Acetylcholine chloride
Synonym(s):Acetylcholine chloride;ACh
- CAS NO.:60-31-1
- Empirical Formula: C7H16ClNO2
- Molecular Weight: 181.66
- MDL number: MFCD00011698
- EINECS: 200-468-8
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-01-27 09:38:02
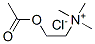
What is Acetylcholine chloride?
Description
Acetylcholine is a neurotransmitter that binds to nicotinic and muscarinic acetylcholine receptors (AChRs) in the central and peripheral nervous systems. It mediates motor function at the neuromuscular junction but also has functions in the parasympathetic and sympathetic nervous systems. It is involved in learning and memory through actions at nicotinic AChRs in the CNS. The actions of acetylcholine are terminated primarily via the action of acetylcholinesterase, which breaks it down into acetate and choline. Acetylcholine (chloride) mimics the effects of acetylcholine and has been used to determine the function of acetylcholine in various biological processes. Acetylcholine (chloride) inhibits peptide aggregation of p53 mutants in vitro at micromolar concentrations. It increases alveolar fluid clearance in a dose-dependent manner and enhances Na+/K+-ATPase activity, effects which are blocked by atropine , in a mouse model of pulmonary edema.
Chemical properties
White or almost white crystalline powder or colourless crystals, very hygroscopic.
The Uses of Acetylcholine chloride
Acetylcholine Chloride is a cholinergic.
The Uses of Acetylcholine chloride
cholinergic, antiarrhythmic, miotic, vasodilator (peripheral)
The Uses of Acetylcholine chloride
endogenous neurotransmitter at cholinergic synapses; amplifies sarcolemma action potential inducing muscle contractions
The Uses of Acetylcholine chloride
A cholinergic neurotransmitter that can induce the opening of calcium channels
What are the applications of Application
Acetylcholine chloride is a cholinergic neurotransmitter that binds to nicotinic and muscarinic acetylcholine receptors and can induce the opening of calcium channels.
Definition
ChEBI: The chloride salt of acetylcholine, and a parasympatomimetic drug.
brand name
Miochol (Novartis).
General Description
ACh chloride exerts a powerfulstimulant effect on the parasympathetic nervous system.Attempts have been made to use it as a cholinergic agent,but its duration of action is too short for sustained effects,because of rapid hydrolysis by esterases and lack of specificitywhen administered for systemic effects. It is a cardiacdepressant and an effective vasodilator. Stimulation of thevagus and the parasympathetic nervous system produces atonic action on smooth muscle and induces a flow from thesalivary and lacrimal glands. Its cardiac-depressant effectresults from (a) a negative chronotropic effect that causes adecrease in heart rate and (b) a negative inotropic action onheart muscle that produces a decrease in the force of myocardialcontractions.
Biological Activity
Endogenous neurotransmitter. Acts at nicotinic and muscarinic acetylcholine receptors.
Biochem/physiol Actions
Acetylcholine chloride, injected at 20 mg/kg body weight, reduces mortality and plasma proinflammatory cytokines in mice with experimentally-induced sepsis . The cholinergic anti-inflammatory mechanism is probably mediated by interaction of acetylcholine with α7n cholinoreceptor on monocytes, macrophages, and neutrophils, which decreases the levels of proinflammatory cytokines such as TNF-α, IL-1β, and IL-6.
Clinical Use
Atropine blocks the depressant effect of ACh on cardiacmuscle and its production of peripheral vasodilation (i.e.,muscarinic effects) but does not affect the skeletal musclecontraction (i.e., nicotinic effect) produced.ACh chloride is a hygroscopic powder that is available inan admixture with mannitol to be dissolved in sterile waterfor injection shortly before use. It is a short-acting mioticwhen introduced into the anterior chamber of the eye and isespecially useful after cataract surgery during the placementof sutures. When applied topically to the eye, it has littletherapeutic value because of poor corneal penetration andrapid hydrolysis by AChE.
