Iodosobenzene
- CAS NO.:536-80-1
- Empirical Formula: C6H5IO
- Molecular Weight: 220.01
- MDL number: MFCD00039507
- EINECS: 208-648-8
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-06-08 17:06:38
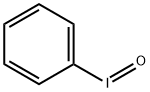
What is Iodosobenzene?
Chemical properties
Iodosobenzene is an amorphous yellow substance; it explodes at 210℃, decomposing with the evolution of iodine vapour, and dissolves in hot water and alcohol. If acids do not oxidise C6H5IO, they give saline compounds in which iodosobenzene appears as a basic oxide of a diatomic metal, C6H5I. Thus, for instance, when an acetic acid solution of iodosobenzene is treated with a solution of nitric acid, it gives large monoclinic crystals of a nitric acid salt having the composition C6H5(NO3)2 [like Ca(NO3)2). Iodosobenzene displaces iodine from potassium iodide (in a solution acidulated with acetic or hydrochloric acid)-ie, it acts with its oxygen like HClO. The action of peroxide of hydrogen, chromic acid, and other similar oxidising agents gives C6H5IO2, which is a neutral substance-i.e, is incapable of giving salts with acids.
Iodosobenzene is one of the very first oxidants and remains in use because it has excellent oxygen-transfer behavior and mechanistic cleanliness (Hill & Schardt, 1980; Rezaeifard et al., 2007; Po?towicz et al., 2006).
The Principles of Chemistry Volume 1
The Uses of Iodosobenzene
Oxygen transfer reagent for stiochiometric or catalytic cross-functionalization of alkenes, alcohols, sulfides, and organometallo Compounds.
Iodosobenzene is used as an oxidizing and acetoxylating agent in organic synthesis. It is actively involved in the preparation of (Z)-3,7-dimethyl-2,6-octadien-1-al(neral) from (Z)-3,7-dimethyl-2,6-octadien-1-ol (nerol) in presence of 2,2,6,6-tetramethylpiperidin-1-oxyl (TEMPO). It is a useful reagent for the synthesis of a wide variety of heterocyclic compounds. It is also used in the Pd-catalyzed 2-arylation of indoles.
The Uses of Iodosobenzene
lodosobenzene is a relatively new, selective oxidizing agent which is particularly useful for the preparation of sulfoxides from unsaturated or otherwise sensitive sulfides. The preparation of diallyl, di-2-hydroxyethyl and phenyl 2-chloroethyl sulfoxides illustrates its use.
Iodosobenzene diacetate behaves similarly and oxidizes diphenyl and 4-nitro-phenyl 4'-carboxyphenyl sulfide exclusively to the sulfoxides. This reagent failed, however, to oxidize bis(2-nitro-4-trifuoromethylphenyl) sulfide and in the case of bis(2-aminophenyl) sulfide it gave complex products. Iodosobenzene diacetate caused diacetoxylation of the heterocycle of 2,5-diphenyl-1 ,4-dithiadiene rather than oxidation of the sulfide function, and with 2,5-diphenylI-1 ,4-dithiadiene-1-oxide unexpected results were obtained, as discussed in section C-4.
Organic Sulfur Compounds
What are the applications of Application
Iodosobenzene is an iodinated benzene compound for proteomics research
Synthesis
Iodosobenzene has been prepared by the action of sodium or potassium hydroxide solution on iodobenzene dichloride and by addition of water to the dichloride.
Iodosobenzene is prepared from iodobenzene.It is prepared by first oxidizing iodobenzene by peracetic acid. Hydrolysis of resulting diacetate affords "PhIO":
C6H5I + CH3CO3H + CH3CO2H → C6H5I(O2CCH3)2 + H2O
C6H5I(O2CCH3)2 + H2O → C6H5IO + 2CH3CO2H
http://orgsyn.org
Properties of Iodosobenzene
| Melting point: | 210°C (rough estimate) |
| Density | 1.8665 (estimate) |
| storage temp. | Freezer |
| solubility | Methanol (Slightly), TFA (Slightly) |
| Water Solubility | Slightly soluble in water |
| form | powder to crystal |
| color | White to Yellow to Green |
| Merck | 14,5044 |
| EPA Substance Registry System | Benzene, iodosyl- (536-80-1) |
Safety information for Iodosobenzene
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H228:Flammable solids H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P240:Ground/bond container and receiving equipment. P241:Use explosion-proof electrical/ventilating/lighting/…/equipment. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P280:Wear protective gloves/protective clothing/eye protection/face protection. |
Computed Descriptors for Iodosobenzene
| InChIKey | JYJVVHFRSFVEJM-UHFFFAOYSA-N |
Abamectin manufacturer
New Products
4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium SODIUM AAS SOLUTION ZINC AAS SOLUTION BUFFER SOLUTION PH 10.0(BORATE) GOOCH CRUCIBLE SINTERED AQUANIL 5 BERYLLIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran
![1-HYDROXY-1,3-DIOXO-1,3-DIHYDRO-1L5-BENZO[D][1,2]IODOXOLE-4-CARBOXYLIC ACID](https://img.chemicalbook.in/CAS/GIF/426832-99-7.gif)
You may like
-
 536-80-1 Iodoso benzene 98%View Details
536-80-1 Iodoso benzene 98%View Details
536-80-1 -
 536-80-1 98%View Details
536-80-1 98%View Details
536-80-1 -
 Iodoso-benzene 98%View Details
Iodoso-benzene 98%View Details
536-80-1 -
 536-80-1 98%View Details
536-80-1 98%View Details
536-80-1 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4