IBOGAINE
- CAS NO.:83-74-9
- Empirical Formula: C20H26N2O
- Molecular Weight: 310.43
- MDL number: MFCD03265619
- EINECS: 201-498-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:15:30
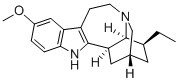
What is IBOGAINE?
Description
This indole alkaloid occurs in the root of Tabernanthe iboga Baill. It was first
examined in detail by Raymond-Hamet who assigned to it the empirical formula
C19H24(26)ON2, now altered to that given above. The base forms colourless
crystals from EtOH and is laevorotatory with [Q1D - 53° (EtOH). It is insoluble
in Et20, slightly so in Me2CO or CHC1 3 but dissolves freely in MeOH, EtOH or
H20. The alkaloid yields a hydrochloride, m.p. 299°C (dec.); lQlhs - 67°
(MeOH) or - 37.3° (H20). It contains one methoxyl group and gives the typical
indole reactions. On distillation with Zn dust or soda-lime it furnishes products
which are indole derivatives with the ~-position free.
In a manner similar to that of cocaine, the alkaloid potentiates the pressor
action of adrenaline and abolishes the sino-carotid reflexes. Unlike cocaine,
however, it also augments the action of tyramine and that of dl-ephedrine to a
slight extent. Vincent and Sero have reported that it inhibits the action of serum
cholinesterase.
Description
Ibogaine is an indole alkaloid that occurs in West African shrubs in the Apocynaceae family, including?Tabernanthe iboga, for which it is named. Iboga root has long been used in the Bwiti rituals of peoples in Gabon, Cameroon, and the Republic of the Congo.
Ibogaine is hallucinogenic and neurotoxic, but it has shown some success in reducing withdrawal effects from drugs of abuse, such as alcohol, nicotine, and methamphetamine. Some governments, fearing abuse, have banned it outright. The US Food and Drug Administration classifies it as a Schedule I controlled substance, but it has permitted research on ibogaine for combating addiction.
Definition
ChEBI: Ibogaine is an organic heteropentacyclic compound that is ibogamine in which the indole hydrogen para to the indole nitrogen has been replaced by a methoxy group. It has a role as a plant metabolite, an inhibitor, a hallucinogen and a oneirogen. It is a monoterpenoid indole alkaloid, an organic heteropentacyclic compound and an aromatic ether. It is functionally related to an ibogamine. It is a conjugate base of an ibogaine(1+).
Purification Methods
Crystallise it from EtOH or aqueous EtOH and sublime it at 150o/0.01mm. It is soluble in organic solvents but insoluble in H2O. The hydrochloride, m 299-300o(dec), is soluble in H2O and alcohols. [Büchi et al. J Am Chem Soc 88 3099 1866, Rosenmund Chem Ber 108 1871 1975, Beilstein 23 III/IV 2742.]
References
Raymond-Hamet., Bull. Soc. Chirn. Fr., 9,620 (1942)
Delourme-Houde., Chern. Abstr., 41, 1390 (1947)
Bartlett, Dickel, Taylor.,J. Arner. Chern. Soc., 80, 126 (1958)
Arai, Coppola, Jeffery., Acta Cryst., 13, 553 (1960)
Shamma, Soyster., Experientia, 20,36 (1964)
Synthesis:
Buchi et ai., f. Arner. Chern. Soc., 88,3099 (1966)
Pharmacology:
Raymond-Hamet, Rothlin., C.R. Soc. Bioi., 127,592 (1938)
Raymond-Hamet, Rothlin., Arch. inst. Pharrnacodyn., 63, 27 (1939)
Raymond-Hamet., Cornpt. rend., 201,285 (1940)
Raymond-Hamet., C.R. Soc. Bioi., 135, 176 (1941)
Raymond-Hamet, Perrot., Bull. Acad. Med., 24,423 (1941)
Vincent, Sero., C.R. Soc. Bioi., 136,612 (1942)
Properties of IBOGAINE
| Melting point: | 152-153° |
| Boiling point: | 450.59°C (rough estimate) |
| alpha | D20 -53° (in 95% ethanol) |
| Density | 1.0633 (rough estimate) |
| refractive index | 1.6800 (estimate) |
| storage temp. | 2-8°C |
| pka | 8.1 in 80% methylcellosolve |
| EPA Substance Registry System | Ibogaine (83-74-9) |
Safety information for IBOGAINE
Computed Descriptors for IBOGAINE
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid 1-Bromo-3,5-Di-Tert-Butylbenzene S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran

You may like
-
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1