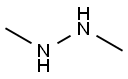
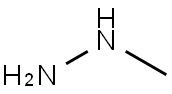
1,1-Dimethylhydrazine
Synonym(s):asym-Dimethylhydrazine;1,1-Dimethylhydrazine
- CAS NO.:57-14-7
- Empirical Formula: C2H8N2
- Molecular Weight: 60.1
- MDL number: MFCD00007628
- EINECS: 200-316-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 13:37:16
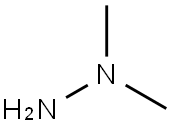
What is 1,1-Dimethylhydrazine?
Description
1,1-Dimethylhydrazine, frequently called unsymmetrical dimethylhydrazine (UDMH), is the third member of the hydrazine family to appear in Molecule of the Week. The first two were the parent molecule hydrazine in 2010 and methylhydrazine earlier this year. All these highly toxic hydrazines are used in rocket fuels.
Australian chemist Henry H. Hatt reported a synthesis of the hydrochloride salt1 of UDMH in the 1936 edition of Organic Syntheses. Hatt treated dimethylamine with nitric acid to produce the N-nitroso derivative, which was then reduced with zinc to form UDMH, isolated as the salt. Hatt later made UDMH by treating dimethylamine with chloramine; this is one of two current industrial production processes. In the other, acetylhydrazine is treated with formaldehyde and hydrogen to form a dimethyl derivative, which is then hydrolyzed to produce UDMH.
UDMH is a hypergolic rocket fuel, which means that it ignites when it is mixed with another component of the fuel. The second component is usually dinitrogen tetroxide. UDMH is generally preferred to hydrazine in rocket fuels because it autoignites at a higher temperature and is thus more stable in storage.
1. CAS Reg. No. 593-82-8.
Description
UDMH is a fuming colorless liquid that turnsyellow on contact with air, with a fishy, amine-like odor.The odor threshold is 6.114 ppm. Molecularweight= 60.12; Boiling point = 63.9℃; Freezing/Meltingpoint = 257.7℃; Vapor pressure= 103 mmHg at 20℃;Flash point = 215℃; Autoignition temperature= 249℃.Explosive limits: LEL = 2.0%; UEL= 95%. HazardIdentification (based on NFPA-704 M Rating System):Health 4, Flammability 3, Reactivity 1. Highly soluble inwater.
Chemical properties
1,1-Dimethylhydrazine is a colorless, flammable, hygroscopic liquid that gradually turns yellow on contact with air and is miscible with water. 1,1-Dimethylhydrazine is primar- ily used as a high-energy fuel in military applications, as a rocket propellant and fuel for thrusters, and small electrical power-generating units. 1,1-Dimethylhydrazine is also used in the manufacture of a plant growth regulator, in chemical synthesis, in photographic chemicals, as a stabilizer for fuel additives, and as an absorbent for acid gases. Exposure to 1,1-dimethylhydrazine usually occurs at the workplace during use and handling of the chemical substance. No information is available on the carcinogenic effects of 1,1-dimethylhydrazine in humans. Carcinogenic effects were observed in mice and rats exposed to 1,1-dimethylhydrazine by inhalation, but the carcinogenicity could not be defi nitively attributed to 1,1-dimethylhydrazine because of the presence of contaminants in the study. The US EPA has not classifi ed 1,1-dimethylhydrazine for potential carcinogenicity, while the IARC has classifi ed 1,1-dimethylhydrazine as Group 2B, meaning a possible human carcinogen.
Physical properties
Clear, colorless fuming liquid with an amine-like odor. Turns yellow on exposure to air. Odor detection threshold concentrations ranged from 6.1 to 14 ppmv (Jacobson et al., 1955).
The Uses of 1,1-Dimethylhydrazine
1,1-Dimethylhydrazine is used in rocket fuel.
The Uses of 1,1-Dimethylhydrazine
Base in rocket fuel formulations; intermediate in organic synthesis
The Uses of 1,1-Dimethylhydrazine
N,N-Dimethylhydrazine is used as a reagent in the synthesis of novel substituted pyridinylbenzenesulfonamide derivatives which have herbicidal activity. N,N-Dimethylhydrazine is also used in the preparation of the antibacterial agent, (-)-Platencin.
Definition
ChEBI: A member of the class of hydrazines that is hydrazine substituted by two methyl groups at position 1.
