TRI-AMMONIUM ORTHOPHOSPHATE
- CAS NO.:10124-31-9
- Empirical Formula: H6NO4P
- Molecular Weight: 115.02
- MDL number: MFCD00036305
- EINECS: 233-330-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:08:57
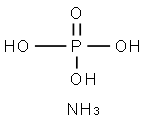
What is TRI-AMMONIUM ORTHOPHOSPHATE?
The Uses of TRI-AMMONIUM ORTHOPHOSPHATE
Large quantities of the ammonium phosphates are used as fertilizers and in fertilizer formulations. The compounds furnish both nitrogen and phosphorus essential to plant growth. The compounds also are used as fire retardants in wood building materials, paper and fabric products, and in matches to prevent afterglow. Solutions of the ammonium phosphates sometimes are air dropped to retard forest fires, serving the double purpose of fire fighting and fertilizing the soil to accelerate new plant growth. The compounds are used in baking powder formulations, as nutrients in the production of yeast, as nutritional supplements in animal feeds, for controlling the acidity of dye baths, and as a source of phosphorus in certain kinds of ceramics.
Production Methods
Ammonium phosphates usually are manufactured by neutralizing phosphoric acid with NH3. Control of the pH (acidity/alkalinity) determines which of the ammonium phosphates will be produced. Pure grades can be easily made by crystallization of solutions obtained from furnace grade phosphoric acid. Fertilizer grades, made from wet-process phosphoric acid, do not crystallize well and usually are prepared by a granulation technique. First, a highly concentrated solution or slurry is obtained by neutralization. Then the slurry is mixed with from 6× to 10× its weight of previously dried material, after which the mixture is dried in a rotary drier. The dry material is then screened to separate the desired product size. Oversize particles are crushed and mixed with fines from the screen operation and then returned to the granulation step where they act as nuclei for the production of further particles. Other ingredients often are added during the granulation of fertilizer grades. The ratio of nitrogen to phosphorus can be altered by the inclusion of ammonium nitrate, ammonium sulfate, or urea. Potassium salts sometimes are added to provide a 3-component fertilizer (N, P, K). A typical fertilizer grade diammonium phosphate will contain 18% N and 46% P2O5 (weight).
There has been a trend toward the production of ammonium phosphates in powder form. Concentrated phosphoric acid is neutralized under pressure, and the heat of neutralization is used to remove the water in a spray tower. The powdered product then is collected at the bottom of the tower. Ammonium nitrate/ammonium phosphate combination products can be obtained either by neutralizing mixed nitric acid and phosphoric acid, or by the addition of ammonium phosphate to an ammonium nitrate melt.
Properties of TRI-AMMONIUM ORTHOPHOSPHATE
| EPA Substance Registry System | Ammonium phosphate (10124-31-9) |
Safety information for TRI-AMMONIUM ORTHOPHOSPHATE
Computed Descriptors for TRI-AMMONIUM ORTHOPHOSPHATE
New Products
Tert-butyl bis(2-chloroethyl)carbamate (S)-3-Aminobutanenitrile hydrochloride N-Boc-D-alaninol N-BOC-D/L-ALANINOL N-octanoyl benzotriazole 4-Hydrazinobenzoic acid 3,4-Dibenzyloxybenzaldehyde Electrolytic Iron Powder 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 4-HYDROXY BENZYL ALCOHOL 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) S-2-CHLORO PROPIONIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 3-(Hydroxymethyl)benzoate N-Boc-2-chloroethylamine 1-Bromo-2-methoxy-3-nitrobenzene N-Methyl-3-cyclopenten-1-amine 2-Bromo-3-hydroxybenzaldehyde 1H-indazole-5-carboxamideRelated products of tetrahydrofuran

You may like
-
 7441-43-2 98%View Details
7441-43-2 98%View Details
7441-43-2 -
 1260741-78-3 6-Bromo-3-iodo-1-methyl-1H-indazole 98%View Details
1260741-78-3 6-Bromo-3-iodo-1-methyl-1H-indazole 98%View Details
1260741-78-3 -
 (3-Benzyloxypropyl)triphenyl phosphonium 98%View Details
(3-Benzyloxypropyl)triphenyl phosphonium 98%View Details
54314-85-1 -
 4-bromo-3,5-dimethylbenzenesulfonyl chloride 1581266-79-6 98%View Details
4-bromo-3,5-dimethylbenzenesulfonyl chloride 1581266-79-6 98%View Details
1581266-79-6 -
 2490430-37-8 98%View Details
2490430-37-8 98%View Details
2490430-37-8 -
 N-(5-Amino-2-methylphenyl)acetamide 5434-30-0 98%View Details
N-(5-Amino-2-methylphenyl)acetamide 5434-30-0 98%View Details
5434-30-0 -
 124371-59-1 98%View Details
124371-59-1 98%View Details
124371-59-1 -
 53857-52-2 98%View Details
53857-52-2 98%View Details
53857-52-2