(1S,4Z,7S,10S,11E,20R)-4-ethylidene-7,20-dipropan-2-yl-9-oxa-15,16-dit hia-3,6,18,21-tetrazabicyclo[8.7.6]tricos-11-ene-2,5,8,19,22-pentone
Synonym(s):Cyclo[(2Z)-2-amino-2-butenoyl-L-valyl-(3S,4E)-3-hydroxy-7-mercapto-4-heptenoyl-D-valyl-D-cysteinyl] cyclic (35)-disulfide;Depsipeptide;FK228;FR901228;L-Valine, N-((3S,4E)-3-hydroxy-7-mercapto-1-oxo-4-heptenyl)-D-valyl-D-cysteinyl-(2Z)-2-amino-2-butenoxyl-, (4-1)-lactone, cyclic (1-2)-disulfide
- CAS NO.:128517-07-7
- Empirical Formula: C24H36N4O6S2
- Molecular Weight: 540.7
- MDL number: MFCD18433404
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-16 21:30:20
![(1S,4Z,7S,10S,11E,20R)-4-ethylidene-7,20-dipropan-2-yl-9-oxa-15,16-dit hia-3,6,18,21-tetrazabicyclo[8.7.6]tricos-11-ene-2,5,8,19,22-pentone Structural](https://img.chemicalbook.in/CAS/GIF/128517-07-7.gif)
What is (1S,4Z,7S,10S,11E,20R)-4-ethylidene-7,20-dipropan-2-yl-9-oxa-15,16-dit hia-3,6,18,21-tetrazabicyclo[8.7.6]tricos-11-ene-2,5,8,19,22-pentone?
Absorption
Romidepsin exhibited linear pharmacokinetics at standard doses.
Toxicity
Risk factor D in pregnancy. It is not known if romidepsin is excreted in breast milk. Due to the potential for serious adverse reactions in the nursing infant, the manufacturer recommends a decision be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of treatment to the mother. The majority of patients receiving romidepsin experience nausea, vomiting, and anorexia.
Description
The U.S. FDA approved romidepsin (also referredto as FK228) in 2009 for the treatment of cutaneous T-cell lymphoma (CTCL) for patients who received at least one systemic therapy. Romidepsin is a natural product that was first isolated from the fermentation broth of C. violaceum. Romidepsin is the second histone deacetylase (HDAC) inhibitor approved for CTCL, the other being vorinostat,whichwas approved by the FDA in 2006. Unlike vorinostat which is a pan-HDAC inhibitor, romidepsin shows modest selectivity for class I HDACs in in vitro assays. It has been shown that after romidepsin enters the cytoplasm, the disulfide bond is cleaved by glutathione to release the sulfhydryl groupwhich chelateswith the activesite zinc of class IHDACs and inhibits the enzymatic activity at nanomolar concentrations. Although romidepsin inhibits class I HDACs, it is 17–23 times less potent as the parent than the corresponding reduced form at each isozyme. For example, the IC50 of romidepsin at HDAC1 is 36±16nM while that of the reduced form is IC50=1.6± 0.9nM.Romidepsinhasalsobeenshownto induce cell cycle arrest, differentiation, and apoptosis in tumor cells by mechanisms that cannot be completely explained by HDAC inhibition alone.
Chemical properties
Pale Yellow Solid
Originator
Fujisawa (Astellas Pharma) (Japan)
The Uses of (1S,4Z,7S,10S,11E,20R)-4-ethylidene-7,20-dipropan-2-yl-9-oxa-15,16-dit hia-3,6,18,21-tetrazabicyclo[8.7.6]tricos-11-ene-2,5,8,19,22-pentone
Labelled Romidepsin (R425060). Romidepsin is a histone deacetylase inhibitor that can alter chromatin structure and gene transcription leading to multiple changes in cellular protein production. This may result in cell cycle arrest and tumor growth inhibition. Romidepsin has shown anti-proliferative activity in vitro against multiple mouse and human tumor cell lines and in vivo in human tumor xeno graft models. Romidepsin can be administered with a second agent, such as a cytotoxic agent, a steroidal agent, a proteasome inhibitor, or a kinase inhibitor.
