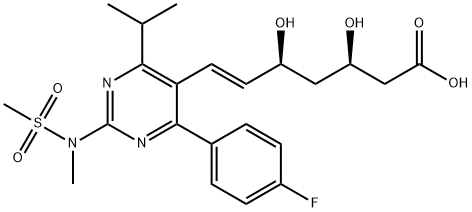
Rosuvastatin calcium
Synonym(s):(E)-7-[4-(4-Fluorophenyl)-6-isopropyl-2-[methyl(methylsulfonyl)amino]pyrim idin-5-yl]-(3R,5S)-3,5-dihydroxyhept-6-enoic acid, calcium salt (2:1);Rosuvastatin calcium salt;Rosuvastatin hemicalcium
- CAS NO.:147098-20-2
- Empirical Formula: C22H30CaFN3O6S
- Molecular Weight: 523.63
- MDL number: MFCD04112702
- EINECS: 627-028-1
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-16 16:15:04
What is Rosuvastatin calcium?
Description
Rosuvastatin, one of the two new statins launched for the treatment of hypercholesterolemia, has high hepato-selectivity and more potent inhibitory effect on HMG-CoA reductase than the previously marketed statins. In rat hepatocytes, it inhibits cholesterol biosynthesis with an IC50 of 1.12 nM, which is ~100-fold higher potency than pravastatin. Rosuvastatin is synthesized in a 12-step sequence, entailing the construction of a pyrimidinyl aldehyde intermediate in eight steps and subsequent introduction of the dihydroxyheptenoate side chain via Wittig reaction with a bketophosphorane reagent and stereoselective carbonyl reduction of the resultant enone. Pharmacokinetic properties of rosuvastatin in humans, dosed at 5–80 mg, are approximately linear with dose. Following oral administration, rosuvastatin is rapidly absorbed with an oral bioavailability of ~20% and tmax of ~3 h. It has a prolonged duration of action, with terminal t1/2 of ~20 h, compatible with once-daily dosing. In humans, rosuvastatin is minimally metabolized through CYP2C9 and CYP2C19, with little or no metabolism via the CYP3A4. Approximately 90% of the administered oral dose is eliminated in the feces (92% as the parent compound) and the rest in the urine. Rosuvastatin is considered a “superstatin” due to its ability, at well-tolerated doses, to lower LDL cholesterol and triglycerides to a much greater extent than first generation statins. In patients with hypercholesterolemia, rosuvastatin treatment at doses of 5 and 10 mg/day over 12-week period resulted in 40–43% reduction of LDL-cholesterol levels, 12–13% increase in HDL-cholesterol, and 17– 19% reduction in triglycerides. In comparison, the efficacy range of LDL-cholesterol reductions by atorvastatin (10 mg/day), pravastatin (20 mg/day), and simvastatin (20 mg/day) was 28–35%. Rosuvastatin is a well-tolerated drug at doses of 1–20 mg and the most common side effects at these doses are headache, myalgia, pain and pharyngitis, which are consistent with those previously reported for statin therapy.
Chemical properties
White to Off-White Crystalline Solid
Originator
Shinogi (Japan)
The Uses of Rosuvastatin calcium
Rosuvastatin Calcium Salt is a selective, competitive HMG-CoA reductase inhibitor. Antilipemic.
The Uses of Rosuvastatin calcium
antiinflammatory, glucocorticoid
The Uses of Rosuvastatin calcium
antirheumatic
What are the applications of Application
Rosuvastatin calcium is commonly used to lower bad cholesterol levels (LDL-C) and fats (triglycerides) in the blood. It also increases good cholesterol levels (HDL). Improving your cholesterol levels helps decrease your risk of heart disease, stroke, and heart attack.
Rosuvastatin calcium may also be used for other conditions as determined by your healthcare provider.
Definition
ChEBI: An organic calcium salt that is the hemicalcium salt of rosuvastatin.
brand name
Crestor (AstraZeneca).
General Description
Rosuvastatin calcium (ROS) has a molecular mass of 1001.14. ROS is a member of the "statins" group and is a 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor.