General Description
A clear colorless liquid with an ammonia-like odor. Flash point 0°F. Corrosive to the skin. Less dense than water and soluble in water. Vapors are heavier than air and very toxic by inhalation, attacking the eyes and respiratory system. Prolonged exposure of containers to heat may result in their violent rupturing and rocketing due to decomposition. Generates toxic oxides of nitrogen when burned. Vapors may travel to a source of ignition and a flame can flashback to the source of vapors. Used as a rocket propellant and to make other chemicals.
Air & Water Reactions
Highly flammable over a wide range of vapor concentrations. May ignite spontaneously when spread on a large surface exposed to the air. [Def. Res. and Eng., pp 299-300(1963)]. Dissolves and slowly decomposes in water.
Reactivity Profile
1,1-Dimethylhydrazine is a powerful reducing agent. Ignition can occur on contact with oxidizing agents such hydrogen peroxide and fuming nitric acid, [Haz. Chem. Data(1966)]. Also reacts as a base to neutralize acids in exothermic reactions.
Health Hazard
1,1-Dimethylhydrazine exhibits high acute toxicity as a result of exposure by all routes. Death or permanent injury may result after very short exposure to small quantities. Chronic exposure may cause pneumonia, liver damage, and kidney damage.
Health Hazard
The vapors of this compound are irritating tothe eyes, nose, and throat. The liquid is corrosive to the skin. The toxic properties aresimilar to those of 1,1-dimethylhydrazine.Exposure to 280 ppm for 4 hours was fatalto rats. The oral LD50 value in th.
Fire Hazard
Vapor may explode if ignited in an enclosed area. Vapors may travel to a source of ignition and flashback. Runoff to sewer may create fire or explosion hazard. When 1,1-Dimethylhydrazine decomposes, 1,1-Dimethylhydrazine gives off toxic nitrogen compound fumes. Dissolves, swells, and disintegrates many plastics. Dangerous when exposed to heat, flame, or oxidizers. Hazardous polymerization may not occur.
Safety Profile
Confirmed carcinogen with experimental carcinogenic, tumorigenic, and teratogenic data. Other experimental reproductive effects. Poison byingestion, intraperitoneal, intravenous, and intracerebral routes. Moderately toxic by inhalation and skin contact. Human mutation data reported. A plant growth control agent. Corrosive. A powerful reducing agent. A dangerous fire hazard. It is hypergolic with many oxidants (e.g., dinitrogen tetroxide, hydrogen peroxide, and nitric acid). Dangerous when exposed to heat, flame, or oxidizers; can react vigorously with oxidning materials such as air, fuming HNO3, (HNO3 + N2O4), NO. A high-energy propellant for liquid-fueled rockets. To fight fire, use alcohol foam, CO2, dry chemical. When heated to decomposition it emits highly toxic fumes of NOx. See also HYDRAZINE.
Potential Exposure
Tumorigen,Mutagen; Reproductive Effector; Human Data. This material is used as a component in liquid rocket propellant combinations; it is also used in photography; as an absorbent;and to make other chemicals.
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. If victim is conscious, administer water ormilk. Do not induce vomiting. Medical observation isrecommended for 2448 h after breathing overexposure, aspulmonary edema may be delayed. As first aid for pulmonary edema, a doctor or authorized paramedic may consideradministering a corticosteroid spray.
Carcinogenicity
1,1-Dimethylhydrazine is reasonably anticipated to be a human carcinogen based on sufficient evidence of carcinogenicity from studies in experimental animals.
Environmental Fate
Chemical/Physical. Releases toxic nitrogen oxides when heated to decomposition (Sax and
Lewis, 1987). Ignites spontaneously in air or in contact with hydrogen peroxide, nitric acid, or
other oxidizers (Patnaik, 1992).
N-Nitrosodimethylamine was the major product of ozonation of 1,1-dimethylhydrazine in the
dark. Hydrogen peroxide, methyl hydroperoxide, and methyl diazene were also identified (HSDB,
1989).