The Uses of (1S,4Z,7S,10S,11E,20R)-4-ethylidene-7,20-dipropan-2-yl-9-oxa-15,16-dit hia-3,6,18,21-tetrazabicyclo[8.7.6]tricos-11-ene-2,5,8,19,22-pentone
Romidepsin is a histone deacetylase inhibitor that can alter chromatin structure and gene transcription leading to multiple changes in cellular protein production. This may result in cell cycle arrest and tumor growth inhibition. Romidepsin has shown anti-proliferative activity in vitro against multiple mouse and human tumor cell lines and in vivo in human tumor xenograft models. Romidepsin can be administered with a second agent, such as a cytotoxic agent, a steroidal agent, a proteasome inhibitor, or a kinase inhibitor.
Background
Romidepsin is a selective inhibitor of histone deacetylase, approved in the US in 2009 for the treatment of cutaneous T-cell lymphoma (CTCL) in patients who have received at least one prior systemic therapy.
Indications
Romidepsin is indicated for the treatment of cutaneous T-cell lymphoma (CTCL) in adult patients who have received at least one prior systemic therapy.
What are the applications of Application
Romidepsin is an HDAC1, HDAC2, HDAC4, and HDAC6 inhibitor
Definition
ChEBI: A cyclodepsipeptide consisting of the cyclic disulfide of (2Z)-2-aminobut-2-enoyl, L-valyl, (3S,4E)-3-hydroxy-7-sulfanylhept-4-enoyl, D-valyl and D-cystei yl residues coupled in sequence and cyclised head-to tail.
brand name
Chromadax (Gloucester);Istodax.
Biochem/physiol Actions
Romidepsin is a very potent natural prodrug inhibitor of HDAC1 and HDAC2 that is converted to active form by glutathione. Romidepsin has IC50 values of 36 nM and 47 nM for HDAC1 and HDAC2, respectively. Romidepsin kills lymphoma cell lines overexpressing Bcl-2 and Bcl-XL, and has been approved for the treatment for cutaneous T-cell lymphoma (CTCL) and peripheral T-cell lymphoma, and a variety of other cancers.
Clinical Use
Romidepsin, a histone deacetylase inhibitor, originally developed by Fujisawa (now Astellas Pharma), causes cell cycle arrest, differentiation, and apoptosis in various cancer cells. In 2004, the FDA granted fast-track designation for romidepsin as monotherapy for the treatment of cutaneous Tcell lymphoma (CTCL) in patients who have relapsed following, or become refractory to, other systemic therapies. The FDA designated romidepsin as an orphan drug and it was approved in 2009 for this indication and it was commercialized in 2010. In 2007, another fast-track designation was granted for the product as monotherapy of previously treated peripheral T-cell lymphoma. Romidepsin (FR901228) was originally discovered and isolated from the fermentation broth of Chromobacterium violaceum No. 968. It was identified through efforts in the search for novel agents which selectively reverse the morphological phenotype of Ras oncogene-transformed cells since the Ras signaling pathway plays a critical role in cancer development. Therefore, the drug could also have multiple molecular targets for its anticancer activity besides HDAC. FR901228 is a bicyclic depsipeptide which is structurally unrelated to any known class of cyclic peptides with an unusual disulfide bond connecting a thiol and Dcysteine.
