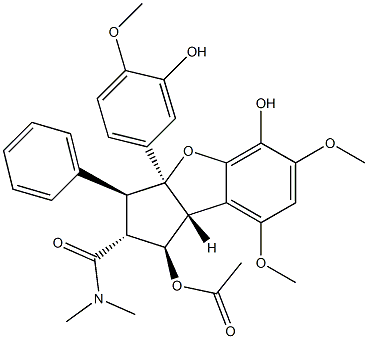
ROCAGLAMIDE
Synonym(s):(1R,2R,3S,3aR,8bS)-2,3,3a,8b-Tetrahydro-1,8b-dihydroxy-6,8-dimethoxy-3a-(4-methoxyphenyl)-N,N-dimethyl-3-phenyl-1H-cyclopenta[b]benzofuran-2-carboxamide;NSC 326408;Roc-A;Rocaglamide A
- CAS NO.:84573-16-0
- Empirical Formula: C29H31NO7
- Molecular Weight: 505.56
- MDL number: MFCD08702699
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-11-14 14:09:35
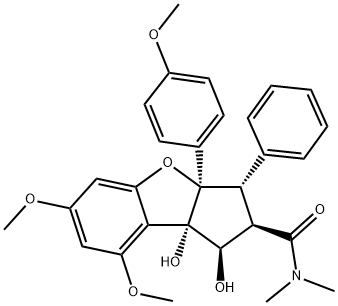
What is ROCAGLAMIDE?
Description
Rocaglamide is an anti-
The Uses of ROCAGLAMIDE
Rocaglamide is derived from a Chinese medicinal plant Aglaia. Rocaglamide induces apoptosis in various human leukemia cell lines and in acute lymphoblastic leukemia, chronic myeloid leukemia and acute myeloid leukemia cells freshly isolated from patients.
What are the applications of Application
Rocaglamide is an immunosuppressant tetrahydrobenzofuran compound and an inhibitor of NFκB activity.
Definition
ChEBI: An organic heterotricyclic compound that is 2,3,3a,8b-tetrahydro-1H-benzo[b]cyclopenta[d]furan substituted by hydroxy groups at positions 1 and 8b, methoxy groups at positions 6 and 8, a 4-methoxyphenyl group at position 3a, a phenyl group at position 3 and a N,N-dimethylcarbamoyl group at position 1. Isolated from Aglaia odorata and Aglaia duperreana, it exhibits antineoplastic activity.
General Description
Rocaglamide is known to be derived from tetrahydro benzofuran and is also an active chemical compound of Aglaia plants.
Biochem/physiol Actions
Rocaglamide is a potent anticancer agent isolated from the genus Aglaia. Rocaglamides inhibit protein synthesis without affecting DNA or RNA synthesis. Recent study shows that Rocaglamide binds to prohibitin (PHB) 1 and 2, which prevents interaction between PHB and CRaf and inhibits CRaf activation and subsequently CRaf-MEK-ERK signaling. Also, Rocaglamide is an immunosuppressant that inhibits activation of NF-kB and NF-AT.
References
1) Baumann?et al.?(2002),?Rocaglamide Derivatives Are Potent Inhibitors of NF-kB Activation in T-cells; J. Biol. Chem.?277?44791 2) Prolsch?et al.?(2005),?Rocaglamide Derivatives Are Immunosuppressive Phytochemicals That Target NF-AT Activity in T Cells; J. Immunol.?174?7075 3) Polier?et al.?(2012),?The natural anticancer compounds rocaglamides inhibit the Raf-MEK-ERK pathway by targeting prohibitin1 and 2; Chem. Biol.?19?1093 4) Neumann?et al.?(2014),?The natural anticancer compound rocaglamide selectively inhibits the G1-S phase transition in cancer cells through the ATM/ATR-mediated Chk1/2 cell cycle checkpoints; Int. J. Cancer?134?1991 5) Zhu?et al.?(2007),?The traditional Chinese herbal compound rocaglamide preferentially induces apoptosis in leukemia cells by modulation of mitogen-activated protein kinase activities; Int. J. Cancer?121?1839
Properties of ROCAGLAMIDE
| Melting point: | 117-118 °C |
| Boiling point: | 667.3±55.0 °C(Predicted) |
| Density | 1.321±0.06 g/cm3(Predicted) |
| storage temp. | -20°C |
| solubility | Soluble in DMSO or Ethanol |
| pka | 11.70±0.70(Predicted) |
| form | film |
| color | colorless |
| Stability: | Stable for 2 years from date of purchase as supplied. Solutions in DMSO or ethanol may be stored at -20°C for up to 1 month. Protect from exposure to light and moisture. |
Safety information for ROCAGLAMIDE
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for ROCAGLAMIDE
New Products
Tert-butyl bis(2-chloroethyl)carbamate (S)-3-Aminobutanenitrile hydrochloride N-Boc-D-alaninol N-BOC-D/L-ALANINOL 3-(2,4-Dimethoxybenzyl)dihydropyrimidine-2,4(1H,3H)-dione 6-Bromo-3-iodo-1-methyl-1H-indazole N-octanoyl benzotriazole 3,4-Dibenzyloxybenzaldehyde 4-Hydrazinobenzoic acid Electrolytic Iron Powder 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 4-HYDROXY BENZYL ALCOHOL 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) S-2-CHLORO PROPIONIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine N-Boc-L-proline methyl ester 2-(BOC-Amino)4-picoline 1-(4-Methylphenylsulfonyl)-1H-1,2,3-benzotriazole 1-(2-Chlorobenzyl)-4-nitro-1H-pyrazoleRelated products of tetrahydrofuran

You may like
-
 Rocaglamide CAS 84573-16-0View Details
Rocaglamide CAS 84573-16-0View Details
84573-16-0 -
 55441-95-7 2 2-BIS(2-HYDROXYETHOXY)-1 1-BINAPHTHYL 99%View Details
55441-95-7 2 2-BIS(2-HYDROXYETHOXY)-1 1-BINAPHTHYL 99%View Details
55441-95-7 -
 Ste-Glu-AEEA-AEEA-OSUView Details
Ste-Glu-AEEA-AEEA-OSUView Details
1169630-40-3 -
 1446013-08-6 Fmoc-His-Aib-OH TFA 98%View Details
1446013-08-6 Fmoc-His-Aib-OH TFA 98%View Details
1446013-08-6 -
 127464-43-1 99%View Details
127464-43-1 99%View Details
127464-43-1 -
 Chloro Uracil 99%View Details
Chloro Uracil 99%View Details
1820-81-1 -
 2-ETHYLPYRIDINE 100-71-0 99%View Details
2-ETHYLPYRIDINE 100-71-0 99%View Details
100-71-0 -
 13162-05-5 99%View Details
13162-05-5 99%View Details
13162-05-5