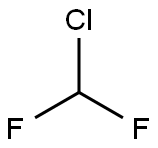
Difluoromethane
- CAS NO.:75-10-5
- Empirical Formula: CH2F2
- Molecular Weight: 52.02
- MDL number: MFCD00039283
- EINECS: 200-839-4
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-10-29 10:02:07

What is Difluoromethane?
Chemical properties
Difluoromethane is a colorless and flammable gas that has a light ethereal odor with poor warning properties.
The Uses of Difluoromethane
Difluoromethane is use as refrigerant, as an important component in some refrigeration mixtures, and as a reactive ion etching gas in semiconductor materials processing applications.
Definition
ChEBI: Difluoromethane is a member of fluoromethanes. It has a role as a refrigerant.
Preparation
Difluoromethane(HFC-32) is manufactured in closed systems. Possible routes are hydrodechlorination of chlorodifluoromethane (HCFC-22) and hydrofluorination of dichloromethane.
General Description
A colorless odorless gas. Insoluble in water and has a high thermal stability. Its vapors are heavier than air. Under prolonged exposure to fire or intense heat the containers may rupture violently and rocket. Contact with the unconfined liquid can cause frostbite. Used as a refrigerant.
Air & Water Reactions
Highly flammable. Insoluble in water. Easily ignited by heat, sparks or flames. Forms explosive mixtures with air
Reactivity Profile
Difluoromethane can react with some metals to form dangerous products. May react violently with aluminum. Incompatible with strong oxidizing and reducing agents. Also incompatible with many amines, nitrides, azo/diazo compounds, alkali metals, and epoxides.
Health Hazard
Vapors may cause dizziness or asphyxiation without warning. Some may be irritating if inhaled at high concentrations. Contact with gas or liquefied gas may cause burns, severe injury and/or frostbite. Fire may produce irritating and/or toxic gases.
Fire Hazard
EXTREMELY FLAMMABLE. Will be easily ignited by heat, sparks or flames. Will form explosive mixtures with air. Vapors from liquefied gas are initially heavier than air and spread along ground. CAUTION: Hydrogen (UN1049), Deuterium (UN1957), Hydrogen, refrigerated liquid (UN1966) and Methane (UN1971) are lighter than air and will rise. Hydrogen and Deuterium fires are difficult to detect since they burn with an invisible flame. Use an alternate method of detection (thermal camera, broom handle, etc.) Vapors may travel to source of ignition and flash back. Cylinders exposed to fire may vent and release flammable gas through pressure relief devices. Containers may explode when heated. Ruptured cylinders may rocket.
Properties of Difluoromethane
| Melting point: | −136 °C(lit.) |
| Boiling point: | −51.6 °C(lit.) |
| Density | 1.1 g/mL at 25 °C(lit.) |
| refractive index | 1.1960 |
| CAS DataBase Reference | 75-10-5(CAS DataBase Reference) |
| NIST Chemistry Reference | Methane, difluoro-(75-10-5) |
| EPA Substance Registry System | HFC-32 (75-10-5) |
Safety information for Difluoromethane
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Gas Cylinder Compressed Gases GHS04  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H220:Flammable gases H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P280:Wear protective gloves/protective clothing/eye protection/face protection. |
Computed Descriptors for Difluoromethane
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran
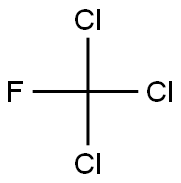



You may like
-
 VESTAR 1.0T 3 STAR INVERTER SPLIT AC R32 GAS- VASKE123IA15HView Details
VESTAR 1.0T 3 STAR INVERTER SPLIT AC R32 GAS- VASKE123IA15HView Details
75-10-5 -
 MITSUBISHI ELECTRIC 1.0T 4 STAR INVERTER SPLIT AC R32 GAS- MSY/MUY-GN13VFView Details
MITSUBISHI ELECTRIC 1.0T 4 STAR INVERTER SPLIT AC R32 GAS- MSY/MUY-GN13VFView Details
75-10-5 -
 1.1 Tr Split Mitsubishi Air Conditioner, R 32, 3 StarView Details
1.1 Tr Split Mitsubishi Air Conditioner, R 32, 3 StarView Details
75-10-5 -
 TOSHIBA 1.5T 3 STAR INVERTER SPLIT AC R32 GAS- RAS-18G3KCVG-IN / RAS-18G3ACVG-INView Details
TOSHIBA 1.5T 3 STAR INVERTER SPLIT AC R32 GAS- RAS-18G3KCVG-IN / RAS-18G3ACVG-INView Details
75-10-5 -
 1.0 Ton Blue Star 3 Star Fixed Speed Wac, R-32, WindowView Details
1.0 Ton Blue Star 3 Star Fixed Speed Wac, R-32, WindowView Details
75-10-5 -
 TOSHIBA 1.0T 3 STAR (NON INVERTER) SPLIT AC R32 GAS- RAS-13G3KG-IN/RAS-13G3AG-INView Details
TOSHIBA 1.0T 3 STAR (NON INVERTER) SPLIT AC R32 GAS- RAS-13G3KG-IN/RAS-13G3AG-INView Details
75-10-5 -
 R-32 Panasonic Inverter Split AC 1.5 TR CS/CU-YU18AKYD, 10 Years, 3 StarView Details
R-32 Panasonic Inverter Split AC 1.5 TR CS/CU-YU18AKYD, 10 Years, 3 StarView Details
75-10-5 -
 20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0
