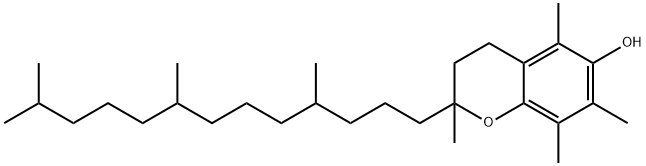
Tocopherol
- CAS NO.:1406-18-4
- Empirical Formula: C29H50O2
- Molecular Weight: 430.71
- MDL number: MFCD00072051
- EINECS: 215-798-8
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:15:30
What is Tocopherol?
Chemical properties
Viscous oils. Soluble in fats; insoluble in water. Stable to heat in the absence of oxygen, to strong acids, and to visible light; unstable to UV light, alkalies, and oxidation.
The Uses of Tocopherol
Fat-soluble vitamin E, which is a light yellow oil readily degradable by heat. As a vitamin, it is essential for normal muscle growth and prevents vitamin A destruction by deterioration. It also functions as an antioxidant. It prevents the oxidation of certain fatty acids and is stable unless the food becomes rancid. Vegetable oils contain a higher concentration of natural antioxidants, including tocopherols, than animal fats and are thus more stable. Tocopherol is obtained from vegetable oils, beans, eggs, and milk. It is also termed alpha-tocopherol.
The Uses of Tocopherol
Medicine, nutrition, antioxidants for fats, animal feed additive.
Definition
(vitamin E). Any of a group of related substances (α-, β-, γ-, andΔ-tocopherol) that constitute vitamin E. The α-form (which occurs naturally as the d-isomer) is the most potent. Occurs naturally in plants, especially wheat germ. All are derivatives of dihydrobenzo-γ-pyran and differ from each other only in the number and position of methyl groups. Vitamin E is required by certain rodents for normal reproduction. Muscular and central nervous system depletion along with generalized edema are deficiency symptoms in all animals. It is not required as a dietary supplement for humans.
Preparation
Tocopherol is synthesised in two ways. One is to extract it from natural plant sources and the other is to obtain it by chemical synthesis. The following are the chemical synthesis routes:
The most important natural raw materials for production of tocopherols by extraction are deodoriser sludges, which are distillates obtained in the deodorisation of vegetable oils. Such distillates contain sterols, sterol esters and triacylglycerols, as well as tocopherols and tocotrienols. The concentration of tocopherols depends on the deodorisation parameters (temperature, vacuum, quantity of injected steam and equipment) but their amount is lower than 10%, usually 8–9 %, of unsaponifiable matter present. Separation of tocopherol from the other distilled compounds is possible by several methods: (1) by esterification with a lower alcohol, washing and vacuum distillation; (2) by saponification, or (3) by fractional liquid–liquid extraction. The concentrates obtained in this way may be purified further by molecular distillation, extraction, crystallisation, or combinations of these procedures.
The tocopherol concentrates recommended as antioxidants are mixtures with relatively high contents of γ-tocopherol and δ-tocopherol (being obtained from soybean oil), but α-tocopherol is also present. The total tocopherol concentration usually lies between 30 and 80%. The rest is constituted of triacylglycerols.
Pharmacology
The antioxidant effects of tocopherol can be translated into different changes at the pharmacodynamic level. In vitro studies have shown that this antioxidant activity can produce modification in protein kinase C (PKC) which will later be translated into an inhibition of cell death. Some other derivate effects are the anti-inflammatory properties of tocopherol which can be related to the modulation of cytokines or prostaglandins, prostanoids and thromboxanes.
Toxicology
Tocopherol is considered as non-toxic but if very high doses are administered, there are reports of hemorrhagic activity. Reproductive and developmental toxicity tests are negative. These negative results were also observed in the analysis of mutagenicity and carcinogenicity.
Properties of Tocopherol
| Melting point: | 292 °C |
| storage temp. | 0-6°C |
| Odor | Typical vegetable oil |
| CAS DataBase Reference | 1406-18-4(CAS DataBase Reference) |
| NIST Chemistry Reference | «alpha»-Tocopherol(1406-18-4) |
| EPA Substance Registry System | Vitamin E (1406-18-4) |
Safety information for Tocopherol
Computed Descriptors for Tocopherol
New Products
4-Fluorophenylacetic acid 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate (6-METHYL-[1,3]DITHIOLO[4,5-b]QUINOXALIN-2-ONE INDAZOLE-3-CARBOXYLIC ACID 4-IODO BENZOIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5,6-Dimethoxyindanone 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran





You may like
-
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1