POLYTHIAZIDE (200 MG)
- CAS NO.:346-18-9
- Empirical Formula: C11H13ClF3N3O4S3
- Molecular Weight: 439.8818296
- MDL number: MFCD00072071
- EINECS: 2064684
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:08:52
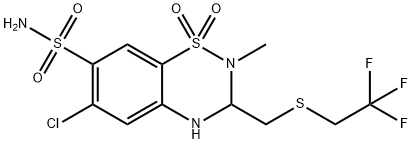
What is POLYTHIAZIDE (200 MG)?
Description
Polythiazide exhibits a more pronounced antihypertensive effect than chlorothiazide and it may be used independently for the same indications as the aforementioned drugs. However, it is primarily used as an ingredient of a combination drugs intended for lowering pressure, in particular in minizide, which is a combination of prazozine and polythiazide.
Originator
Renese ,Pfizer, US ,1961
The Uses of POLYTHIAZIDE (200 MG)
Polythiazide is a thiazide diuretic. Polythiazide is used in the treatment of hypertension as well as nephrotic syndrome, cirrhosis of the liver, and nutritional edema.
Indications
Polythiazide is a thiazide diuretic used to decrease edema and decrease blood pressure.
Background
A thiazide diuretic with actions and uses similar to those of hydrochlorothiazide. (From Martindale, The Extra Pharmacopoeia, 30th ed, p826)
Definition
ChEBI: Polythiazide is a benzothiadiazine.
Manufacturing Process
(A) Preparation of trifluoroethylthioacetaldehyde dimethylacetal: To 4.6 g (0.2 mol) of metallic sodium dissolved in 75 ml of absolute methanol is rapidly added 24.4 g (0.2 mol) of mercaptoacetaldehyde dimethylacetal followed by dropwise addition of 42.0 g (0.2 mol) of trifluoroethyl iodide.
The resulting reddish mixture is refluxed on a steam bath for one hour. One half of the alcohol is removed by concentration and the remainder diluted with several volumes of water and extracted with ether. The combined ether extracts are dried over sodium sulfate, the ether then removed at reduced pressure and the residue distilled to about 30 g (BP 82°C/25 mm).
(B) Preparation of 4-Amino-2-Chloro-5-(Methylsulfamyl)Benzenesulfonamide: The 5-substituted-2,4-disulfamyl anilines may be prepared by procedures described in the literature, for example, the general procedures in Monatsch. Chem. vol. 48, p 87 (1927), which involves the treatment of a m-substituted aniline with from 10 to 20 parts by weight of chlorosulfonic acid followed by the gradual addition of from about 90 to 170 parts by weight of sodium chloride. The resultant mixture is heated at approximately 150°C for about 2hours after which the reaction mixture is poured into water and the resultant 5substituted aniline-2,4-disulfonyl chloride is filtered and is then treated with concentrated ammonium hydroxide or suitable amine by standard procedures to obtain the corresponding disulfonamide.
(C) Preparation of 2-Methyl-3-(2,2,2-Trifluoroethyl)Thiomethyl-6-Chloro-7Sulfamyl-3,4-Dihydro-1,2,4-Benzothiadiazine-1,1-Dioxide: To 4.6 g (0.015 mol) of 4-amino-2-chloro5-(methylsulfamyl)benzenesulfonamide in 30 ml of the dimethyl ether of ethylene glycol is added 4.08 g (0.02 mol) of 2,2,2trifluoroethylmercaptoacetaldehyde dimethylacetal followed by 1 ml of ethyl acetate saturated with hydrogen chloride gas. The resulting solution is refluxed for 1.5 hours, cooled and then slowly added to cold water dropwise with stirring. The crude product is filtered, dried and recrystallized from isopropanol (3.2 g), MP 202° to 202.5°C. A second recrystallization from isopropanol raised the MP to 202° to 203°C.
brand name
Renese (Pfizer).
Therapeutic Function
Diuretic
General Description
Crystals or white powder.
Air & Water Reactions
Its rate of decomposition in solution increases with an increase in pH. . Insoluble in water.
Reactivity Profile
POLYTHIAZIDE (200 MG) is a sulfonamide derivative. With strong reducing agents will produce hydrogen sulfide gas.
Fire Hazard
Flash point data for POLYTHIAZIDE (200 MG) are not available, but POLYTHIAZIDE (200 MG) is probably combustible.
Pharmacokinetics
As a thiazide diuretic, Polythiazide inhibits the sodium-chloride symporter which decreases solute reabsorption leading to a retention of water in the urine, as water normally follows solutes. More frequent urination is due to the increased loss of water that has not been retained from the body as a result of a concomitant relationship with sodium loss from the convoluted tubule. The short-term anti-hypertensive action is based on the fact that thiazides decrease preload, decreasing blood pressure
Synthesis
Polythiazide, 1,1-dioxide 2-methyl-3-(2,2,2-trifluoroethylthiomethyl)-6- chloro-3,4-dihydro-2H-1,2,4-benzothiadiazin-7-sulfonamide (21.3.8), is also synthesized by an analogous scheme, which is by condensing 4-aminosulfonyl-5-chloro-2-methylaminosulfonylaniline (21.3.7) with 2,2,2-trifluoroethylthioacetaldehyde dimethylacetal.
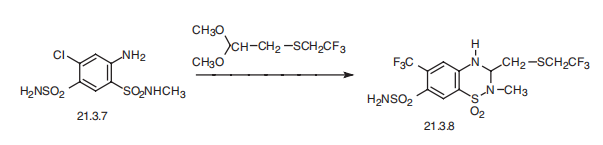
Metabolism
Not Available
Properties of POLYTHIAZIDE (200 MG)
| Melting point: | 202.5° |
| Boiling point: | 580.1±60.0 °C(Predicted) |
| Density | 1.8346 (rough estimate) |
| refractive index | 1.6100 (estimate) |
| storage temp. | Refrigerator |
| solubility | DMSO (Slightly), Methanol (Slightly, Heated) |
| pka | pKa 9.58(H2O,t =25) (Uncertain) |
| form | Solid |
| color | White to Off-White |
| EPA Substance Registry System | Polythiazide (346-18-9) |
Safety information for POLYTHIAZIDE (200 MG)
Computed Descriptors for POLYTHIAZIDE (200 MG)
New Products
4-Fluorophenylacetic acid 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate (6-METHYL-[1,3]DITHIOLO[4,5-b]QUINOXALIN-2-ONE INDAZOLE-3-CARBOXYLIC ACID 4-IODO BENZOIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5,6-Dimethoxyindanone 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran

You may like
-
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1