D-AMPHETAMINE SULFATE
- CAS NO.:51-63-8
- Empirical Formula: C18H28N2O4S
- Molecular Weight: 368.49
- MDL number: MFCD00069209
- EINECS: 200-111-6
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:15:32
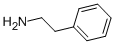
What is D-AMPHETAMINE SULFATE?
Chemical properties
white powder
Originator
Dexedrine Sulfate,SKF,US,1944
The Uses of D-AMPHETAMINE SULFATE
Amfetamine
Manufacturing Process
Two mols, for example, 270 grams, of racemic α-methylphenethylamine base
are reacted with one mol (150 grams) of d-tartaric acid, thereby forming dl-αmethylphenethylamine d-tartrate, a neutral salt. The neutral salt thus
obtained is fully dissolved by the addition of sufficient, say about 1 liter, of
absolute ethanol, and heating to about the boiling point. The solution is then
allowed to cool to room temperature with occasional stirring to effect
crystallization. The crystals are filtered off and will be found to contain a
preponderance of the levo enantiomorph.
The residual solid in the mother liquors is repeatedly and systematically
crystallized, yielding a further fraction of 1-α-methylphenethylamine d-tartrate
which may be purified by recrystallization. d-α-Methylphenethylamine may be
readily recovered from the mother liquors by the addition of tartaric acid
thereto for the formation of acid tartrates and separation of d-αmethylphenethylamine d-bitartrate by crystallization.
The free base of either optical isomer may be obtained by addition to the dtartrate in the case of the levo isomer and the d-bitartrate in the case of the
dextro isomer of alkali in excess, as, for example, by the addition of an
aqueous solution of caustic soda, which will cause the base to separate as an oil which may be recovered and purified by any well-known procedure. The
base is exactly neutralized with sulfuric acid to give the sulfate.
Therapeutic Function
Central stimulant
Biological Activity
CNS stimulant. Targets monoamine transporters to elevate synaptic levels of noradrenalin, dopamine and serotonin.
Safety Profile
Poison by ingestion, intraperitoneal, subcutaneous, and intravenous routes. A human teratogen that causes developmental abnormalities of the central nervous system. Experimental reproductive effects including other teratogenic effects. A habit-forming stimulant. When heated to decomposition it emits very toxic fumes of SO, and NO,. See also other benzidrine compounds and SULFATES.
storage
Desiccate at RT
Properties of D-AMPHETAMINE SULFATE
| Melting point: | >300° |
| alpha | D20 +21.8° (c = 2) |
| Density | 1.1500 |
| refractive index | 1.6930 (estimate) |
| storage temp. | Desiccate at RT |
| solubility | H2O: soluble |
| form | Powder |
| color | Plates |
| Water Solubility | Soluble to 100 mM in water |
| Stability: | Stable. Incompatible with strong oxidizing agents. |
| EPA Substance Registry System | Benzeneethanamine, .alpha.-methyl-, (.alpha.S)-, sulfate (2:1) (51-63-8) |
Safety information for D-AMPHETAMINE SULFATE
Computed Descriptors for D-AMPHETAMINE SULFATE
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate N-octanoyl benzotriazole 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5,6-Dimethoxyindanone 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran

You may like
-
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1