Abemaciclib
- CAS NO.:1231929-97-7
- Empirical Formula: C27H32F2N8
- Molecular Weight: 506.59
- MDL number: MFCD22665744
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-10-14 18:20:50
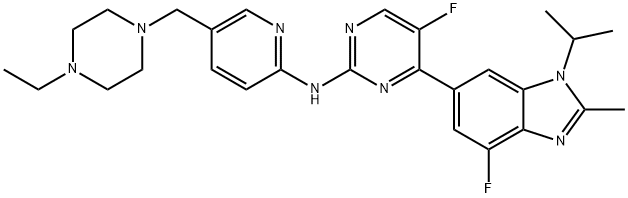
What is Abemaciclib?
Absorption
The plasma concentration of the drug increases in a dose-proportional manner. Following a single oral dose administration of 200 mg abemaciclib, the mean peak plasma concentration (Cmax) of 158 ng/mL is reached after 6 hours. The median time to reach maximum plasma concentration (Tmax) ranges from 4-6 hours following an oral administration of abemaciclib over a range of 50–275 mg , but may range up to 24 hours . The absolute bioavailability of the drug is reported to be 45% .
Toxicity
According to the bacterial reverse mutation (Ames) assay, abemaciclib and its active metbolites M2 and M20 did not display mutagenic properties. Abemaciclib was not clastogenic in vitro rat bone marrow micronucleus assay. Repeat-dose toxicity studies were performed to assess the effects of abemaciclib in testis, epididymis, prostate, and seminal vesicle at doses ≥10 mg/kg/day in rats and ≥0.3 mg/kg/day in dogs which exceed the recommeded therapeutic doses in humans. The findings included decreased organ weights, intratubular cellular debris, hypospermia, tubular distillation, atrophy and degeneration or necrosis .
Characteristics
Class: serine/threonine protein kinase
Treatment: Breast cancer
Oral bioavailability = 46%
Elimination half-life = 18 h
Protein binding = 95–98%
The Uses of Abemaciclib
Abemaciclib is a potent and selective inhibitor of cyclin-dependent kinases, CDK4 and CDK6, as a method to inhibit the proliferation of cancer cells.
Background
Abemaciclib is an antitumor agent and dual inhibitor of cyclin-dependent kinases 4 (CDK4) and 6 (CDK6) that are involved in the cell cycle and promotion of cancer cell growth in case of unregulated activity. On September 28, 2017, FDA granted approval of abemaciclib treatment under the market name Verzenio for the treatment of HR-positive and HER2-negative advanced or metastatic breast cancer that has progressed after unsuccessful endocrine therapy. It is either given alone in patients who has undergone endocrine therapy and chemotherapy after the metastasis of cancer, or in combination with Fulvestrant. Following oral treatment in patients with HR-positive, HER2-negative breast cancer, abemaciclib demonstrated increased progression-free survival rates and objective response rates. Abemaciclib has been used in trials studying the treatment of melanoma, lymphoma, neoplasm, solid tumor, and glioblastoma.
Indications
Indicated in combination with fulvestrant for the treatment of women with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer with disease progression following endocrine therapy.
Inidicated as monotherapy for the treatment of adult patients with HR-positive, HER2-negative advanced or metastatic breast cancer with disease progression following endocrine therapy and prior chemotherapy in the metastatic setting.
Reduce drug development failure ratesBuild, train, & validate machine-learning modelswith evidence-based and structured datasets.See how Build, train, & validate predictive machine-learning models with structured datasets.See how
Indications
Abemaciclib is used to treat breast cancer that is estrogen receptor-positive (ER-positive) and HER2-negative. It can be taken by both men and women. Patients may be offered abemaciclib if they have:
Early primary breast cancer that the treatment team thinks has a higher risk of coming back (recurrence). Abemaciclib may be provided as an adjuvant treatment, meaning a treatment given after initial treatment, such as surgery;
Breast cancer that has spread to the tissues and lymph nodes around the chest, neck, and under the breastbone (locally advanced breast cancer);
Breast cancer has spread to other parts of the body, such as the bones, lungs, liver, or brain (secondary breast cancer).
Biological Activity
ly2835219 is an orally available cyclin-dependent kinase (cdk) inhibitor that targets the cdk4 (cyclin d1) and cdk6 (cyclin d3) cell cycle pathway, with potential antineoplastic activity. cdk4/6 dual inhibitor ly2835219 specifically inhibits cdk4 and 6, thereby inhibiting retinoblastoma (rb) protein phosphorylation in early g1. inhibition of rb phosphorylation prevents cdk-mediated g1-s phase transition, thereby arresting the cell cycle in the g1 phase, suppressing dna synthesis and inhibiting cancer cell growth. overexpression of the serine/threonine kinases cdk4/6, as seen in certain types of cancer, causes cell cycle deregulation.
Biochem/physiol Actions
LY2835219 (abemaciclib) was identified via compound and biochemical screening by scientists at Eli Lilly and Company Research Laboratories and selected for its biological activity and highly selective inhibition of the complexes CDK4/ cyclin D1 (IC50 =2 nmol/L) and CDK6/cyclin D1 (IC50 =10 nmol/L), with no activity against other CDK/cyclin complexes or cell-cycle-related kinases within the nanomolar ranges, except for inhibition of CDK9 at IC50 at least five times higher. The compound was shown to act as a competitive inhibitor of the ATP-binding domain of the CDK4 and CDK6 and to be 14 times more potent against CDK4 than against CDK6. In comparison to palbociclib and ribociclib, abemaciclib shows higher selectivity for the complex CDK4/cyclin D1, with IC50 values five times lower than those of the two other compounds[1].
