Rimonabant
- CAS NO.:168273-06-1
- Empirical Formula: C22H21Cl3N4O
- Molecular Weight: 463.79
- MDL number: MFCD04034714
- EINECS: 200-223-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-11-19 23:02:33
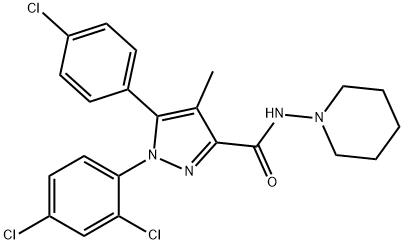
What is Rimonabant?
Absorption
Undetermined
Toxicity
Almost twice as many people discontinued rimonabant compared with placebo because of adverse events (13.8% vs. 7.2%). These consistently involved psychiatric disorders (8.5% vs. 3.2%), including depression and anxiety. Other common side effects included insomnia, nausea, vomiting, diarrhoea and fatigue.
Description
Rimonabant is a first-in-class drug launched as an oral treatment for obesity, and its mechanism of action involves the selective antagonism of cannabinoid type 1 (CB1) receptor. It is specifically indicated as an adjunct to diet and exercise for the treatment of obese patients (body mass index [BMI]≥30 kg/m2), or overweight patients (BMI>27 kg/m2) with associated risk factors such as type 2 diabetes or dyslipidemia. Additionally, rimonabant is currently under development as a treatment for nicotine dependence. The CB1 and CB2 receptors, along with their endogenous ligands, constitute the endocannabinoid system. The CB1 receptor is expressed in the brain, adipose tissue, and several peripheral organs; the CB2 receptor is predominantly expressed in immune cells. Activation of the CB1 receptor in the CNS is associated with appetite stimulation and the modulation of brain reward mechanism, whereas activation in the periphery favors metabolic processes that lead to hepatic lipogenesis and impaired glucose homeostasis. Rimonabant acts by selectively blocking the action of central and peripheral CB1 receptors, thereby reducing food intake and improving lipid and glucose metabolism.
Originator
Sanofi-Synthelabo (France)
The Uses of Rimonabant
Rimonabant is a selective antagonist of CB1 with IC50 of 13.6 nM and EC50 of 17.3 nM in hCB1 transfected HEK 293 membrane
What are the applications of Application
Rimonabant is an immunomodulatory CB1 receptor inverse agonist
Background
Rimonabant is an anorectic anti-obesity drug produced and marketed by Sanofi-Aventis. It is an inverse agonist for the cannabinoid receptor CB1. Its main avenue of effect is reduction in appetite. Rimonabant is the first selective CB1 receptor blocker to be approved for use anywhere in the world. Rimonabant is approved in 38 countries including the E.U., Mexico, and Brazil. It was rejected for approval for use in the United States. This decision was made after a U.S. advisory panel recommended the medicine not be approved because it may increase suicidal thinking and depression.
Indications
For use in conjunction with diet and exercise for patients with a body mass index greater than 30 kg/m2, or patients wih a BMI greater than 27 kg/m2 with associated risk factors, such as type 2 diabetes or dyslipidaemia.
Definition
ChEBI: Rimonabant is a carbohydrazide obtained by formal condensation of the carboxy group of 5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl-1H-pyrazole-3-carboxylic acid with the amino group of 1-aminopiperidine. It is a potent and selective cannabinoid receptor 1 (CB1R) antagonist. Besides its antagonistic properties, numerous studies have shown that, at micromolar concentrations rimonabant behaves as an inverse agonist at CB1 receptors. The drug was the first selective CB1 antagonist/inverse agonist introduced into clinical practice to treat obesity and metabolic-related disorders. It was later withdrawn from market due to CNS-related adverse effects including depression and suicidal ideation. It has a role as an anti-obesity agent, a CB1 receptor antagonist and an appetite depressant. It is a member of pyrazoles, a dichlorobenzene, a carbohydrazide, an amidopiperidine and a member of monochlorobenzenes.
brand name
Acomplia (Sanofi-Synthe-labo).
Pharmacokinetics
In the RIO-North America trial, 3040 patients were randomized to receive either placebo or one of two doses of rimonabant (5 mg or 20 mg per day). Patients taking 20 mg rimonabant had significant weigh loss, decrease in waist circumference, improved insulin sensitivity, and increases in HDL cholesterol, compared to patients on placebo.
