Amosulalol
- CAS NO.:85320-68-9
- Empirical Formula: C18H24N2O5S
- Molecular Weight: 380.46
- MDL number: MFCD00242746
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-11-19 20:33:22
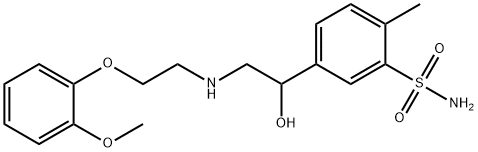
What is Amosulalol?
Description
Amosulalol is a potent antihypertensive with combined alpha and beta blocking properties. Indicated for the treatment of essential hypertension, it appears to possess a minimum of side-effects. Its beta-blocking property reduces the tachycardia associated with alpha-blockers, while its alpha-blocking activity reduces the peripheral vasoconstriction associated with beta-blockade.
Originator
Yamanouchi (Japan)
Definition
ChEBI: Amosulalol is a sulfonamide.
Manufacturing Process
A mixture of N-benzyl-2-(2-methoxyphenoxy)ethylamine, methyl ethyl ketone,
and 5-bromoacetyl-2-methylbenzenesulfonamide was refluxed with stirring.
After cooling the reaction mixture, ethyl ketone was distilled off under reduced
pressure and the residue formed was dissolved in benzene. Then, ether was
added to the solution and after removing the hydrobromide of N-benzyl-2-(2-
methoxyphenoxy)ethylamine precipitated, the solvent was distilled off under
reduced pressure to provide a viscous oily product.
The viscous oily product was dissolved in ethanol and after adding to the
solution an excess amount of sodium borohydride, the mixture was stirred at
room temperature followed by distilling off ethanol under reduced pressure.
The residue was dissolved in ethyl acetate and the ethyl acetate layer
recovered was washed with water, dried over anhydrous sodium sulfate, and
concentrated under reduced pressure to provide a viscous oily product. The
product was subjected to a silica gel column chromatography and eluted using
benzene and then a mixture of benzene and ethyl acetate of 10:1 by volume
ratio to provide 5-{1-hydroxy-2-[N-benzyl-2-(2-
methoxyphenoxy)ethylamino]ethyl}-2-methyl-benzenesulfonamide as a
viscous oily product.
In 200 ml of methanol was dissolved 20.0 g of 5-{1-hydroxy-2-[N-benzyl-2-
(2-methoxyphenoxy)ethylamino]ethyl}-2-methyl-benzenesulfonamide. After
adding thereto 20 ml of ethanol containing about 10% hydrogen chloride and
1.0 g of 10% palladium charcoal, the mixture was shaken in hydrogen gas
stream. When the absorption of hydrogen stopped, the catalyst was filtered
away and the filtrate was distilled off under reduced pressure. The residue
was dissolved in 100 ml of ethanol while it was hot and the solution was
allowed to stand overnight in ice chamber, whereby 12.8 g of the α-type
crystals of 5-{1-hydroxy-2-[2-(2-methoxyphenoxy)ethylamino]ethyl}-2-
methylbenzenesulfonamide were obtained as the colorless crystals, melting
point 169°-171°C.
In practice it is usually used as monohydrochloride
brand name
Lowgan
Therapeutic Function
Alpha-adrenergic blocker, Beta-adrenergic blocker, Antihypertensive
Properties of Amosulalol
| form | Solid |
| color | White to off-white |
Safety information for Amosulalol
Computed Descriptors for Amosulalol
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium THOMAIND PAPER PH 2.0 TO 4.5 1 BOX BUFFER CAPSULE PH 9.2 - 10 CAP SODIUM CHLORIDE 0.1N CVS ALLOXAN MONOHYDRATE 98% PLATINUM 0.5% ON 3 MM ALUMINA PELLETS (TYPE 73) LITHIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran

You may like
-
 1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
99903-60-3 -
 88491-46-7 98%View Details
88491-46-7 98%View Details
88491-46-7 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
1805639-70-6 -
 1784294-80-9 98%View Details
1784294-80-9 98%View Details
1784294-80-9 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4