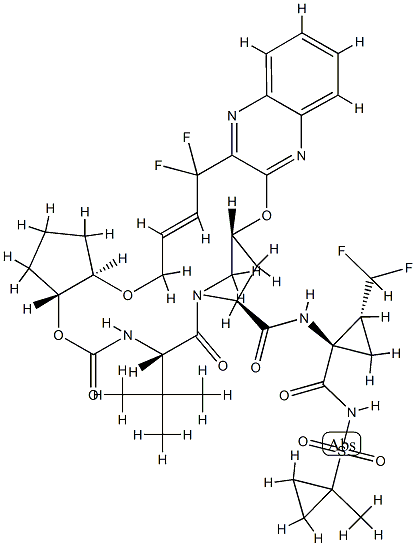
ABT-530
- CAS NO.:1353900-92-1
- Empirical Formula: C57H65F5N10O8
- Molecular Weight: 1113.18
- MDL number: MFCD30747902
- EINECS: 829-351-5
- Update Date: 2024-11-19 23:02:33
What is ABT-530?
Absorption
In healthy subjects, the time it takes to reach the peak plasma concentration (Tmax) is approximately 5 hours. The mean peak plasma concentration (Cmax) is 110ng/mL in non-cirrhotic HCV-infected subjects. Relative to fasting conditions, the consumption of meals increases the absorption of pibrentasvir by 40-53% .
Toxicity
Pibrentasvir is not shown to be genotoxic according to in vitro or in vivo studies. It also shows to have no effect on mating, female or male fertility, or early embryonic development in rodent studies. Carcinogenicity studies with pibrentasvir have not been conducted .
The Uses of ABT-530
Pibrentasvir (CAS# 1353900-92-1) is an antiviral used for hepatitis C virus NS3/4A protease inhibition.
Background
Pibrentasvir is a direct acting antiviral agent and Hepatitis C virus (HCV) NS5A inhibitor that targets the the viral RNA replication and viron assembly. In combination with Glecaprevir, pibrentastiv is a useful therapy for patients who experienced therapeutic failure from other NS5A inhibitors. In cell cultures, the emergence of amino acid substitutions at known NS5A inhibitor resistance-associated positions in HCV genotype 1a, 2a or 3a replicons led to reduced susceptibility and resistance to pibrentasvir . These resistance-associated amino acid substitutions included Q30D/deletion, Y93D/H/N or H58D +Y93H in genotype 1a replicons, F28S + M31I or P29S + K30G in genotype 2a replicons, and Y93H in genotype 3a replicons. Individual NS5A amino acid substitutions that reduced susceptibility to pibrentasvir include M28G or Q30D in a genotype 1a replicon and P32-deletion in a genotype 1b replicon .
Pibrentasvir is available as an oral combination therapy with Glecaprevir under the brand name Mavyret. This fixed-dose combination therapy was FDA-approved in August 2017 to treat adults with chronic hepatitis C virus (HCV) genotypes 1-6 without cirrhosis (liver disease) or with mild cirrhosis, including patients with moderate to severe kidney disease and those who are on dialysis . Mavyret is also indicated for HCV genotype 1-infected patients who have been previously treated with regimens either containing an NS5A inhibitor or an NS3/4A protease inhibitor, but not both . Hepatitis C viral infection often leads to decreased liver function and subsequent liver failure, causing a significantly negative impact on the patients' quality of life. The ultimate goal of the combination treatment is to achieve sustained virologic response (SVR) and cure the patients from the infection. In clinical trials, this combination therapy achieved SVR12 rate, or undetectable Hepatitis C for twelve or more weeks after the end of treatment, of ≥93% across genotypes 1a, 2a, 3a, 4, 5 and 6 .
Indications
Indicated for the treatment of adult patients with chronic hepatitis C virus (HCV) genotype 1, 2, 3, 4, 5 or 6 infection without cirrhosis or with compensated cirrhosis (Child-Pugh A). MAVYRET is also indicated for the treatment of adult patients with HCV genotype 1 infection, who previously have been treated with a regimen containing an HCV NS5A inhibitor or an NS3/4A protease inhibitor (PI), but not both .
