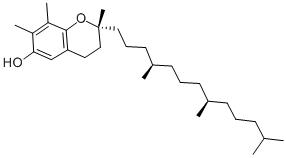
DL-α-Tocopherol
Synonym(s):Vitamin E;all-rac-α-Tocopherol;(±)-α-Tocopherol;TGF-β3;Vitamin E alcohol
- CAS NO.:10191-41-0
- Empirical Formula: C29H50O2
- Molecular Weight: 430.71
- MDL number: MFCD00072051
- EINECS: 233-466-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-18 14:26:18
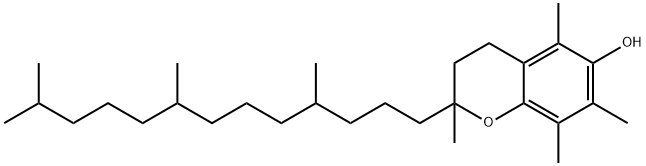
What is DL-α-Tocopherol?
Chemical properties
1mg = 1.1 IU
Chemical properties
Alpha tocopherol is a natural product. The PhEur 6.0 describes alpha-tocopherol as a clear, colorless or yellowish-brown, viscous, oily liquid.
The Uses of DL-α-Tocopherol
dl-α-Tocopherol is the racemic analog of α-Tocopherol (T526125), the most bioactive of the naturally occurring forms of Vitamin E. Richest sources are green vegetables, grains, and oils, particularly palm, safflower and sunflower oils. dl-α-Tocopherol is an antioxidant that protects cell membrane lipids from oxidative damage.
The Uses of DL-α-Tocopherol
DL-α-Tocopherol is used as an antioxidant that protects cell membrane lipids from oxidative damage.
The Uses of DL-α-Tocopherol
These Secondary Standards are qualified as Certified Reference Materials. These are suitable for use in several analytical applications including but not limited to pharma release testing, pharma method development for qualitative and quantitative analyses, food and beverage quality control testing, and other calibration requirements.
What are the applications of Application
DL-α-Tocopherol is an antioxidant that protects cell membrane lipids from oxidative damage
Production Methods
Naturally occurring tocopherols are obtained by the extraction or molecular distillation of steam distillates of vegetable oils; for example, alpha tocopherol occurs in concentrations of 0.1–0.3% in corn, rapeseed, soybean, sunflower, and wheat germ oils.Beta and gamma tocopherol are usually found in natural sources along with alpha tocopherol. Racemic synthetic tocopherols may be prepared by the condensation of the appropriate methylated hydroquinone with racemic isophytol.
Definition
ChEBI: 2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridecyl)-3,4-dihydro-2H-1-benzopyran-6-ol is a tocopherol.
General Description
TGF-β3 (transforming growth factor-β3) belongs to the TGF β superfamily. The TGFβ3 gene is mapped to human chromosome 14q24.3
Flammability and Explosibility
Non flammable
Pharmaceutical Applications
Alpha tocopherol is primarily recognized as a source of vitamin E,
and the commercially available materials and specifications reflect
this purpose. While alpha tocopherol also exhibits antioxidant
properties, the beta, delta, and gamma tocopherols are considered
to be more effective as antioxidants.
Alpha-tocopherol is a highly lipophilic compound, and is an
excellent solvent for many poorly soluble drugs.Of widespread
regulatory acceptability, tocopherols are of value in oil- or fat-based
pharmaceutical products and are normally used in the concentration
range 0.001–0.05% v/v. There is frequently an optimum
concentration; thus the autoxidation of linoleic acid and methyl
linolenate is reduced at low concentrations of alpha tocopherol, and
is accelerated by higher concentrations. Antioxidant effectiveness
can be increased by the addition of oil-soluble synergists such as
lecithin and ascorbyl palmitate.
Alpha tocopherol may be used as an efficient plasticizer. It has
been used in the development of deformable liposomes as topical
formulations.
d-Alpha-tocopherol has also been used as a non-ionic surfactant
in oral and injectable formulations.
Biochem/physiol Actions
TGF-β3 (transforming growth factor-β3) regulates lymphocyte proliferation, apoptosis, hematopoiesis and embryogenesis to maintain immune homeostasis. TGF-β plays an important role in cell growth, differentiation, and survival. TGF-β3 specifically promotes chondrogenic differentiation.TGF-β3 is a strong growth inhibitor for normal and transformed epithelial, lymphoid, fibroblast, and keratinocyte cells. TGF-2 inhibits antitumor action of NK (natural killer) cells, T-cells, macrophages, monocytes and neutrophils. TGF-β3 is a tumor suppressor in the early stages of carcinogenesis, but in the later stages acts as a tumor promoter by inducing epithelial-mesenchymal transition and stimulating angiogenesis. TGF-β isoforms is known to participate in wound healing and tissue fibrosis. TGF-β3 is crucial for tissue restoration and scarless tissue repair. Mutation in the TGFβ3 gene is associated with development of non-syndromic cleft palate only (NS CPO), a rare congenital disease.
Safety
Tocopherols (vitamin E) occur in many food substances that are
consumed as part of the normal diet. The daily nutritional
requirement has not been clearly defined but is estimated to be
3.0–20.0 mg. Absorption from the gastrointestinal tract is dependent
upon normal pancreatic function and the presence of bile.
Tocopherols are widely distributed throughout the body, with some
ingested tocopherol metabolized in the liver; excretion of metabolites
is via the urine or bile. Individuals with vitamin E deficiency are
usually treated by oral administration of tocopherols, although
intramuscular and intravenous administration may sometimes be
used.