storage
(1) Color Code—Red: Flammability Hazard: Storein a flammable liquid storage area or approved cabinetaway from ignition sources and corrosive and reactivematerials. (2) Color Code—Yellow Stripe (strong reducingagent): Reactivity Hazard; Store separately in an area isolated from flammables, combustibles, or other yellow-codedmaterials. (3) Color Code—Blue: Health Hazard/Poison:Store in a secure poison location. Prior to working withUDMH you should be trained on its proper handling andstorage. Outside or detached storage is preferred. Beforeentering confined space where this chemical may be present, check to make sure that an explosive concentrationdoes not exist. Store in tightly closed containers in a cool,well-ventilated area. Keep dry and separate from porousmaterials, oxidizing agents, and other incompatible materials, some of which are listed above. Store in an inert atmosphere below 50℃. Do not use copper containers. Metalcontainers involving the transfer of this chemical should begrounded and bonded. Where possible, automatically pumpliquid from drums or other storage containers to processcontainers. Drums must be equipped with self-closingvalves, pressure vacuum bungs, and flame arresters. Useonly nonsparking tools and equipment, especially whenopening and closing containers of this chemical. Sources ofignition, such as smoking and open flames, are prohibitedwhere this chemical is used, handled, or stored in a mannerthat could create a potential fire or explosion hazard.Wherever this chemical is used, handled, manufactured, orstored, use explosion-proof electrical equipment and fittings. A regulated, marked area should be established wherethis chemical is handled, used, or stored in compliance withOSHA Standard 1910.1045.
Shipping
This compound requires a shipping label of“POISONOUS/TOXIC MATERIALS, POISON GAS,CORROSIVE.” It falls in Hazard Class 6.1 and PackingGroup I. Procedures for the handling, use, and storage ofcylinders should be in compliance with OSHA 1910.101and 1910.169 with the recommendations of the CompressedGas Association.
Purification Methods
Fractionally distil the hydrazine through a 4-ft column packed with glass helices. Precipitate it as its oxalate from diethyl ether solution. After crystallising from 95% EtOH, the salt is decomposed with aqueous saturated NaOH, and the free base is distilled, dried over BaO and redistilled [McBride & Kruse J Am Chem Soc 79 572 1957]. Distillation and storage should be under nitrogen. [Beilstein 4 IV 3322.]
Incompatibilities
Forms explosive mixture with air; mayspontaneously ignite on contact with air and porous materials, such as asbestos, wood, earth, cloth, etc. Reacts withoxygen causing fire and explosion hazard. A strong reducing agent, this chemical is incompatible with strong acids,halogens, metallic mercury, copper alloys, brass, iron, ironsalts. Contact with strong oxidizers, such as nitric acid,nitrogen tetroxide, hydrogen peroxide, may cause spontaneous ignition. A strong base, this chemical is corrosive andreacts violently with acids. Attacks some plastics, rubber,and coatings. May accumulate static electrical charges, andmay cause ignition of its vapors
Properties of 1,1-Dimethylhydrazine
| Melting point: | -57.2 °C |
| Boiling point: | 60-62 °C(lit.) |
| Density | 0.79 g/mL at 20 °C(lit.) |
| vapor density | 1.94 (vs air) |
| vapor pressure | 103 mm Hg ( 20 °C) |
| refractive index | n |
| Flash point: | 34 °F |
| storage temp. | 2-8°C |
| solubility | Miscible with alcohol, N,N-dimethylformamide, ether, hydrocarbons (Windholz et al., 1983), and
many other polar solvents. |
| form | Colorless liquid |
| appearance | colorless liquid |
| pka | 8.19±0.18(Predicted) |
| explosive limit | 95% |
| Water Solubility | miscible |
| Sensitive | Hygroscopic |
| Merck | 14,3247 |
| BRN | 605261 |
| Henry's Law Constant | 2.45(x 10-9 atm?m3/mol) at 25 °C (quoted, Mercer et al., 1990) |
| Exposure limits | TLV-TWA skin 0.5 ppm (1.0 mg/m3 )
(ACGIH, MSHA, and OSHA); carcinogenicity:
Animal Sufficient Evidence (IARC), Suspected Carcinogen (ACGIH). |
| CAS DataBase Reference | 57-14-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Hydrazine, 1,1-dimethyl-(57-14-7) |
| IARC | 2B (Vol. 4, Sup 7, 71) 1999 |
| EPA Substance Registry System | 1,1-Dimethylhydrazine (57-14-7) |
Safety information for 1,1-Dimethylhydrazine
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Corrosion Corrosives GHS05  Skull and Crossbones Acute Toxicity GHS06  Health Hazard GHS08  Environment GHS09 |
| GHS Hazard Statements |
H225:Flammable liquids H314:Skin corrosion/irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation H350:Carcinogenicity H411:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for 1,1-Dimethylhydrazine
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid 1-Bromo-3,5-Di-Tert-Butylbenzene S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran

You may like
-
 N,N-Dimethylhydrazine CAS 57-14-7View Details
N,N-Dimethylhydrazine CAS 57-14-7View Details
57-14-7 -
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1