Biochem/physiol Actions
Rosuvastatin calcium is a competitive inhibitor of hydroxymethylglutaryl-coenzyme A (HMG-CoA) reductase, the enzyme that catalyzes the conversion of HMG-CoA to mevalonic acid, the rate-limiting step in cholesterol biosynthesis. Rosuvastatin calcium is antilipemic and is used to reduce plasma cholesterol levels and prevent cardiovascular disease.
Mechanism of action
Rosuvastatin is a statin medication and a competitive inhibitor of the enzyme HMG-CoA (3-hydroxy-3-methylglutaryl coenzyme A) reductase, which catalyzes the conversion of HMG-CoA to mevalonate, an early rate-limiting step in cholesterol biosynthesis.24 Rosuvastatin acts primarily in the liver, where decreased hepatic cholesterol concentrations stimulate the upregulation of hepatic low density lipoprotein (LDL) receptors which increases hepatic uptake of LDL. Rosuvastatin also inhibits hepatic synthesis of very low density lipoprotein (VLDL).44 The overall effect is a decrease in plasma LDL and VLDL.
Synthesis
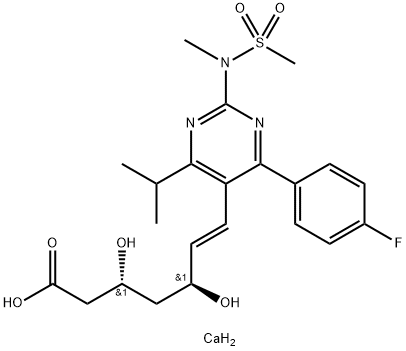
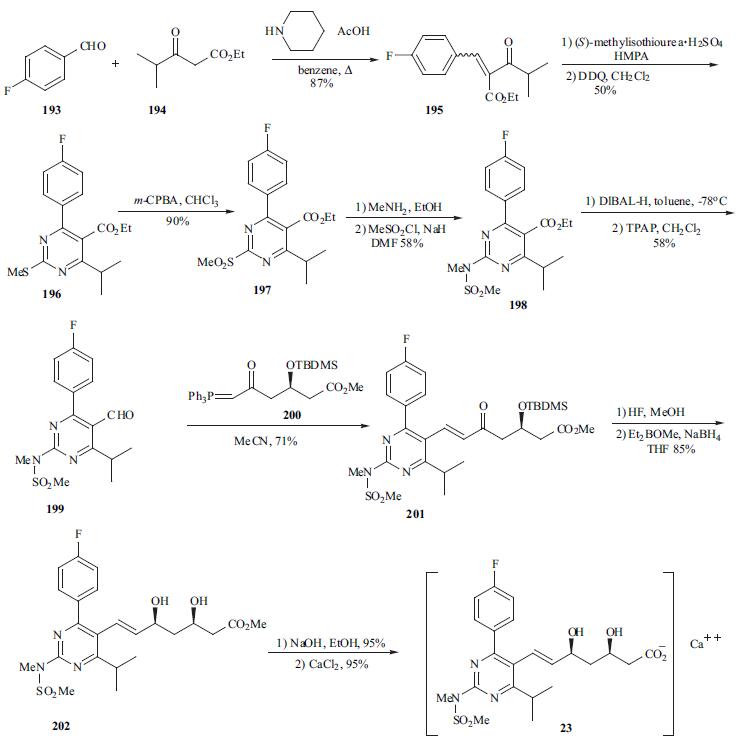
The synthesis of optically pure rosuvastatin (23) begins from the Knoevenagel reaction of p-fluorobenzaldehyde (193) with ethyl isobutylacetate (194) to give unsaturated ketoester 195. Compound 195 was condensed with (S ) - methylisothiourea and then aromatized in situ using DDQ in methylene chloride to give pyrimidine 196 in 50% yield. Pyrimidine sulfide 196 was then oxided by m-CPBA to give sulfone 197 in 96% yield. Sulfone 197 was reacted with methylamine in methanol followed by treatment with methanesulfonyl chloride to give the N - methanesulfonylamino pyrimidine 198 in 58% yield. Reduction of ester 198 with DIBAL-H followed by TPAP oxidation afforded aldehyde 199 in 58% yield. Aldehyde 199 was subjected to Wittig reaction with optically pure ylide, (3R)-3-(t-butyldimethylsilyloxy)-5-oxo-6-triphenylphosphoranylidenehexanoate (200), to give heptenoate compound 201 in 71% yield. Compound 201 was deprotected with HF in acetonitrile, and stereoselective chelation-controlled reduction with Et2BOMe and NaBH4 in THF-MeOH mixed solvent gave methyl (3R, 5S, 6E)- dihydroxyheptenoate 202 in 85% yield. Diol 202 was hydrolyzed with aqueous NaOH to afford the corresponding sodium salt. Rosuvastatin calcium salt (23) was obtained as white powder from the sodium salt on treatment with aqueous CaCl2.