Tocopherols are well tolerated, although excessive oral intake
may cause headache, fatigue, weakness, digestive disturbance, and
nausea. Prolonged and intensive skin contact may lead to erythema
and contact dermatitis.
The use of tocopherols as antioxidants in pharmaceuticals and
food products is unlikely to pose any hazard to human health since
the daily intake from such uses is small compared with the intake of
naturally occurring tocopherols in the diet.
The WHO has set an acceptable daily intake of tocopherol used
as an antioxidant at 0.15–2.0 mg/kg body-weight.
Metabolism
Not Available
Storage
Tocopherols are oxidized slowly by atmospheric oxygen and
rapidly by ferric and silver salts. Oxidation products include
tocopheroxide, tocopherylquinone, and tocopherylhydroquinone,
as well as dimers and trimers. Tocopherol esters are more stable to
oxidation than the free tocopherols but are in consequence less
effective antioxidants.
Tocopherols should be stored under an inert gas, in an airtight
container in a cool, dry place and protected from light.
Purification Methods
Dissolve dl--tocopherol in anhydrous MeOH (15mL/g) cool to -6o for 1hour, then chill in a Dry-ice/acetone bath; crystallisation is induced by scratching with a glass rod. The dl--acetate [52225-20-4] (see DL-vitamin E actetate below) is a viscous yellow liquid with m -7o, b 184o/0.01mm, 224o/0.3mm, d 4 20 0.953, n D 20 1.496. It is used as a standard for Vitamin E activity where the unit of activity is attained with 1mg of pure dl--acetate. [Friedrich “Vitamins” Water de Guyter Publ, Berlin 1988, Beilstein 17/4 V 168.]
Incompatibilities
Tocopherols are incompatible with peroxides and metal ions, especially iron, copper, and silver. Tocopherols may be absorbed into plastic.
Regulatory Status
GRAS listed. Accepted in Europe as a food additive. Included in the FDA Inactive Ingredients Database (IV injections, powder, lyophilized powder for liposomal suspension; oral capsules, tablets, and topical preparations). Included in the Canadian List of Acceptable Non-medicinal Ingredients. Included in nonparenteral medicines licensed in the UK.
Properties of DL-α-Tocopherol
| Melting point: | 2-4°C |
| Boiling point: | 200-220°C 0,1mm |
| alpha | [α]D20 - 0.02 - +0.02゜ (neat) |
| Density | 0.950 g/mL at 20 °C(lit.) |
| refractive index | n |
| Flash point: | 240°C |
| storage temp. | 2-8°C |
| solubility | H2O: insoluble |
| form | Pale yellow oil |
| pka | 11.40±0.40(Predicted) |
| color | Colourless to Dark Yellow |
| Odor | at 100.00?%. bland |
| Water Solubility | Miscible with chloroform, vegetable oils, ether, acetone and alcohol. Immiscible with water. |
| Sensitive | Light Sensitive |
| Merck | 14,9495 |
| BRN | 94012 |
| CAS DataBase Reference | 10191-41-0(CAS DataBase Reference) |
| EPA Substance Registry System | 2H-1-Benzopyran-6-ol, 3,4-dihydro-2,5,7,8-tetramethyl-2-(4,8,12-trimethyltridecyl)- (10191-41-0) |
Safety information for DL-α-Tocopherol
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H317:Sensitisation, Skin |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P272:Contaminated work clothing should not be allowed out of the workplace. P280:Wear protective gloves/protective clothing/eye protection/face protection. P333+P313:IF SKIN irritation or rash occurs: Get medical advice/attention. P501:Dispose of contents/container to..… |
Computed Descriptors for DL-α-Tocopherol
| InChIKey | GVJHHUAWPYXKBD-IEOSBIPESA-N |
DL-α-Tocopherol manufacturer
Nandlal Bankatlal Private Limited
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran
You may like
-
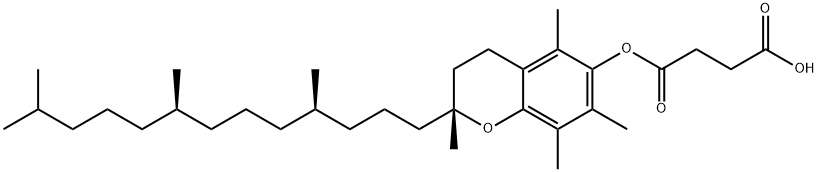
 DL-α-Tocopherol CAS 10191-41-0View Details
DL-α-Tocopherol CAS 10191-41-0View Details
10191-41-0 -
 α-Tocopherol, ≥95.5% CAS 10191-41-0View Details
α-Tocopherol, ≥95.5% CAS 10191-41-0View Details
10191-41-0 -
 all-rac-α-Tocopherol CAS 10191-41-0View Details
all-rac-α-Tocopherol CAS 10191-41-0View Details
10191-41-0 -
 Alpha Tocopherol CAS 10191-41-0View Details
Alpha Tocopherol CAS 10191-41-0View Details
10191-41-0 -
 Alpha Tocopherol CAS 10191-41-0View Details
Alpha Tocopherol CAS 10191-41-0View Details
10191-41-0 -
 Transforming Growth Factor-β3 human CASView Details
Transforming Growth Factor-β3 human CASView Details -
 84View Details
84View Details
10191-41-0 -
 liquid Dl - Alpha -Tocopherol, Grade Standard: Medicine Grade, Packaging Type: CarboyView Details
liquid Dl - Alpha -Tocopherol, Grade Standard: Medicine Grade, Packaging Type: CarboyView Details
10191-41-0
