tilidine
- CAS NO.:20380-58-9
- Empirical Formula: C17H23NO2
- Molecular Weight: 273.374
- EINECS: 2437747
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-05-04 17:34:35
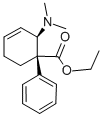
What is tilidine?
Originator
Valoron,Goedecke,W. Germany,1970
The Uses of tilidine
Tilidine is an opioid used primarily as a painkiller it inhibits the enzyme adenylyl cyclase.
Definition
ChEBI: Dextilidine is an ethyl 2-(dimethylamino)-1-phenylcyclohex-3-ene-1-carboxylate that has S configuration at the carbon bearing the phenyl group and R configuration at the carbon bearing the dimethylamino group. The opioid analgesic tilidine is the racemate comprising equimolar amounts of dextilidine and its enantiomer, ent-dextilidine. A prodrug, tilidine is converted by the liver to the active analgesic, nortilidine; virtually all of the opioid activity resides in the (1S,2R) isomer (i.e. the isomer derived from dextilidine). It has a role as an opioid analgesic and a prodrug. It is an enantiomer of an ent-dextilidine.
Manufacturing Process
In a first step, dimethylamine is reacted with crotonaldehyde to give 1-
(dimethylamino)-1,3-butadiene.
A solution of 194 grams (2 mols) of fresh-distilled 1-(dimethylamino)-1,3-
butadiene is combined at room temperature in a 1 liter round-bottom flask
with 352 grams (2 mols) atropic acid ethyl ester. After being stirred for about
10 minutes, the reaction mixture gradually becomes exothermic. By cooling
with ice water, the contents of the flask are kept at a temperature of 40° to
60°C. After the reaction has ceased, the mixture is kept overnight (about 8 to 24 hours) at room temperature. The next day the viscous product is dissolved
in 10 liters of ether and precipitated with ethereal hydrogen chloride forming
the corresponding hydrochloride. By fractional crystallization from ethyl
acetate/methyl ethyl ketone (10:1), an almost complete separation of the
isomeric cis/trans isomers (I) and (II) is achieved. The separation can be
carried out very easily due to the low solubility of the 1 1/2-hydrate of (I).
Therefore, during the crystallization a sufficient quantity of water for the
formation of the 1 1/2-hydrate of (I) is added to the mixture of solvents,
whereby (I) readily precipitates.
Isomer (I): 4-phenyl-3-cis-dimethylamino-4-cis-carbethoxy-?1-
cyclohexenehydrochloride, [ethyl-cis-3-(dimethylamino)-4-phenyl-1-
cyclohexene-4-carboxylate hydrochloride] , MP 84°C (the free base boils at
97.5° to 98°C at 0.01 mm pressure), 64.4% yield.
Isomer (II): 4-phenyl-3-trans-dimethylamino-4-trans-carbethoxy-?1-
cyclohexenehydrochloride, [ethyl-trans-3-(dimethylamino)-4-phenyl-1-
cyclohexene-4-carboxylate hydrochloride], MP 159°C (the free base boils at
95.5° to 96°C at 0.01 mm pressure), 22.2% yield.
Therapeutic Function
Analgesic
Properties of tilidine
| Boiling point: | bp0.01 95.5-96° |
Safety information for tilidine
Computed Descriptors for tilidine
New Products
4-AMINO-TETRAHYDRO-PYRAN-4-CARBOXYLIC ACID HCL 4-(Dimethylamino)tetrahydro-2H-pyran-4-carbonitrile 4-Aminotetrahydropyran-4-carbonitrile Hydrochloride (R)-3-Aminobutanenitrile Hydrochloride 3-((Dimethylamino)methyl)-5-methylhexan-2-one oxalate 1,4-Dioxa-8-azaspiro[4.5]decane 5-Bromo-2-nitropyridine Nimesulide BP Aceclofenac IP/BP/EP Diclofenac Sodium IP/BP/EP/USP Mefenamic Acid IP/BP/EP/USP Ornidazole IP Diclofenac Potassium THOMAIND PAPER PH 2.0 TO 4.5 1 BOX BUFFER CAPSULE PH 9.2 - 10 CAP SODIUM CHLORIDE 0.1N CVS ALLOXAN MONOHYDRATE 98% PLATINUM 0.5% ON 3 MM ALUMINA PELLETS (TYPE 73) LITHIUM AAS SOLUTION 2-Bromo-1-(bromomethyl)-3-chloro-5-nitrobenzene 2-Bromo-3-nitroaniline N-(3-Hydroxypropyl)-N-methylacetamide 3-Bromo-6-chloropyridazine 4-ethyl-3-nitrobenzoic acidRelated products of tetrahydrofuran

You may like
-
 1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
1-Methyl-6-oxo-1,6-dihydropyridazine-3-carbonitrile 98%View Details
99903-60-3 -
 88491-46-7 98%View Details
88491-46-7 98%View Details
88491-46-7 -
 1823368-42-8 98%View Details
1823368-42-8 98%View Details
1823368-42-8 -
 2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
2-(3-(tert-butyl)phenoxy)-2-methylpropanoic acid 1307449-08-6 98%View Details
1307449-08-6 -
 Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
Ethyl 3-(furan-2-yl)-3-hydroxypropanoate 25408-95-1 98%View Details
25408-95-1 -
 2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
2-Chloro-5-fluoro-1-methoxy-3-methylbenzene 98%View Details
1805639-70-6 -
 1784294-80-9 98%View Details
1784294-80-9 98%View Details
1784294-80-9 -
 Lithium ClavulanateView Details
Lithium ClavulanateView Details
61177-44-4