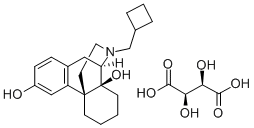
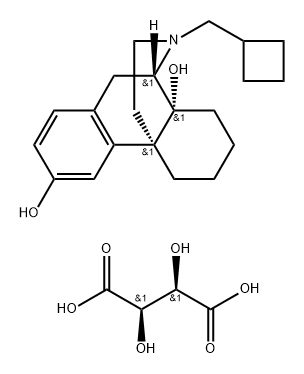
BUTORPHANOL
- CAS NO.:42408-82-2
- Empirical Formula: C21H29NO2
- Molecular Weight: 327.46
- MDL number: MFCD00864249
- EINECS: 255-808-8
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-05-04 15:14:15
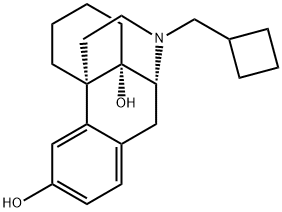
What is BUTORPHANOL?
Absorption
Rapidly absorbed after intramuscular injection and peak plasma levels are reached in 20-40 minutes. The absolute bioavailability is 60-70% and is unchanged in patients with allergic rhinitis. In patients using a nasal vasoconstrictor (oxymetazoline) the fraction of the dose absorbed was unchanged, but the rate of absorption was slowed. Oral bioavailability is only 5-17% because of extensive first-pass metabolism.
Toxicity
The clinical manifestations of butorphanol overdose are those of opioid drugs in general. The most serious symptoms are hypoventilation, cardiovascular insufficiency, coma, and death.
Originator
Stadol,Bristol-Myers,US,1978
The Uses of BUTORPHANOL
Mixed opioid agonist-antagonist. Analgesic (narcotic); antitussive. Controlled substance.
Background
A synthetic morphinan analgesic with narcotic antagonist action. It is used in the management of severe pain.
Indications
For the relief of moderate to severe pain.
Definition
ChEBI: Levorphanol in which a hydrogen at position 14 of the morphinan skeleton is substituted by hydroxy and one of the hydrogens of the N-methyl group is substituted by cyclopropyl. A semi-synthetic opioid agonist-antagonist analgesic, it is sed as its (S,S)-tartaric acid salt for relief or moderate to severe pain.
Manufacturing Process
A mixture of 1.0 g (2.58 mmols) of N-cyclobutylmethyl-14-hydroxy-9- methoxymorphinan and 10 ml of 48% HBr was refluxed, under a nitrogen atmosphere, during five minutes. After cooling, the reaction mixture was diluted with water and made basic with aqueous ammonium hydroxide. The aquous basic mixture was extracted with chloroform and the combined chloroform extracts were dried over anhyrous sodium sulfate. After evaporation of the solvent, the residual oil (730 mg) was taken up in dry ether and the resulting solution filtered through celite-charcoal. The filtrate was treated with a saturated solution of hydrogen chloride in dry ether. The hydrochloride salt thus obtained was collected by filtration and recrystallized from a methanol-acetone mixture to yield 565 mg (56.5%)of Butorphanol hydrochloride crystals melting at 272°-274°C (decomposition).
brand name
Stadol (Bristol-Myers Squibb).
Therapeutic Function
Analgesic, Antitussive
Biological Functions
Butorphanol (Stadol) is chemically related to levorphanol
but pharmacologically similar in action to pentazocine.
As an opioid antagonist it is nearly 30 times as
potent as pentazocine and has one-fortieth the potency
of naloxone. It is a potent opioid analgesic indicated for
the relief of moderate to severe pain. Its potency is 7
times that of morphine and 20 times that of pentazocine
as an analgesic. Its onset of action is similar to that of
morphine.The side effects and signs of toxicity are similar
to those produced by pentazocine. It produces excitatory
effects and sedation and precipitates withdrawal
in opioid-dependent patients. Although generally administered
parenterally because of its low bioavailability
following oral administration, it is also unique in that
a nasal spray formulation is available.The nasal spray is
indicated for the relief of postoperative pain and migraine
headache. The low molecular weight of butorphanol,
its high lipophilicity, and its lack of vasoconstrictor
effects make it particularly suitable for nasal
administration.
