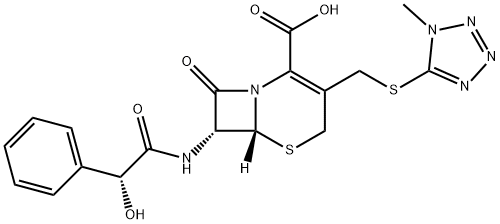
Sodium cefamandole
- CAS NO.:30034-03-8
- Empirical Formula: C18H17N6NaO5S2
- Molecular Weight: 484.48
- MDL number: MFCD07793339
- EINECS: 250-009-0
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-06-08 09:02:24
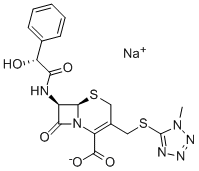
What is Sodium cefamandole?
Description
Cefamandole is a cephalosporin antibiotic that is effective against E. coli (MIC values range from 0.25-2 mg/L depending on strain) as well as H. influenza, S. pneumoniae, and S. aureus. It has been used to study the expression and inhibition of penicillin-binding proteins on bacterial cell walls and to study antibiotic resistance.
Chemical properties
White Solid
The Uses of Sodium cefamandole
Broad-spectrum semi-synthetic cephalosporin antibiotic.
The Uses of Sodium cefamandole
Antibacterial;Bacterial transpeptidase inhibitor
What are the applications of Application
Cefamandole sodium salt is a broad-spectrum semi-synthetic cephalosporin antibiotic
Definition
ChEBI: An organic sodium salt that is the sodium salt of cefamandole.
in vitro
the in-vitro effect of cefamandole was tested against 645 strains of bacteria isolated from clinical sources. against gram-positive organisms cefamandole showed great potency, being three- to four-fold more active than cephalexin or cefoxitin. none of the pseudomonas aeruginosa strains were susceptible to 100 μg of cefamandole per ml [1].
in vivo
the testicular toxicity of cefamandole was evaluated in neonatal rats. results showed that cefamandole caused delayed maturity of the germinal epithelium of neonatal rats. in rats given daily subcutaneous injections during this period, the most mature germinal cells were acrosome phase spermatids [2].
References
[1] eickhoff tc, ehret jm. in vitro comparison of cefoxitin, cefamandole, cephalexin, and cephalothin. antimicrob agents chemother. 1976 jun;9(6):994-9.
[2] hoover dm, buening mk, tamura rn, steinberger e. effects of cefamandole on spermatogenic development of young cd rats. fundam appl toxicol. 1989 nov;13(4):737-46.
[3] delgado dg, brau cj, cobbs cg, dismukes we. clinical and laboratory evaluation of cefamandole in the therapy of haemophilus spp. bronchopulmonary infections. antimicrob agents chemother. 1979 jun;15(6):807-12.
Properties of Sodium cefamandole
| Melting point: | >175°C (dec.) |
| storage temp. | Store at RT. |
| solubility | DMSO (Slightly), Methanol (Slightly), Water (Slightly) |
| form | Solid |
| color | White to Pale Yellow |
| Stability: | Moisture Sensitive |
| CAS DataBase Reference | 30034-03-8(CAS DataBase Reference) |
Safety information for Sodium cefamandole
Computed Descriptors for Sodium cefamandole
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran

You may like
-
 3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details
3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details -
 1-methylindoline-2,3-dione 98%View Details
1-methylindoline-2,3-dione 98%View Details
2058-74-4 -
 614-19-7 98%View Details
614-19-7 98%View Details
614-19-7 -
 3112-85-4 Methyl phenyl sulfone 98%View Details
3112-85-4 Methyl phenyl sulfone 98%View Details
3112-85-4 -
 20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 -
 3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details
3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details -
 57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 -
 4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
5305-40-8