Synthesis
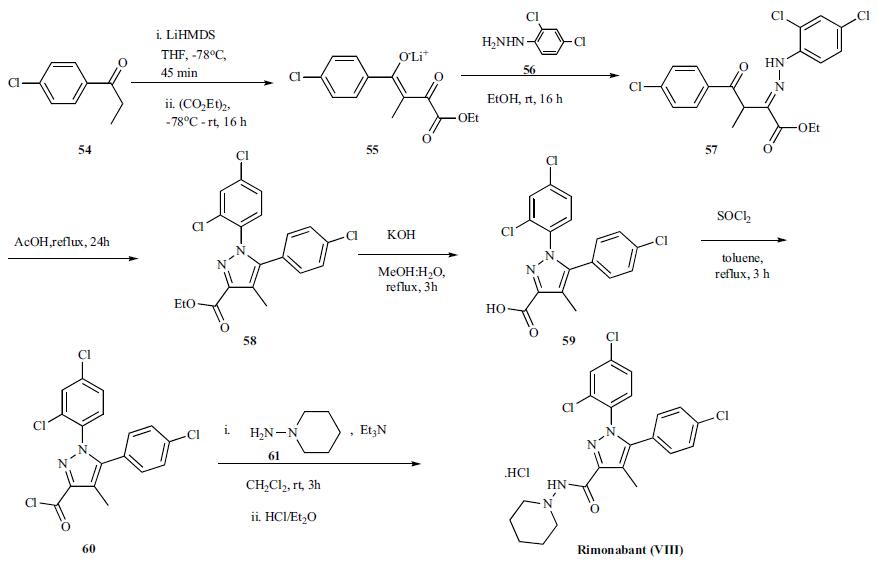
The reported preparation of rimonabant, both in small and large scale, is shown in the scheme. Lithium enolate formation of p-chlorophenyl ethyl ketone 54 with LiHMDS in THF at -78oC for 45 min followed by reaction with diethyl oxalate at -78oC and warming to room temperature over 16 h provided the lithium enolate salt of the diketoester 55. Reaction of diketoester salt 55 with 2,4-dichlorophenyl hydrazine (56) in ethanol at room temperature gave intermediate hydrazone 57 which is then cyclized in refluxing acetic acid for 24 h to obtain pyrazole ester 58. Hydrolysis of ester 58 with KOH in refluxing methanol:water mixture gave acid 59 which was then converted to the acid chloride 60 with thionyl chloride in refluxing toluene in very good yield. On scale, the synthesis of the acid chloride was performed in cyclohexane at 83oC. Reaction of acid chloride 60 with 1-aminopiperidine (61) in the presence of triethylamine at 0oC to room temperature over 3h gave rimonabant (VIII) which was isolated as the HCl salt by treating it with HCl in ether.
Metabolism
Hepatic, CYP3A4 involved.
References
1) Rinaldi-Carmona et al. (1994) SR141716A, a potent and selective antagonist of the brain cannabinoid receptor; FEBS Lett. 350 240 2) Rinaldi-Carmona et al. (1995) Biochemical and pharmacological characterization of SR141716A, the first potent and selective cannabinoid receptor antagonist; Life Sci. 56 1941
Properties of Rimonabant
| Melting point: | 154.7 °C |
| Density | 1.41±0.1 g/cm3(Predicted) |
| storage temp. | RT |
| solubility | Soluble in DMSO (up to 20 mg/ml) or in Ethanol (up to 20 mg/ml). |
| pka | 11.31±0.20(Predicted) |
| form | solid |
| color | White |
| Stability: | Stable for 1 year from date of purchase as supplied. Protect from moisture. Solutions in DMSO or ethanol may be stored at -20°C for up to 3 months. |
| CAS DataBase Reference | 168273-06-1(CAS DataBase Reference) |
Safety information for Rimonabant
Computed Descriptors for Rimonabant
Rimonabant manufacturer
Alfa Omega Pharma
New Products
Tert-butyl bis(2-chloroethyl)carbamate 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid DIETHYL AMINOMALONATE HYDROCHLORIDE 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) N-METHYL INDAZOLE-3-CARBOXYLIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran



You may like
-
 168273-06-1 Rimonabant 98%View Details
168273-06-1 Rimonabant 98%View Details
168273-06-1 -
 Rimonabant 95% CAS 168273-06-1View Details
Rimonabant 95% CAS 168273-06-1View Details
168273-06-1 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1 -
 733039-20-8 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 98+View Details
733039-20-8 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 98+View Details
733039-20-8