Synthesis
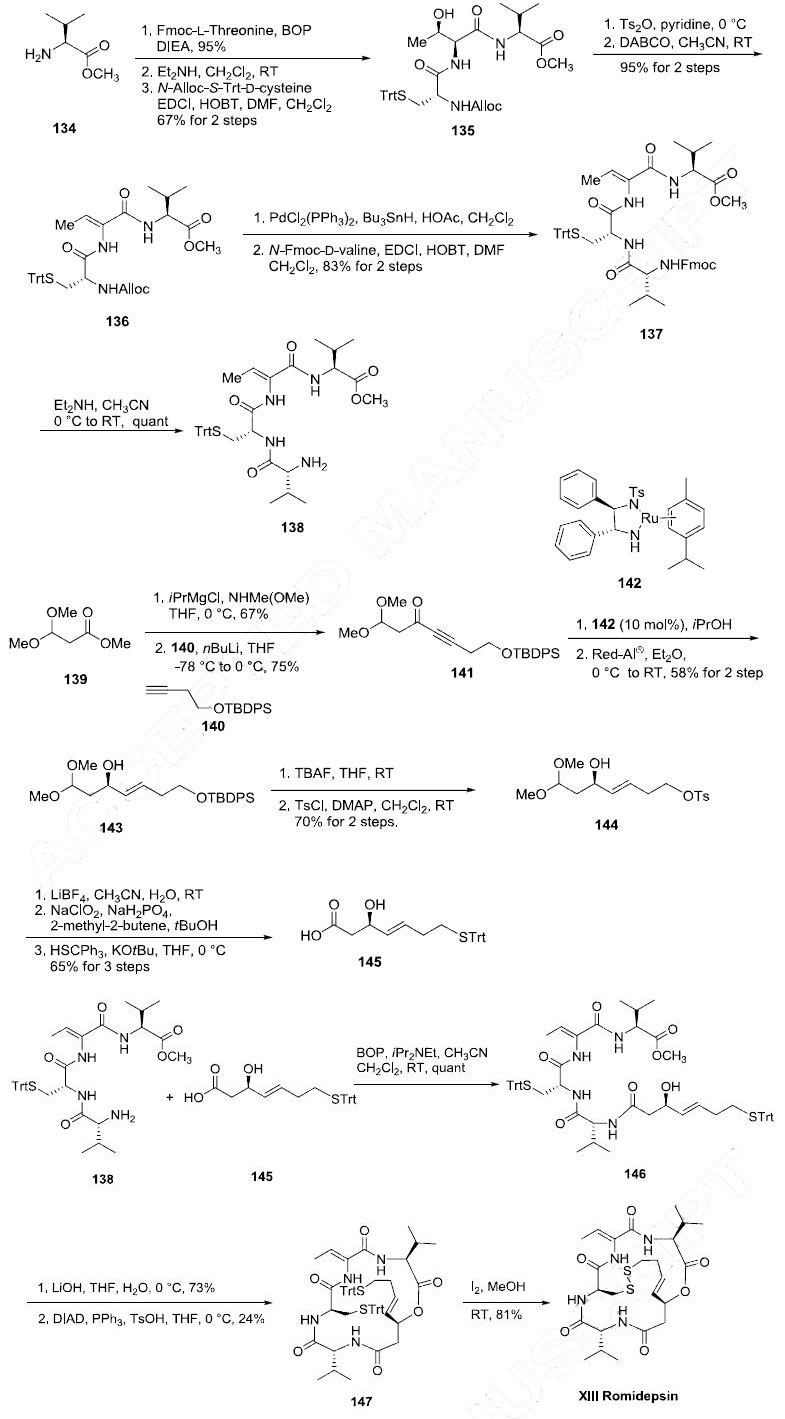
Romidepsin is commercially produced by fermentation; however its interesting and novel structure warrants examination of its synthesis within the context of this review. The synthesis of romidepsin described is based on the total synthesis reported by the Williams and Simon groups. L-Valine methyl ester (134) was coupled to N-Fmoc-L-threonine in the presence of the BOP reagent in 95% yield. The N-Fmoc protecting group was removed with Et2NH and the corresponding free amine was coupled to N-alloc-(S-triphenylmethyl)-D-cysteine with 1-ethyl-3- (3-dimethylaminopropyl)carbodiimide (EDCI) and HOBT in DMF and CH2Cl2 to yield the tripeptide 135 in good yield. The threonine residue of tripeptide 135 was then subjected to dehydrating conditions to give alkene 136 in 95% yield. The N-alloc protecting group of the dehydrated tripeptide 136 was removed with palladium and tin reagents and the corresponding free amine was subsequently coupled with N-Fmoc-D-valine to give tetrapeptide 137 in 83% yield. After removal of the N-Fmoc protecting group of compound 137 with Et2NH amine 138 was obtained in quantitative yield. The acid coupling partner 145 for amine 138 was prepared as follows: methyl 3,3-dimethoxypropionate (139) was converted to its corresponding Weinreb amide by standard conditions and reacted with lithium acetylide 140 to give propargylic ketone 141 in 75% yield. Noyori?ˉs asymmetric reduction of ketone 141 using ruthenium catalyst 142 gave the (R)-propargylic alcohol in 98% ee. This was followed by Red-Al reduction of the alkyne to selectively yield (E)-alkene 143 in 58% yield for the two steps. Liberation of the primary alcohol with tetrabutylammonium fluoride (TBAF) followed by selective tosylation gave 144 in 70% yield in two steps. Hydrolysis of the dimethyl acetal of 144 with LiBF4 was followed by a Pinnick oxidation to give the corresponding carboxylic acid. The tosylate was displaced with trityl mercaptan in the presence of tert-butyl alcohol to give allylic alcohol 145 in 65% yield for the three steps. Aminoamide 138 was then coupled to acid 145 using BOP to give peptide 146 in quantitative yield. The methyl ester of compound 146 was hydrolyzed with lithium hydroxide to provide the free carboxylic acid which underwent macrolactonization under Mitsunobu conditions in the presence of diisopropyl azodicarboxylate (DIAD) and triphenylphosine to give macrocycle 147 in 24% yield. Finally, the disulfide linkage was formed by treating bis-tritylsulfane 147 with iodine in methanol at room temperature to give romidepsin (XIII) in 81% yield.
