Triethyl phosphite
Synonym(s):P(EtO)3;P(OEt)3;Triethylphosphite
- CAS NO.:122-52-1
- Empirical Formula: C6H15O3P
- Molecular Weight: 166.16
- MDL number: MFCD00009084
- EINECS: 204-552-5
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-09-25 17:15:13
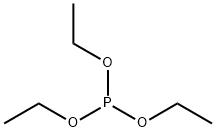
What is Triethyl phosphite?
Chemical properties
Triethyl phosphite is a clear colorless liquid with a strong foul odor. Insoluble in water; soluble in alcohol and ether. Combustible.Vapors heavier than air.
The Uses of Triethyl phosphite
Triethyl phosphite is used as a ligand in organometallic chemistry and as a reagent in organic synthesis. It is used as reference to phosphoric acid standard in 31P NMR spectroscopy. It acts as a reducing agent to prepare 2-phenylindazole from o-nitrobenzylidineaniline as well as reacts with electrophiles to get phosphonates. It forms a stable complex with copper(I) iodide. It finds application in a modified Staudinger reaction for the alkyl bromide to an amine through the azide. Further, it is also used in the preparation of 3-phenyl-2-substituted indoles by reacting with beta-nitro styrenes.
The Uses of Triethyl phosphite
Synthesis, plasticizers, stabilizers, lubricant and grease additives.
The Uses of Triethyl phosphite
Triethyl phosphite is an organophosphorus compound. It is used as a reducing agent; can react with electrophiles to form phosphonates or phosphates; forms a stable complex with copper(I) iodide.
Triethyl phosphite is a very good nucleophile. The carbon adjacent to the bromine is the most electrophilic position, and phosphorus is the only nucleophile. Triethyl phosphite displaces the bromine in an SN2-like process, and back attack by the bromide which is released generates a phosphonate product, in which the α-protons are strongly acidic.
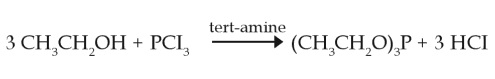
Preparation
Triethyl phosphite is produced in a closed system by reaction of phosphorous trichlorid and ethanol in the presence of an inorganic or organic base. The product is purified by distillation (Bayer AG, 2002a).
Triethyl phosphite is exclusively used as an intermediate for the manufacturing of different products: flame retardants (about 60 %), optical brighteners (about 15 %), pesticides (about 15 %), antioxidants (about 5 %), and pharmaceuticals (about 5 %).
Reactions
This reaction is currently used for the preparation of synthetically useful phosphonate reagents employed in modified retinal studies. Thus, diethyl 3-alkoxycarbonyl-2-propenylphosphonates are prepared in 72-91% yields by the reaction of methyl or ethyl 4-bromocrotonates with triethyl phosphite at 150-160°C. Similarly, diethyl 3-(ethoxycarbonyl)-2 methyl-2-propenylphosphonate is prepared in 81% yield from triethyl phosphite and ethyl 3-methyl-4-chlorocrotonate by heating at 180- 200°C.The diethyl (E)- and (Z)-3-ethoxycarbonyl-3 fluoro-2 -methyl-2-propenylphosphonates are respectively obtained from triethyl phosphite and (E)- or (Z)-4-bromo-2-fuoro-3-methyl-2-butenoates at 140°C.
Reactivity Profile
Triethyl phosphite is colorless, moderately toxic liquid, combustible. Flammable when exposed to heat or flame. When heated to decomposition Triethyl phosphite emits toxic fumes of oxides of phosphorus [Lewis, 3rd ed., 1993, p. 1271].
Health Hazard
Exposure to high concentrations may cause headache, nausea, and dizziness due to reduced chlolinesterase activity.
Fire Hazard
Special Hazards of Combustion Products: May form hazardous decomposition products.
Flammability and Explosibility
Flammable
Safety Profile
Moderately toxic by ingestion. A skin and eye irritant. Flammable liquid when exposed to heat, sparks, or flame. When heated to decomposition it emits toxic fumes of POx.
Toxicity evaluation
The acute toxicity after oral, dermal, and inhalation exposure is relatively low. The oral LD50s in rats ranged between 1840 mg/kg bw (females) and 2470 mg/kg bw (males). Symptoms of rapid breathing and tremors were observed prior to death. In mice LD50 values above 3700 mg/kg bw were recorded. The 6-hour inhalation LC50 with an aerosol of 1.6-3.5 μm MMAD in rats was between 11,100 mg/m3 (females) and 11,600 mg/m3 (males). Clinical signs included eye and upper respiratory irritation, salivation and rapid, shallow breathing. The dermal LD50 in rabbits was between 2800 mg/kg bw (males) and > 3000 mg/kg bw (females).
Properties of Triethyl phosphite
| Melting point: | -112°C |
| Boiling point: | 65 °C |
| Density | 0.969 g/mL at 25 °C(lit.) |
| vapor pressure | <6 hPa (20 °C) |
| refractive index | n |
| Flash point: | 130 °F |
| storage temp. | Flammables area |
| solubility | Chloroform (Slightly), Dichloromethane (Slightly), Methanol (Slightly) |
| form | liquid |
| color | colorless |
| Specific Gravity | 0.969 |
| Odor | characteristic, obnoxious, phosphiteodor |
| explosive limit | 3.75-42.5%(V) |
| Water Solubility | slightly soluble |
| Sensitive | Air & Moisture Sensitive |
| Hydrolytic Sensitivity | 7: reacts slowly with moisture/water |
| BRN | 956578 |
| Dielectric constant | 10.9(65℃) |
| Stability: | Air Sensitive, Moisture Sensitive |
| CAS DataBase Reference | 122-52-1(CAS DataBase Reference) |
| NIST Chemistry Reference | Phosphorous acid, triethyl ester(122-52-1) |
| EPA Substance Registry System | Triethyl phosphite (122-52-1) |
Safety information for Triethyl phosphite
| Signal word | Warning |
| Pictogram(s) |
 Flame Flammables GHS02  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H226:Flammable liquids H302:Acute toxicity,oral H317:Sensitisation, Skin H412:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P233:Keep container tightly closed. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P301+P312:IF SWALLOWED: call a POISON CENTER or doctor/physician IF you feel unwell. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. |
Computed Descriptors for Triethyl phosphite
Triethyl phosphite manufacturer
JSK Chemicals
NSR Amino Organics
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran


You may like
-
 Triethyl phosphite 122-52-1 99%View Details
Triethyl phosphite 122-52-1 99%View Details
122-52-1 -
 122-52-1 98%View Details
122-52-1 98%View Details
122-52-1 -
 Triethyl phosphite, 97% 122-52-1 99%View Details
Triethyl phosphite, 97% 122-52-1 99%View Details
122-52-1 -
 Triethyl phosphite CAS 122-52-1View Details
Triethyl phosphite CAS 122-52-1View Details
122-52-1 -
 TRIETHYL PHOSPHITE For Synthesis CAS 122-52-1View Details
TRIETHYL PHOSPHITE For Synthesis CAS 122-52-1View Details
122-52-1 -
 Triethyl phosphite 97% CAS 122-52-1View Details
Triethyl phosphite 97% CAS 122-52-1View Details
122-52-1 -
 Triethylphosphite CAS 122-52-1View Details
Triethylphosphite CAS 122-52-1View Details
122-52-1 -
 Triethyl phosphite(TEP) 78-40-0 98%View Details
Triethyl phosphite(TEP) 78-40-0 98%View Details
122-52-1