Safety Profile
Poison by subcutaneous, intravenous, and parenteral routes. Moderately toxic by ingestion. When heated to decomposition it emits very toxic fumes of NOx, and Cl-. A cholinergic agent. See also CHOLINE ACETATE (ESTER).
storage
Desiccate at RT
Purification Methods
It is very soluble in H2O (>10%), and is very hygroscopic. If pasty, dry it in a vacuum desiccator over H2SO4 until a solid residue is obtained. Dissolve this in absolute EtOH, filter it and add dry Et2O, when the hydrochloride separates. Collect by filtration and store it under very dry conditions. [Jones & Major J Am Chem Soc 52 307 1930.] The chloroplatinate crystallises from hot H2O in yellow needles and can be recrystallised from 50% EtOH, m 242-244o [Dudley Biochem J 23 1069 1929]; other m given is 256-257o. The perchlorate crystallises from EtOH as prisms m 116-117o. [J Am Pharm Assocn 36 272 1947, Beilstein 4 IV 1446.]
Properties of Acetylcholine chloride
| Melting point: | 146-150 °C (lit.) |
| Density | 1.1235 (rough estimate) |
| refractive index | 1.5560 (estimate) |
| storage temp. | room temp |
| solubility | H2O: 0.1 g/mL, clear, colorless |
| form | Crystalline Powder |
| color | White |
| Water Solubility | Soluble IN COLD WATER |
| Sensitive | Hygroscopic |
| Merck | 14,87 |
| BRN | 3571875 |
| Stability: | Stable. Substances to be avoided include strong oxidizing agents. Protect from moisture - very hygroscopic. |
| CAS DataBase Reference | 60-31-1(CAS DataBase Reference) |
| EPA Substance Registry System | Ethanaminium, 2-(acetyloxy)-N,N,N-trimethyl-, chloride (60-31-1) |
Safety information for Acetylcholine chloride
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H303:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P280:Wear protective gloves/protective clothing/eye protection/face protection. P304+P340:IF INHALED: Remove victim to fresh air and Keep at rest in a position comfortable for breathing. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. P405:Store locked up. |
Computed Descriptors for Acetylcholine chloride
Acetylcholine chloride manufacturer
Anand Agencies
New Products
Cycloleucine 1-Amino-1-cyclohexanecarboxylic acid 6-Bromo-3-iodo-1-methyl-1H-indazole 3-(2,4-Dimethoxybenzyl)dihydropyrimidine-2,4(1H,3H)-dione 7-Bromo-1H-indazole ELECTROLYTIC IRON POWDER 1-(2-Ethoxyethyl)-2-(piperidin-4-yl)-1H-benzo[d]imidazole hydrochloride Decanonitrile tert-butyl 4-(1H-benzo[d]iMidazol-2-yl)piperidine-1-carboxylate N,N'-diallyl-1,3-diaminopropanedihydrochloride 4-Ethylbenzylamine N-(5-Amino-2-methylphenyl)acetamide 4-bromo-3,5-dimethylbenzenesulfonyl chloride 5-Bromo-2,3-dimethoxypyridine 2-Chloro-3-nitropyridine 2-methoxy-4-methyl-5-nitro pyridine 2-iodo-5-bromo pyridine methyl 6-chloro-2-(chloromethyl)nicotinate 2-amino-4-methyl-5-nitro pyridine 5-Fluoro-2-Oxindole (9H-fluoren-9-yl)methyl carbonochloridate 2-methyl-5-nitroaniline Diethyl phosphite 8-hydroxy-2,2,14,14-tetramethylpentadecanedioic acidRelated products of tetrahydrofuran


You may like
-
 Acetylcholine Chloride 98%View Details
Acetylcholine Chloride 98%View Details -
 Acetylcholine chloride, GR 99%+ CAS 60-31-1View Details
Acetylcholine chloride, GR 99%+ CAS 60-31-1View Details
60-31-1 -
 Acetylcholine chloride CAS 60-31-1View Details
Acetylcholine chloride CAS 60-31-1View Details
60-31-1 -
 Acetylcholine chloride 99% CAS 60-31-1View Details
Acetylcholine chloride 99% CAS 60-31-1View Details
60-31-1 -
 Acetylcholine chloride, practical grade CAS 60-31-1View Details
Acetylcholine chloride, practical grade CAS 60-31-1View Details
60-31-1 -
 Acetylcholine Chloride CAS 60-31-1View Details
Acetylcholine Chloride CAS 60-31-1View Details
60-31-1 -
 ACETYL CHOLINE CHLORIDE AR CAS 60-31-1View Details
ACETYL CHOLINE CHLORIDE AR CAS 60-31-1View Details
60-31-1 -
 Acetylcholine chloride CAS 60-31-1View Details
Acetylcholine chloride CAS 60-31-1View Details
60-31-1