733039-20-8 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 98+View Details
733039-20-8 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 98+View Details
733039-20-8