Storage
Store at +4°C
Properties of Rosuvastatin calcium
| Melting point: | 122°C |
| alpha | D24 +14.8° (c = 1.012 in 50% methanol) |
| storage temp. | 2-8°C |
| solubility | DMSO : 25 mg/mL (49.94 mM; Need ultrasonic)H2O : 1 mg/mL (2.00 mM; Need ultrasonic) |
| form | powder |
| color | white to beige |
| optical activity | [α]/D +12 to +18°, c = 1 in methanol: water (1:1) |
| λmax | 243nm(Phosphate buffer sol.)(lit.) |
| Merck | 14,8270 |
| CAS DataBase Reference | 147098-20-2(CAS DataBase Reference) |
Safety information for Rosuvastatin calcium
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Rosuvastatin calcium
| InChIKey | JSVIQRTVYKCCOM-LMRKSJJTNA-N |
| SMILES | C(/C1=C(N=C(N(C)S(=O)(=O)C)N=C1C1C=CC(F)=CC=1)C(C)C)=C\[C@@H](O)C[C@@H](O)CC(=O)O.[Ca] |&1:24,27,r| |
Rosuvastatin calcium manufacturer
Allas Chem Technologies Pvt Ltd
Archerchem Healthcare Pvt., Ltd. (part of Archerchem Group)
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran
![(4R,6S)-6-[(1E)-2-[2-Cyclopropyl-4-(4-fluorophenyl)-3-quinolinyl]ethenyl]-2,2-dimethyl-1,3-dioxane-4-acetic acid tert-butyl ester](https://img.chemicalbook.in/CAS/GIF/147489-06-3.gif)
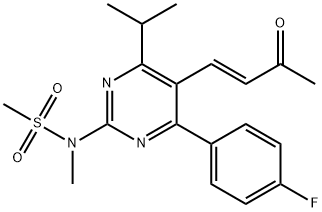
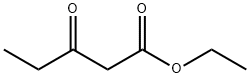
![Phosphonium, [[4-(4-fluorophenyl)-6-(1-methylethyl)-2-[methyl(methylsulfonyl)amino]-5-pyrimidinyl]me](https://img.chemicalbook.in/CAS/GIF/885477-83-8.gif)
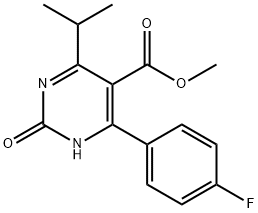
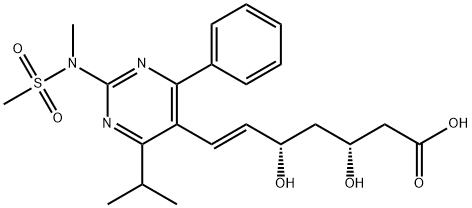
You may like
-
 Rosuvastatin Calcium 97%View Details
Rosuvastatin Calcium 97%View Details -
 Rosuvastatin Calcium 99%View Details
Rosuvastatin Calcium 99%View Details -
 Rosuvastatin S- isomer 98%View Details
Rosuvastatin S- isomer 98%View Details -
 ROSUVASTATIN CALCIUM 99%View Details
ROSUVASTATIN CALCIUM 99%View Details -
 Rosuvastatin Calcium 99%View Details
Rosuvastatin Calcium 99%View Details -
 Rosuvastatin Calcium 98%View Details
Rosuvastatin Calcium 98%View Details -
 Rosuvastatin Calcium 99%View Details
Rosuvastatin Calcium 99%View Details -
 Rosuvastatin Calcium CASView Details
Rosuvastatin Calcium CASView Details
