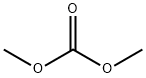
Dimethyl ether
Synonym(s):DME;Methyl ether
- CAS NO.:115-10-6
- Empirical Formula: C2H6O
- Molecular Weight: 46.07
- MDL number: MFCD00008494
- EINECS: 204-065-8
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-17 09:50:35
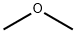
What is Dimethyl ether?
Description
Dimethyl ether (DME), a colorless gas, is the simplest aliphatic ether. Also called methyl ether, it is currently produced by dehydrating methanol; but researchers have an eye on it for sustainable production from biogenic methane or biomass derived from cellulose or lignin.
DME is primarily used to make other small molecules such as acetic acid or dimethyl sulfate. It is also a spray-can propellant1 and a refrigerant to replace chlorofluorocarbons. It has been proposed as a cleaner-burning fuel than hydrocarbons.
Far beyond its terrestrial presence, DME was a significant recent discovery in outer space. In March, researchers at Leiden University (the Netherlands) used the Atacama Large Millimeter/submillimeter Array telescope to identify the molecule in a planet-forming disc in the constellation Ophiuchus, 444 light-years from Earth. With nine atoms, DME is the largest molecule yet observed in space.
Always optimistic, some astrobiologists have speculated that the presence of DME could be an indicator of life “out there”.
1. Erik Rotheim, the Norwegian inventor of the spray can, cited the use of DME in one of his patents, US 1,800,156, in 1931.
Description
Dimethyl ether is a colorless compressed liquefied gas or liquid. Molecular weight= 46.08; Boilingpoint= 224℃; Freezing/Melting point = 2 141℃; Vaporpressure = 5200 hPa at 20℃; Flash point = flammable gas(241℃); Autoignition temperature= 350℃. Explosivelimits: LEL = 3.4%; UEL= 27.0%. Hazard Identification(based on NFPA-704 M Rating System): Health 2,Flammability 4, Reactivity 1. Soluble in water.
Chemical properties
Dimethyl ether is a liquefied gas and exists as a liquid at room temperature when contained under its own vapor pressure, or as a gas when exposed to room temperature and pressure.
It is a clear, colorless, virtually odorless liquid. In high concentrations, the gas has a faint ether-like odor.
The Uses of Dimethyl ether
Dimethyl ether is used as a solvent in aerosol formulations.
The Uses of Dimethyl ether
Aerosol propellant; alternative diesel fuel; chemical intermediate.
The Uses of Dimethyl ether
Methyl ether is used as an aerosol propellantand in refrigeration.
Definition
ChEBI: An ether in which the oxygen atom is connected to two methyl groups.
Production Methods
Dimethyl ether is prepared by the reaction of bituminous or lignite coals with steam in the presence of a finely divided nickel catalyst. This reaction produces formaldehyde, which is then reduced to methanol and dimethyl ether. Dimethyl ether may also be prepared by the dehydration of methanol.
General Description
Dimethyl ether is a colorless gas with a faint ethereal odor. Dimethyl ether is shipped as a liquefied gas under its vapor pressure. Contact with the liquid can cause frostbite. Dimethyl ether is easily ignited. Its vapors are heavier than air. Any leak can be either liquid or vapor. Dimethyl ether can asphyxiate by the displacement of air. Under prolonged exposure to fire or intense heat the containers may rupture violently and rocket.
Air & Water Reactions
Highly flammable. Upon standing and exposure to air (oxygen) tendency to form explosive peroxides. When ethers containing peroxides are heated (distilled) they can detonate [Lewis, 3rd ed., 1993, p. 854].
Reactivity Profile
Dimethyl ether is a colorless, highly flammable gas (b. p. -24° C), slightly toxic. Very dangerous fire and explosion hazard when exposed to flame, sparks, heat or strong oxidizers. Violent reaction with aluminum hydride, lithium aluminum hydride. Upon standing and exposure to air (oxygen) tendency to form explosive peroxides. When ethers containing peroxides are heated (distilled) they can detonate [Lewis, 3rd ed., 1993, p. 854].