Nasal administration of butorphanol decreases the
onset of action to 15 minutes and decreases the firstpass
effect of the drug, which increases bioavailability.
Generally the patient sprays a set dose of 1 mg per hour
for 2 hours. The duration of action is 4 to 5 hours.
The convenience of such administration is a major advantage to patients requiring repeat dosing. The
abuse potential following such administration has not
been extensively studied, although it is thought to be
small. Butorphanol is not a federally controlled
(“scheduled”) drug, so physicians are not required to
obtain the licenses and security safeguards required for
other opioid analgesics.
Adverse effects, contraindications, and drug interactions
are similar to those for pentazocine and morphine.
General Description
Structurally, butorphanol is a morphinan and shares the samecyclobutyl methyl group on the nitrogen as nalbuphine. Likenalbuphine, butorphanol is an agonist at the κ-receptor but atthe μ-receptor butorphanol is both a partial agonist and anantagonist. The affinity for opioid receptors in vitro is1:4:25 for the μ-, δ-, and κ-receptors respectively. Thehigh affinity for the κ-receptors is proposed to give butorphanolits analgesic properties and is also responsible for theCNS adverse effects such as hallucinations, psychosis, andparanoid reactions. Butorphanol binds with μ-receptors as apartial agonist, and administration to humans maintained onhigh-potency μ-agonists such as morphine may precipitatewithdrawal. Butorphanol was found to produce convulsionsin morphine-deprived, morphine-dependent monkeys.
Pharmacokinetics
Butorphanol is a synthetic opioid agonist-antagonist analgesic with a pharmacological and therapeutic profile that has been well established since its launch as a parenteral formulation in 1978. The introduction of a transnasal formulation of butorphanol represents a new and noninvasive presentation of an analgesic for moderate to severe pain. This route of administration bypasses the gastrointestinal tract, and this is an advantage for a drug such as butorphanol that undergoes significant first-pass metabolism after oral administration. The onset of action and systemic bioavailability of butorphanol following transnasal delivery are similar to those after parenteral administration. Butorphanol blocks pain impulses at specific sites in the brain and spinal cord.
Metabolism
Extensively metabolized in the liver. The pharmacological activity of butorphanol metabolites has not been studied in humans; in animal studies, butorphanol metabolites have demonstrated some analgesic activity.
Properties of BUTORPHANOL
| Melting point: | 215-217° |
| Boiling point: | 507.3±50.0 °C(Predicted) |
| alpha | D -70.0° (c = 0.1 in methanol) |
| Density | 1.24±0.1 g/cm3(Predicted) |
| pka | pKa 8.6 (Uncertain) |
Safety information for BUTORPHANOL
Computed Descriptors for BUTORPHANOL
New Products
4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium SODIUM AAS SOLUTION ZINC AAS SOLUTION BUFFER SOLUTION PH 10.0(BORATE) GOOCH CRUCIBLE SINTERED AQUANIL 5 BERYLLIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran
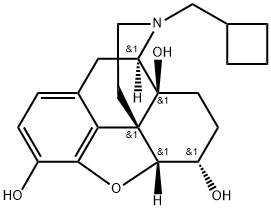
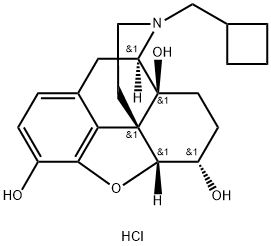
![17-[CYCLOBUTYLMETHYL]-4,5-EPOXYMORPHINAN-3,6,14-TRIOL HYDROCHLORIDE, DIHYDRATE](https://img.chemicalbook.in/CAS/GIF/59052-16-3.gif)


You may like
-
 1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
99903-60-3 -
 88491-46-7 98%View Details
88491-46-7 98%View Details
88491-46-7 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
1805639-70-6 -
 1784294-80-9 98%View Details
1784294-80-9 98%View Details
1784294-80-9 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4