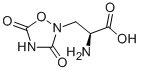
QUISQUALIC ACID
Synonym(s):β-(3,5-Dioxo-1,2,4-oxadiazolidin-2-yl)-L -alanine;3-(3,5-Dioxo-1,2,4-oxadiazolidin-2-yl)-L -alanine
- CAS NO.:52809-07-1
- Empirical Formula: C5H7N3O5
- Molecular Weight: 189.13
- MDL number: MFCD00069337
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-06-30 15:45:59
What is QUISQUALIC ACID?
Description
L-Quisqualic acid is a natural analog of glutamate that acts as an agonist at AMPA-selective and metabotropic glutamate receptors (EC50s = 170, 10, 40, and 29 nM at GluR-A, mGluR1, mGluR3, and mGluR5, respectively), as well as the ionotropic kainate receptor (GRIK4; Ki = 6.43 nM). L-Quisqualic acid is used to study receptor dynamics and as an excitotoxin to selectively destroy neurons in the brain or spinal cord.
Chemical properties
Off-White Solid
The Uses of QUISQUALIC ACID
Quisqualic acid is used to differentiate between mGluR-1 and mGluR-4. L-Quisqualic acid is an activator of GluR.
The Uses of QUISQUALIC ACID
An amino acid that is able to function as an agonist at multiple excitatory amino acid receptor substrates in the central nervous system. It also has high affinity for the kainate, AMPA and the metabotropic receptors. Inhibits the Ca2+/Cl-dependent glutamic acid uptake system in brain synaptic plasma membrane preparations.
The Uses of QUISQUALIC ACID
Quisqualic acid has been used to test ligand-gated receptors in spiral ganglion neurons.
What are the applications of Application
L-Quisqualic acid is an agonist for group I mGluR
Definition
ChEBI: Quisqualic acid is a non-proteinogenic alpha-amino acid.
General Description
Quisqualate/ Quisqualic acid is obtained from the fruits and seeds of Quisqualis chinensis. It is an agonist at two subsets of excitatory amino acid receptors metabotropic receptors that indirectly mediate calcium mobilization from intracellular stores and, ionotropic receptors that directly control membrane channels. L-quisqualic acid is an agonist of the neurotransmitter L-glutamate.
Biological Activity
Glutamate receptor agonist acting at AMPA receptors and metabotropic glutamate receptors positively linked to phosphoinositide hydrolysis. Sensitizes neurons in hippocampus to depolarization by L-AP6 (the so called 'quis' effect). Also available as part of the Group I mGlu Receptor Tocriset™ .
Biochem/physiol Actions
Active enantiomer of quisqualic acid; excitatory amino acid at glutamate receptors.
storage
Store at RT
Purification Methods
It has been purified by ion-exchange chromatography on Dowex 50W (x 8, H+ form); the desired fractions are lyophilised and recrystallised from H2O/EtOH. It has IR (KBr) : 3400-2750br, 1830s, 1775s, 1745s and 1605s cm-1;maxand 1H NMR (NaOD/D2O, pH 13) : 3.55-3.57 (1H m, X of ABX, H-2), 3.72-3.85 (2H, AB of ABX, H-3), 1 3C NMR (D2O) : 50.1t, 53.4d, 154.8s, 159.7s and 171.3s. [Baldwin et al. J Chem Soc, Chem Commun 256 1985.] It is a quasiqualate receptor agonist [Joels et al. Proc Natl Acad Sci USA 86 3404 1989].
Properties of QUISQUALIC ACID
| Melting point: | 185-187°C dec. |
| Boiling point: | 324.36°C (rough estimate) |
| alpha | D20 +17.0° (c = 2.0 in 6M HCl) |
| Density | 1.6649 (rough estimate) |
| refractive index | 1.6190 (estimate) |
| storage temp. | 2-8°C |
| solubility | NH4OH 1 M: 20 mg/mL, clear, colorless |
| form | powder |
| pka | 2.12±0.10(Predicted) |
| color | white to off-white |
| Water Solubility | Soluble in ethanol, water. Insoluble in organic solvents. |
| Merck | 13,8177 |
| BRN | 1078734 |
| Stability: | Stable. Incompatible with strong oxidizing agents. |
Safety information for QUISQUALIC ACID
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H312:Acute toxicity,dermal H332:Acute toxicity,inhalation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P321:Specific treatment (see … on this label). P304+P340:IF INHALED: Remove victim to fresh air and Keep at rest in a position comfortable for breathing. |
Computed Descriptors for QUISQUALIC ACID
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium THOMAIND PAPER PH 2.0 TO 4.5 1 BOX BUFFER CAPSULE PH 9.2 - 10 CAP SODIUM CHLORIDE 0.1N CVS ALLOXAN MONOHYDRATE 98% PLATINUM 0.5% ON 3 MM ALUMINA PELLETS (TYPE 73) LITHIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran

You may like
-
 Quisqualic acid CAS 52809-07-1View Details
Quisqualic acid CAS 52809-07-1View Details
52809-07-1 -
 1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
99903-60-3 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
1805639-70-6 -
 1784294-80-9 98%View Details
1784294-80-9 98%View Details
1784294-80-9 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4