Health Hazard
Methyl ether produced low inhalation toxicity in rats. Caprino and Togna (1975)reported a 30-minute LC50 value of 396 ppmfor rats. In lethal doses it caused sedation,a gradual depression of motor activity, lossof the sighting reflex, hypopnea, coma, anddeath in mice. Exposure to a 40% mixtureof methyl ether in air resulted in an initialslight increase in heart rate in rabbits, whichwas followed by depression of arterial bloodpressure. Death occurred in 45 minutes. Thearterial and venous partial oxygen pressurewas found to decrease while the venous CO2pressure and the blood pH increased
Reuzel et al. (1981) reported that sub chronic inhalation of methyl ether in ratsdid not cause significant adverse effects.No noticeable effect on organ and bodyweights and no treatment-related changeswere observed. In humans, adverse healtheffect from inhalation of this compoundshould be minimal. However, inhalation ofexcessive quantities can produce intoxicationand loss of consciousness.
Fire Hazard
Behavior in Fire: Containers may explode. Vapors are heavier than air and may travel long distance to a source of ignition and flash back.
Chemical Reactivity
Reactivity with Water No reaction; Reactivity with Common Materials: No reaction; Stability During Transport: Stable; Neutralizing Agents for Acids and Caustics: Not pertinent; Polymerization: Not pertinent; Inhibitor of Polymerization: Not pertinent.
Pharmaceutical Applications
Dimethyl ether may be used as an aerosol propellant for topical aerosol formulations in combination with hydrocarbons and other propellants. Generally, it cannot be used alone as a propellant owing to its high vapor pressure. Dimethyl ether is a good solvent and has the unique property of high water solubility, compared to other propellants. It has frequently been used with aqueous aerosols. A coarse, wet, spray is formed when dimethyl ether is used as a propellant.
Dimethyl ether is also used as a propellant in cosmetics such as hair sprays,and in other aerosol products such as air fresheners and fly sprays.
Dimethyl ether is additionally used as a refrigerant.
Safety
Dimethyl ether may be used as a propellant and solvent in topical pharmaceutical aerosols, and is generally regarded as an essentially nontoxic and nonirritant material when used in such applications. However,inhalation of high concentrations of dimethyl ether vapor is harmful. Additionally, skin contact with dimethyl ether liquid may result in freezing of the skin and severe frostbite.
When used in topical formulations, dimethyl ether may exert a chilling effect on the skin, although if it is used as directed the propellant quickly vaporizes and is nonirritating.
LD50 (mouse, inhalation): 386000ppm/30min
LD50 (rat, inhalation): 308g/m3
Potential Exposure
Used as a refrigerant, solvent, propellant for aerosol sprays; and in making plastics.Incompatibilities: Flammable gas. Forms explosive mixturewith air. Forms unstable peroxides in containers that havebeen opened or remain in storage for more than 6 months.Peroxides can be detonated by friction, impact or heating.Violent reaction with strong oxidizers, aluminum hydride,lithium aluminum hydride. Keep away from heat, air,sunlight
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit. Iffrostbite has occurred, seek medical attention immediately;do NOT rub the affected areas or flush them with water. Inorder to prevent further tissue damage, do NOT attempt toremove frozen clothing from frostbitten areas. If frostbitehas NOT occurred, immediately and thoroughly wash contaminated skin with soap and water
Carcinogenicity
A lifetime study in rats did not produce cancer or clear, statistically significant evidence of chronic toxicity at 25,000 ppm of dimethyl ether.
Environmental Fate
DME released to the atmosphere would be expected to exist almost entirely in the vapor phase since the vapor pressure is 4450 mmHg at 25 ℃. It is susceptible to photooxidation via vapor phase reaction with photochemically produced hydroxyl radicals. An atmospheric half-life of 5.4 days has been calculated. It will also exhibit very highmobility in soil and, therefore, itmay leach to groundwater. If DME is released to water, it will not be expected either to significantly absorb to sediment or suspended particulate matter, bioconcentrate in aquatic organisms, or directly photolyze. No data concerning the biodegradation of DME in environmental media were located but many ethers are known to be resistant to biodegradation. DME would not be expected to bioconcentrate in aquatic organisms.