Pharmacokinetics
In combination with fulvestrant, the progression-free survival for patients with HR-positive, HER2-negative breast cancer was 16.4 months compared to 9.3 months for patients taking a placebo with fulvestrant. As a monotherapy, 19.7% of patients taking abemaciclib achieved complete or partial shrinkage of their tumors for a median 8.6 months after treatment . Abemaciclib induces cell cycle arrest and exerts an antitumor activity in human tumor xenograft models .
In patient investigations and a healthy volunteer study, abemaciclib is not shown to induce any clinically significant changes in the QTc interval .
Synthesis
A concise total synthesis of CDK 4/6 inhibitor abemaciclib is described below. The synthesis uses a Suzuki coupling and a Hartwig–Buchwald amination to join three of the four subunits. The final step is a reductive amination utilizing Leuckart–Wallach conditions. Key to the Leuckart–Wallach reaction was the addition of trimethyl orthoformate to remove water formed during the reaction, allowing the reaction to complete[4].
Enzyme inhibitor
This oral cell cycle inhibitor (FWfree-base = 506.61 g/mol; FWmesylate-salt = 602.70 g/mol; CAS 1231930-82-7 (mesylate salt) ), also known as LY2835219 and N-[5-[ (4-ethyl-1-piperazinyl) methyl]-2-pyridinyl]-5- fluoro-4-[4-fluoro-2-methyl-1- (1-methylethyl) -1H-benzimidazol-6-yl]-2- pyrimidinamine, targets the cyclin-dependent kinase CDK4, or cyclin D1 (IC50 = 2 nM) and CDK6, or cyclin D3 (IC50 = 6 nM), inhibiting retinoblastoma (Rb) protein phosphorylation in early G1, thereby arresting the cell cycle in the G1, suppressing DNA synthesis, and inhibiting cancer cell growth. LY2835219 inhibits activation of AKT and ERK, but not mTOR.
Metabolism
Abemaciclib mainly undergoes hepatic metabolism mediated by CYP3A4. The major metabolite formed is N-desethylabemaciclib (M2), while other metabolites hydroxyabemaciclib (M20), hydroxy-N-desethylabemaciclib (M18), and an oxidative metabolite (M1) are also formed. M2, M18, and M20 are equipotent to abemaciclib and their AUCs accounted for 25%, 13%, and 26% of the total circulating analytes in plasma, respectively .
References
[1] Silvia Paola Corona, Daniele Generali. “Abemaciclib: a CDK4/6 inhibitor for the treatment of HR+/HER2- advanced breast cancer.” Drug Design, Development and Therapy (2018): 321–330.
[2] “OS Trending Positive for Abemaciclib.” Cancer discovery 13 1 (2023): OF3.
[3] Erin R Scheidemann. “Resistance to abemaciclib is associated with increased metastatic potential and lysosomal protein deregulation in breast cancer cells.” Molecular Carcinogenesis (2024): 209–223.
[4] Michael O. Frederick, Douglas P. Kjell. “A synthesis of abemaciclib utilizing a Leuckart–Wallach reaction.” Tetrahedron Letters 56 7 (2015): Pages 949-951.
Properties of Abemaciclib
| Boiling point: | 689.3±65.0 °C(Predicted) |
| Density | 1.32±0.1 g/cm3(Predicted) |
| storage temp. | 4°C, protect from light |
| solubility | insoluble in H2O; ≥4.83 mg/mL in DMSO with gentle warming and ultrasonic; ≥6.34 mg/mL in EtOH with gentle warming |
| form | solid |
| pka | 7.69±0.10(Predicted) |
| color | Off-white to yellow |
Safety information for Abemaciclib
Computed Descriptors for Abemaciclib
| InChIKey | UZWDCWONPYILKI-UHFFFAOYSA-N |
| SMILES | C1(NC2=NC=C(CN3CCN(CC)CC3)C=C2)=NC=C(F)C(C2C=C3N(C(C)C)C(C)=NC3=C(F)C=2)=N1 |
Abemaciclib manufacturer
New Products
(S)-3-Aminobutanenitrile hydrochloride 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 4-IODO BENZOIC ACID 3-NITRO-2-METHYL ANILINE 1-(2,4-DICHLOROPHENYL) ETHANAMINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5,6-Dimethoxyindanone 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 1-(4-(aminomethyl)benzyl)urea hydrochloride 2-aminopropyl benzoate hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran
You may like
-
 1231929-97-7 Abemaciclib 98%View Details
1231929-97-7 Abemaciclib 98%View Details
1231929-97-7 -
 1231929-97-7 98%View Details
1231929-97-7 98%View Details
1231929-97-7 -
 Abemaciclib 99%View Details
Abemaciclib 99%View Details
1231929-97-7 -
 Abemaciclib 1231929-97-7 98%View Details
Abemaciclib 1231929-97-7 98%View Details
1231929-97-7 -
![N-{5-[(4-ethylpiperazin-1-yl)methyl]pyridin-2-yl}-5-fluoro-4-(7-fluoro-3-isopropyl-2-methyl-1,3-benzodiazol-5-yl)pyrimidin-2-amine 95% CAS 1231929-97-7](https://img.chemicalbook.in//Content/image/CP5.jpg) N-{5-[(4-ethylpiperazin-1-yl)methyl]pyridin-2-yl}-5-fluoro-4-(7-fluoro-3-isopropyl-2-methyl-1,3-benzodiazol-5-yl)pyrimidin-2-amine 95% CAS 1231929-97-7View Details
N-{5-[(4-ethylpiperazin-1-yl)methyl]pyridin-2-yl}-5-fluoro-4-(7-fluoro-3-isopropyl-2-methyl-1,3-benzodiazol-5-yl)pyrimidin-2-amine 95% CAS 1231929-97-7View Details
1231929-97-7 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1