Potassium superoxide
Synonym(s):Potassium peroxide;Potassium superoxide
- CAS NO.:12030-88-5
- Empirical Formula: KO2*
- Molecular Weight: 71.1
- MDL number: MFCD00082411
- EINECS: 234-746-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-01-27 09:38:02
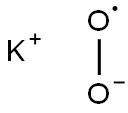
What is Potassium superoxide ?
Chemical properties
light yellow powder or chunks
The Uses of Potassium superoxide
Reagent and intermediate.
One use of potassium superoxide,KO2, is for generating oxygen. It has the ability to absorb carbon dioxide, while giving out oxygen at the same time:
4KO2(s)+ 2CO2(g)--->2K2CO3(s)+ 3O2(g)
This property has been made use of in breathing equipment,e.g.for mountaineers, in submarines and in spacecraft.
The Uses of Potassium superoxide
Potassium oxide is used as a carbon dioxide scrubber, water dehumidifier and oxygen generator. It finds application in rebreathers for fighting with fire and mine rescue work. It is also used in spacecraft, submarines and spacesuit life support systems.
General Description
A yellowish to white solid. Melting point 948°F. Mixtures with combustible material readily ignite by friction, heat, or contact with moisture. Prolonged exposure to fire or heat may cause vigorous decomposition of the material and rupturing of the container.
Air & Water Reactions
Reacts explosively with water [Mellor 2, Supp. 3: 1631. 1963].
Reactivity Profile
Potassium superoxide is a powerful oxidizer. Forms on the surface of potassium metal, solid or molten, that is exposed to the air. Attempts to extinguish burning potassium with powdered graphite has resulted in violent explosions [Chem. Abstr. 63:424. 1965]. Highly oxidized potassium metal was dropped into a dish of ethyl alcohol, an immediate explosion shattered the dish. Potassium superoxide was considered the cause of the reaction [Health and Safety Inf. 251. 1967]. Potassium superoxide should not be added to pure organic materials (hydrocarbons), as ignition and violent explosion may occur. Oxidation of arsenic, antimony, copper, potassium, tin, or zinc proceeds with incandescence, [Mellor, 1941, Vol. 2, 493]. Interaction between the superoxide and diselenium dichloride is violent, [Mellor, 1947, Vol. 10, 897].
Hazard
Corrosive to tissue.
Health Hazard
TOXIC; inhalation, ingestion or contact (skin, eyes) with vapors, dusts or substance may cause severe injury, burns or death. Fire may produce irritating and/or toxic gases. Toxic fumes or dust may accumulate in confined areas (basement, tanks, hopper/tank cars, etc.). Runoff from fire control or dilution water may cause pollution.
Fire Hazard
May explode from friction, heat or contamination. These substances will accelerate burning when involved in a fire. May ignite combustibles (wood, paper, oil, clothing, etc.). Some will react explosively with hydrocarbons (fuels). Containers may explode when heated. Runoff may create fire or explosion hazard.
Safety Profile
Explosive reaction when heated with carbon, 2-aminophenol + tetrahydrofuran (at 65°C). Forms a friction- sensitive explosive mixture with hydrocarbons. Violent reaction with lselenium dichloride, ethanol, potassium- sodium alloy. May ignite on contact with organic compounds. Incandescent reaction with metals (e.g., arsenic, antimony, copper, potassium, tin, and zinc). When heated to decomposition it emits toxic fumes of K2O. See also PEROXIDES.
Properties of Potassium superoxide
| Melting point: | 400 °C |
| Density | 2,14 g/cm3 |
| storage temp. | Store at +15°C to +25°C. |
| solubility | Soluble in ethanol and ether. |
| form | powder |
| color | Yellow-green |
| Specific Gravity | 2.14 |
| PH | >7 (H2O, 20℃) |
| Water Solubility | reacts |
| Sensitive | Air & Moisture Sensitive |
| Stability: | Stable, but reacts violently with water. Incompatible with moisture, alcohols, strong reducing agents, strong acids, finely powdered metals. |
| CAS DataBase Reference | 12030-88-5(CAS DataBase Reference) |
| NIST Chemistry Reference | Potassium superoxide(12030-88-5) |
| EPA Substance Registry System | Potassium superoxide (K(O2)) (12030-88-5) |
Safety information for Potassium superoxide
| Signal word | Danger |
| Pictogram(s) |
 Flame Over Circle Oxidizers GHS03  Corrosion Corrosives GHS05 |
| GHS Hazard Statements |
H271:Oxidising liquids;Oxidising solids H314:Skin corrosion/irritation |
| Precautionary Statement Codes |
P220:Keep/Store away from clothing/…/combustible materials. P280:Wear protective gloves/protective clothing/eye protection/face protection. P310:Immediately call a POISON CENTER or doctor/physician. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Potassium superoxide
New Products
Indole Methyl Resin tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Boc-His(Boc)-OH 2-CTC Resin 4-Chloro-7-tosy1-7Hpyrrolo[2,3-d]pyrimidine 5,7-Dibromo-1H-indole 2,5-dichloro-N-hydroxy-4,6-dimethylpyridine-3-carboximidamide 2,2-Dimethoxy-7-azaspiro[3.5]nonane hydrochloride 4-chloromethyl-5-methyl-1,3-dioxol-2-one (DMDO-Cl) R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDIIMIDAZOLE 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) N-METHYL INDAZOLE-3-CARBOXYLIC ACID 4-((2-hydroxyethyl)thio)benzoic acid 1-(TERT-BUTOXYCARBONYL)-2-PYRROLIDINONE Methyl 6-methylnicotinate 3-Pyridineacrylic acid tert-Butyl carbazate TETRAHYDRO-2H-PYRAN-3-OL 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile 2,4-dihydroxybenzaldehyde 1,3-Diethyl-1,3-Diphenylurea Methyl 2-methylquinoline-6-carboxylateRelated products of tetrahydrofuran

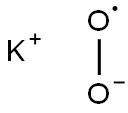

You may like
-
 Potassium dioxide CAS 12030-88-5View Details
Potassium dioxide CAS 12030-88-5View Details
12030-88-5 -
 Potassium Superoxide CAS: 12030-88-5View Details
Potassium Superoxide CAS: 12030-88-5View Details
12030-88-5 -
 Pyridine 99.5% HPLC /UV SpectroscopyView Details
Pyridine 99.5% HPLC /UV SpectroscopyView Details
110-86-1 -
 Piperazine Spot supply, best priceView Details
Piperazine Spot supply, best priceView Details
110-85-0 -
 Dibutyl PhthalateView Details
Dibutyl PhthalateView Details
84-74-2 -
 Imidazole Spot supply, competitive priceView Details
Imidazole Spot supply, competitive priceView Details
288-32-4 -
 Octadecyl 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate 98% (GC)View Details
Octadecyl 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate 98% (GC)View Details
2082-79-3 -
 Thiourea 99% ARView Details
Thiourea 99% ARView Details
62-56-6
