Ranibizumab
- CAS NO.:347396-82-1
- Empirical Formula: C11H12N2O2
- Molecular Weight: 204.22518
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-07-02 08:55:00
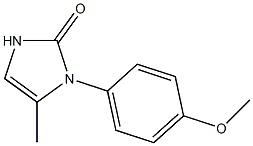
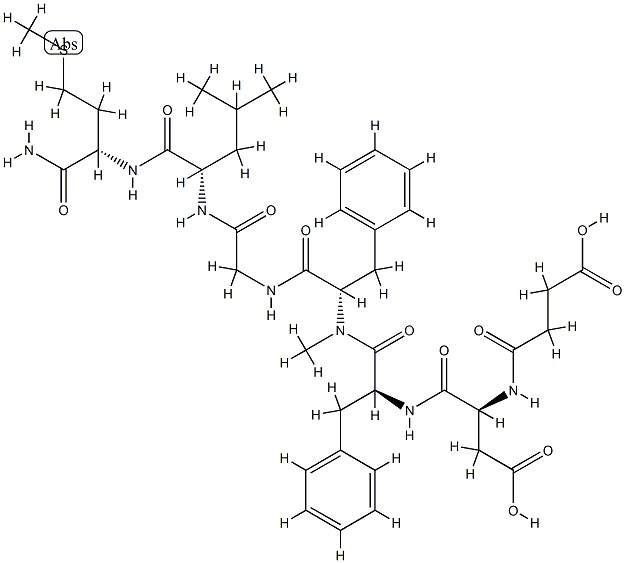
What is Ranibizumab?
Description
Ranibizumab is a recombinant, humanized, IgG1 monoclonal antibody fragment (Fab) that neutralizes all active forms of vascular endothelial growth factor A (VEGF-A), and it is indicated for the treatment of neovascular age-related macular degeneration (AMD). It consists of a nonbinding human sequence and a high-affinity binding epitope (Fab fragment) derived from the mouse. The full-length RhuMab VEGF (bevacizumab) was launched previously by Genentech for the treatment of colorectal cancer. Both the antibody fragment and the full-length antibody bind to and inhibit all active forms of VEGF-A and are derived from the same mouse monoclonal antibody. However, ranibizumab has been genetically engineered through a process of selective mutation to increase its affinity for binding and inhibiting the growth factor.
Originator
Genentech (US)
The Uses of Ranibizumab
Ranibizumab, a humanized antigen binding portion of a murine anti- VEGF monoclonal antibody with a mature high affinity for all VEGF isoforms, has been approved as an intravitreal treatment for neovascular AMD and is the first such treatment to improve visual acuity (VA) in neovascular AMD.
CRUISE is multicenter, randomized, double-masked, sham injectioncontrolled Phase III study of 392 participants designed to assess the efficiency and safety profile of ranimizumab for macular edema associated to CRVO. The month 6 result showed that 46.2% (61/132) of patients received 0.3 mg of ranimizumab and 47.7% (62/130) received 0.5 mg of ranimizumab had their vision improved by 15 letters or more compared to 16.9% (22/130) of patients receiving sham injections and mean gain was observed beginning at day seven with an 8.8 and 9.3 letter gain in the 0.3 mg and 0.5 mg study arms of ranimizumab, respectively, compared with 1.1 letters in the sham injection arm.
The Uses of Ranibizumab
antiangiogenic monoclonal antibody macular degeneration therapy
brand name
Lucentis
Properties of Ranibizumab
| form | Liquid |
| color | Colorless to light yellow |
| CAS DataBase Reference | 347396-82-1 |
Safety information for Ranibizumab
Computed Descriptors for Ranibizumab
New Products
4-Fluorophenylacetic acid 4-Methylphenylacetic acid N-Boc-D-alaninol N-BOC-D/L-ALANINOL Tert-butyl bis(2-chloroethyl)carbamate 3-Morpholino-1-(4-nitrophenyl)-5,6-dihydropyridin- 2(1H)-one Furan-2,5-Dicarboxylic Acid Tropic acid S-2-CHLORO PROPIONIC ACID ETHYL ISOCYANOACETATE 2-Bromo-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate (6-METHYL-[1,3]DITHIOLO[4,5-b]QUINOXALIN-2-ONE INDAZOLE-3-CARBOXYLIC ACID 4-IODO BENZOIC ACID (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5,6-Dimethoxyindanone 2-(Cyanocyclohexyl)acetic acid 4-methoxy-3,5-dinitropyridine 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate tert-butyl 4- (ureidomethyl)benzylcarbamate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran







You may like
-
 2033-24-1 98%View Details
2033-24-1 98%View Details
2033-24-1 -
 42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID 98%View Details
42831-50-5 -
 1975-50-4 98%View Details
1975-50-4 98%View Details
1975-50-4 -
 2-HYDROXY BENZYL ALCOHOL 98%View Details
2-HYDROXY BENZYL ALCOHOL 98%View Details
90-01-7 -
 2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
2-Chloro-1,3-Bis(Dimethylamino)Trimethinium Hexafluorophosphate 221615-75-4 98%View Details
221615-75-4 -
 61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 CIS BROMO BENZOATE 98%View Details
61397-56-6 -
 14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 (2-Hydroxyphenyl)acetonitrile 98+View Details
14714-50-2 -
 118753-70-1 98+View Details
118753-70-1 98+View Details
118753-70-1