Storage
The liquefied gas is stable when used as a propellant. However, exposure to the air for long periods of time may result in explosive peroxides being slowly formed.
Solutions of liquid dimethyl ether should not be concentrated either by distillation or by evaporation. Dimethyl ether should be stored in tightly closed metal cylinders in a cool, dry place.
Shipping
This compound requires a shipping label of“FLAMMABLE GAS.” It falls in Hazard Class 2.1 andthere is no Packing Group listed
Purification Methods
Dry methyl ether by passing over alumina and then BaO, or over CaH2, followed by fractional distillation at low temperatures. Its solubility is 37mL per mL of H2O at 18o, and it is very soluble
Toxicity evaluation
Higher concentrations of DME act on the central nervous system (CNS) to produce narcosis. The effects are rapidly reversible which is consistent with the very rapid bioelimination of the molecule. DME has, in the past, been considered for use as a human anesthetic. It should be noted that this chemical can produce cardiac sensitization similar to the effects of epinephrine.
Incompatibilities
Dimethyl ether is an aggressive solvent and may affect the gasket materials used in aerosol packaging. Oxidizing agents, acetic acid, organic acids, and anhydrides should not be used with dimethyl ether.
Regulatory Status
Included in the FDA Inactive Ingredients Database (topical aerosols). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.
Properties of Dimethyl ether
| Melting point: | −141 °C(lit.) |
| Boiling point: | −24.8 °C(lit.) |
| Density | 1.617 |
| vapor density | 1.62 (vs air) |
| vapor pressure | >760 mm Hg ( 25 °C) |
| refractive index | 1.2984 |
| Flash point: | -41°C |
| solubility | Soluble in acetone, chloroform, ethanol (95%), ether, and 1 in 3 parts of water. Dimethyl ether is generally miscible with water, nonpolar materials, and some semipolar materials. For pharmaceutical aerosols, ethanol (95%) is the most useful cosolvent. Glycols, oils, and other similar materials exhibit varying degrees of miscibility with dimethyl ether. |
| appearance | colorless gas |
| Odor | Chloroform-like; sweet. |
| explosive limit | 27% |
| Odor Threshold | 500ppm |
| Water Solubility | soluble |
| Merck | 6071 |
| BRN | 1730743 |
| Dielectric constant | 5.0(25℃) |
| Stability: | Stable. Extremely flammable. Note low flash point. May form explosive mixtures with air. May form peroxides during prolonged storage. |
| CAS DataBase Reference | 115-10-6(CAS DataBase Reference) |
| NIST Chemistry Reference | Dimethyl ether(115-10-6) |
| EPA Substance Registry System | Dimethyl ether (115-10-6) |
Safety information for Dimethyl ether
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Gas Cylinder Compressed Gases GHS04 |
| GHS Hazard Statements |
H220:Flammable gases H280:Gases under pressure |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P377:Leaking gas fire: Do not extinguish, unless leak can be stopped safely. P381:Eliminate all ignition sources if safe to do so. P410+P403:Protect from sunlight. Store in a well-ventilated place. |
Computed Descriptors for Dimethyl ether
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran



You may like
-
 115-10-6 Dimethyl ether 98%View Details
115-10-6 Dimethyl ether 98%View Details
115-10-6 -
 Dimethyl ether CAS 115-10-6View Details
Dimethyl ether CAS 115-10-6View Details
115-10-6 -
 Dimethyl ether CAS 115-10-6View Details
Dimethyl ether CAS 115-10-6View Details
115-10-6 -
 Dimethyl ether CAS 115-10-6View Details
Dimethyl ether CAS 115-10-6View Details
115-10-6 -
 3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details
3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details -
 20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 -
 3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details
3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details -
 57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4
