2-Nitro-1-naphthol
- CAS NO.:607-24-9
- Empirical Formula: C10H7NO3
- Molecular Weight: 189.17
- MDL number: MFCD00003956
- EINECS: 210-131-7
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-07-16 09:52:10
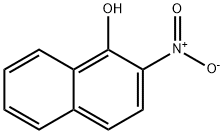
What is 2-Nitro-1-naphthol?
Description
2-Nitro-1-naphthol is an important organic compound with anti-inflammatory properties used as an analytical method for measuring albumin concentration and as an enzyme-labelled substrate in electrochemical immunoassays. The substance reacts with albumin to form a coloured complex which can be detected spectrophotometrically. It is also used as a model system for the study of inflammatory diseases and inhibits p38 kinase activity. In addition, a derivative of 2-Nitro-1-naphthol (4-nitro-1-naphthol) can be used to prepare chromogenic substrates for neutral or slightly acidic hydrolases.
Chemical properties
yellow-green to ochre-brown powder and/or chunks
The Uses of 2-Nitro-1-naphthol
2-Nitro-1-naphthol was employed as substrate for use in an amperometric 3-electrode system for the determination of alkaline phosphatase, the enzyme label most commonly used in electrochemical immunoassays. It was also used in the preparation of:
- di-(2-nitro-1-naphthyl) hydrogen phosphate
- 1-(2′-methyl-3′-indenyl)-2-naphthylamine and -2-naphthol (axially chiral ligands)
What are the applications of Application
2-Nitro-1-naphthol is commonly used as an intermediate in organic synthesis. It is a derivative of 1-nitro-2-naphthol and 4-nitro-1-naphthol. 1-Nitro-2-naphthol has been found to be a potent inhibitor of enzyme-dependent activation of acetyl-CoA, while 4-nitro-1-naphthol facilitates the preparation of chromogenic substrates for neutral or slightly acidic hydrolases[1-2].
Purification Methods
Crystallise the naphthol (repeatedly) from EtOH. [Beilstein 6 H 615, 6 III 2938, 6 IV 4236.]
References
[1] JIZHENG DANG . Chromogenic substrate from 4-nitro-1-naphthol for hydrolytic enzyme of neutral or slightly acidic optimum pH: 4-Nitro-1-naphthyl-β-d-galactopyranoside as an example[J]. Bioorganic & Medicinal Chemistry Letters, 2013. DOI:10.1016/j.bmcl.2012.11.120.
[2] ATSUKO SHINOHARA. Inhibition of acetyl-coenzyme A dependent activation of N-hydroxyarylamines by phenolic compounds, pentachlorophenol and 1-nitro-2-naphthol[J]. Chemico-Biological Interactions, 1986. DOI:10.1016/0009-2797(86)90058-X.
Properties of 2-Nitro-1-naphthol
| Melting point: | 123-125 °C (lit.) |
| Boiling point: | 324.41°C (rough estimate) |
| Density | 1.3175 (rough estimate) |
| refractive index | 1.5400 (estimate) |
| storage temp. | Sealed in dry,Room Temperature |
| pka | 6.13±0.50(Predicted) |
| InChI | InChI=1S/C10H7NO3/c12-10-8-4-2-1-3-7(8)5-6-9(10)11(13)14/h1-6,12H |
| CAS DataBase Reference | 607-24-9(CAS DataBase Reference) |
| NIST Chemistry Reference | 2-Nitro-1-naphthol(607-24-9) |
Safety information for 2-Nitro-1-naphthol
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P264:Wash hands thoroughly after handling. P264:Wash skin thouroughly after handling. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for 2-Nitro-1-naphthol
| InChIKey | MUCCHGOWMZTLHK-UHFFFAOYSA-N |
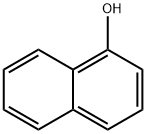
| SMILES | C1(O)=C2C(C=CC=C2)=CC=C1[N+]([O-])=O |
New Products
Indole Methyl Resin tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Boc-His(Boc)-OH 2-CTC Resin 4-Chloro-7-tosy1-7Hpyrrolo[2,3-d]pyrimidine 5,7-Dibromo-1H-indole 2,5-dichloro-N-hydroxy-4,6-dimethylpyridine-3-carboximidamide 2,2-Dimethoxy-7-azaspiro[3.5]nonane hydrochloride 4-chloromethyl-5-methyl-1,3-dioxol-2-one (DMDO-Cl) R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDIIMIDAZOLE 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) N-METHYL INDAZOLE-3-CARBOXYLIC ACID 4-((2-hydroxyethyl)thio)benzoic acid 1-(TERT-BUTOXYCARBONYL)-2-PYRROLIDINONE Methyl 6-methylnicotinate 3-Pyridineacrylic acid tert-Butyl carbazate TETRAHYDRO-2H-PYRAN-3-OL 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile 2,4-dihydroxybenzaldehyde 1,3-Diethyl-1,3-Diphenylurea Methyl 2-methylquinoline-6-carboxylateRelated products of tetrahydrofuran
You may like
-
 2-Nitro-1-naphthol CAS 607-24-9View Details
2-Nitro-1-naphthol CAS 607-24-9View Details
607-24-9 -
 Pyridine 99.5% HPLC /UV SpectroscopyView Details
Pyridine 99.5% HPLC /UV SpectroscopyView Details
110-86-1 -
 Guanine , 99%View Details
Guanine , 99%View Details
73-40-5 -
 Piperazine Spot supply, best priceView Details
Piperazine Spot supply, best priceView Details
110-85-0 -
 Dibutyl PhthalateView Details
Dibutyl PhthalateView Details
84-74-2 -
 Imidazole Spot supply, competitive priceView Details
Imidazole Spot supply, competitive priceView Details
288-32-4 -
 Octadecyl 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate 98% (GC)View Details
Octadecyl 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate 98% (GC)View Details
2082-79-3 -
 Thiourea 99% ARView Details
Thiourea 99% ARView Details
62-56-6