Metabolism
Romidepsin undergoes extensive hepatic metabolism in vitro primarily by CYP3A4 with minor contribution from CYP3A5, CYP1A1, CYP2B6 and CYP2C19.
Storage
-20°C
References
1) Furumai et al. (2002), FK228 (depsipeptide) as a natural prodrug that inhibits class I histone deacetylases; Cancer Res., 62 4916 2) Panicker et al. (2010), Romidepsin (FK228/depsipeptide) controls growth and induces apoptosis in neuroblastoma tumor cells; Cell Cycle, 9 1830 3) Ueda et al. (1994), FR901228, a novel anti-tumor bicyclic depsipeptide produced by Chromobacterium violaceum No. 968. III. Antitumor activities on experimental tumors in mice; J. Antibiot. (Tokyo), 47 315 4) VanderMolin et al. (2011), Romodepsin (Istodax, NSC 630176, FR901228, FK228, depsipeptide): a natural product recently approved for cutaneous T-cell lymphoma; J. Antibiotic. (Tokyo), 64 525
Properties of (1S,4Z,7S,10S,11E,20R)-4-ethylidene-7,20-dipropan-2-yl-9-oxa-15,16-dit hia-3,6,18,21-tetrazabicyclo[8.7.6]tricos-11-ene-2,5,8,19,22-pentone
| Melting point: | 219-224°C |
| Boiling point: | 942.8±65.0 °C(Predicted) |
| Density | 1.174 |
| storage temp. | -20°C |
| solubility | Soluble in DMSO (up to 10 mg/ml). |
| pka | 11.33±0.60(Predicted) |
| form | powder |
| color | white to beige |
| Stability: | Stable for 2 years from date of purchase as supplied. Solutions in DMSO may be stored at -20° for up to 1 month. |
Safety information for (1S,4Z,7S,10S,11E,20R)-4-ethylidene-7,20-dipropan-2-yl-9-oxa-15,16-dit hia-3,6,18,21-tetrazabicyclo[8.7.6]tricos-11-ene-2,5,8,19,22-pentone
Computed Descriptors for (1S,4Z,7S,10S,11E,20R)-4-ethylidene-7,20-dipropan-2-yl-9-oxa-15,16-dit hia-3,6,18,21-tetrazabicyclo[8.7.6]tricos-11-ene-2,5,8,19,22-pentone
New Products
tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Boc-Arg-OH HCl H2O Indole Methyl Resin 2-CTC Resin Gabapentin EP Impurity A 2,5-dichloro-N-hydroxy-4,6-dimethylpyridine-3-carboximidamide 4-Chloro-7-tosy1-7Hpyrrolo[2,3-d]pyrimidine 1,3-Diphenylurea 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID N-METHYL INDAZOLE-3-CARBOXYLIC ACID 2-Hydroxy-5-nitroacetophenone 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,4-dihydroxybenzaldehyde 1,3-Diethyl-1,3-Diphenylurea 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2-HYDROXY BENZYL CYANIDE DIETHYL AMINOMALONATE HYDROCHLORIDE 5-BROMO-2CYANO PYRIDINE Boldenone Clarithromycin Ethinyl Estradiol StanozololRelated products of tetrahydrofuran

You may like
-
 128517-07-7 Romidepsin 98%View Details
128517-07-7 Romidepsin 98%View Details
128517-07-7 -
 Romidepsin 98%View Details
Romidepsin 98%View Details
128517-07-7 -
 Romidepsin 98%View Details
Romidepsin 98%View Details
128517-07-7 -
 Romidepsin 99%View Details
Romidepsin 99%View Details -
 Romidepsin >95% CAS 128517-07-7View Details
Romidepsin >95% CAS 128517-07-7View Details
128517-07-7 -
 FK228 >95% CAS 128517-07-7View Details
FK228 >95% CAS 128517-07-7View Details
128517-07-7 -
 Romidepsin CAS 128517-07-7View Details
Romidepsin CAS 128517-07-7View Details
128517-07-7 -
 3-NITRO 2-METHYL BENZOIC ACID 98%View Details
3-NITRO 2-METHYL BENZOIC ACID 98%View Details
1975-50-4