Pharmacokinetics
Pibrentasvir is a pan-genotypic . According to HCV replicon assays, pibrentasvir has EC50 values ranging from 0.08-4.6 nM agaisnt laboratory and clinical isolates from subtypes 1a, 1b, 2a, 2b, 3a, 4a, 4d, 5a, and 6a, or EC50 values of 0.5-4.3 pM against laboratory and clinical isolates from subtypes 1a, 1b, 2a, 2b, 3a, 4a, 4b, 4d, 5a, 6a, 6e and 6p . It is active against common resistance-conferring substitutions in HCV genotypes 1 to 6 that confers resistance and decreased therapeutic response from other NS5A inhibitors, inluding positions 24, 28, 30, 31, 58, 92, or 93 in NS5A . In a QT study, pibrentasvir is not shown to prolong the QTc interval.
Metabolism
Pibrentasvir is not metabolized .
Properties of ABT-530
| Melting point: | >230°C (subl.) |
| Density | 1.363±0.06 g/cm3(Predicted) |
| storage temp. | -20°C Freezer, Under inert atmosphere |
| solubility | DMSO (Slightly), Methanol (Slightly), Water (Slightly) |
| form | Solid |
| pka | 9.76±0.10(Predicted) |
| color | White to Pale Yellow Low-Melting |
Safety information for ABT-530
Computed Descriptors for ABT-530
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium 3-Bromopyrazole (3aR,4R,5R,6aS)-hexahydro-5-Triethyl silyloxy-4-((E)-3-oxo-5-phenylpent-1- enyl)cyclopenta[b]furan-2-one. 1-Chlorocarbonyl-4-piperidinopiperidine 1-Bromo-4-phenyl-2-Butanone 4-Amino-2-fluoro-N-methylbenzamide 1,1'-Carbonyldiimidazole SODIUM AAS SOLUTION ZINC AAS SOLUTION BUFFER SOLUTION PH 10.0(BORATE) GOOCH CRUCIBLE SINTERED AQUANIL 5 BERYLLIUM AAS SOLUTIONRelated products of tetrahydrofuran
You may like
-
![Dimethyl [2-oxo-3-[3-(trifluoromethyl)phenoxy]propyl]phosphonate 99%](https://img.chemicalbook.in//Content/image/CP5.jpg) Dimethyl [2-oxo-3-[3-(trifluoromethyl)phenoxy]propyl]phosphonate 99%View Details
Dimethyl [2-oxo-3-[3-(trifluoromethyl)phenoxy]propyl]phosphonate 99%View Details
54094-19-8 -
 85-81-4 99%View Details
85-81-4 99%View Details
85-81-4 -
 Cyclopentane carboxxylic acid 3400-45-1 99%View Details
Cyclopentane carboxxylic acid 3400-45-1 99%View Details
3400-45-1 -
![208111-98-2 (3aR,4R,5R,6aS)-5-(Benzoyloxy)hexahydro-4-[(1E)-3-oxo-4-[3-(trifluoromethyl)phenoxy]-1-buten- 1-yl]-2H-cyclopenta[b]furan-2-one 99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 208111-98-2 (3aR,4R,5R,6aS)-5-(Benzoyloxy)hexahydro-4-[(1E)-3-oxo-4-[3-(trifluoromethyl)phenoxy]-1-buten- 1-yl]-2H-cyclopenta[b]furan-2-one 99%View Details
208111-98-2 (3aR,4R,5R,6aS)-5-(Benzoyloxy)hexahydro-4-[(1E)-3-oxo-4-[3-(trifluoromethyl)phenoxy]-1-buten- 1-yl]-2H-cyclopenta[b]furan-2-one 99%View Details
208111-98-2 -
 2033-24-1 99%View Details
2033-24-1 99%View Details
2033-24-1 -
 Meldrums acid 2033-24-1 99%View Details
Meldrums acid 2033-24-1 99%View Details
2033-24-1 -
 Cyaclopentane carboxylic acid 99%View Details
Cyaclopentane carboxylic acid 99%View Details
3400-45-1 -
 2-Aminopyridine 504-29-0 99%View Details
2-Aminopyridine 504-29-0 99%View Details
504-29-0