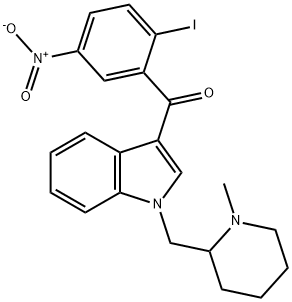
(R,S)-AM1241
Synonym(s):(R,S)-3-(2-Iodo-5-nitrobenzoyl)-1-(1-methyl-2-piperidinylmethyl)-1H-indole
- CAS NO.:444912-48-5
- Empirical Formula: C22H22IN3O3
- Molecular Weight: 503.33
- MDL number: MFCD09970778
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-11-19 23:02:33
What is (R,S)-AM1241?
Description
AM1241 is a cannabinoid (CB) receptor agonist that is selective for CB2 over CB1 with Ki values of 7.1 and 580 nM for human recombinant receptors transfected into HEK and CHO cells, respectively, in a radioligand binding assay. It is considered a protean agonist as it has neutral antagonist and partial agonist activity, depending on the assay utilized. It is also acts in a species-dependent manner in vitro, acting as an agonist at human CB2 receptors (EC50 = 190 nM) but an inverse agonist at rat and mouse CB2 receptors (EC50s = 216 and 463 nM, respectively). AM1241 produces antinociception to thermal stimuli in rat hindpaw. The antinociceptive actions of AM1241 were blocked by the CB2 receptor-selective antagonist AM630 but not by the CB1 receptor-selective antagonist AM251 . AM1241 is neuroprotective, preventing HIV-1 glycoprotein Gp120-induced apoptosis in primary human and murine neural progenitor cells and increasing cell survival and differentiation. It increases hippocampal neurogenesis and decreases astro- and gliogenesis in GFAP/Gp120 transgenic mice when administered at a dose of 10 mg/kg daily for ten days. AM1241 also delays motor impairment in a murine model of amytrophic lateral sclerosis (ALS).
The Uses of (R,S)-AM1241
(2-Iodo-5-nitrophenyl)[1-[(1-methyl-2-piperidinyl)methyl]-1H-indol-3-yl]methanone is a compound from the aminkalkylindole family which exerts potent and selective agonist activity for the cannabinoid receptor CB2.
The Uses of (R,S)-AM1241
(R,S)-AM1241 has been used as a cannabinoid CB2 agonist:
- to study its inhibitory effect on bone cancer-induced pain and bone loss
- to study the effect of its interaction with 17βestradiol on proliferation activity in primary human osteoblasts
- to evaluate the sites of CB2 mediated antinociception in vivo.
Biochem/physiol Actions
AM1241 acts as an antinociceptive agent in several animal pain models. It has a potential to delay disease progression in amyotrophic lateral sclerosis (ALS) mouse model. Intrathecal, intravenous or intraperitoneal administration of AM1241 reduces hyperalgesia and allodynia in neuropathic rats.
Properties of (R,S)-AM1241
| Boiling point: | 630.7±55.0 °C(Predicted) |
| Density | 1.60±0.1 g/cm3(Predicted) |
| storage temp. | Store at -20°C |
| solubility | DMSO: ~18mg/mL at 60°C |
| pka | 9.73±0.10(Predicted) |
| form | solid |
| color | yellow |
Safety information for (R,S)-AM1241
| Signal word | Danger |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07  Health Hazard GHS08 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H334:Sensitisation, respiratory H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for (R,S)-AM1241
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium THOMAIND PAPER PH 2.0 TO 4.5 1 BOX BUFFER CAPSULE PH 9.2 - 10 CAP SODIUM CHLORIDE 0.1N CVS ALLOXAN MONOHYDRATE 98% PLATINUM 0.5% ON 3 MM ALUMINA PELLETS (TYPE 73) LITHIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran
You may like
-
 AM-1241 98.00% CAS 444912-48-5View Details
AM-1241 98.00% CAS 444912-48-5View Details
444912-48-5 -
 (R,S)-AM1241 CAS 444912-48-5View Details
(R,S)-AM1241 CAS 444912-48-5View Details
444912-48-5 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
1805639-70-6 -
 1784294-80-9 98%View Details
1784294-80-9 98%View Details
1784294-80-9 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4