Pyrene
Synonym(s):Benzo[def]phenanthrene;Benzo[def]phenanthrene;Pyrene
- CAS NO.:129-00-0
- Empirical Formula: C16H10
- Molecular Weight: 202.25
- MDL number: MFCD00004136
- EINECS: 204-927-3
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-11-07 08:30:04
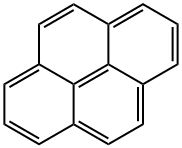
What is Pyrene?
Description
Pyrene is an organic compound with chemical formula C16H10, light yellow monoclinic crystal (pure product is colorless), aromatic, combustible[1], insoluble in water and soluble in ethanol and ether. It can carry out electrophilic substitution, such as halogenation, nitration, sulfonation and other reactions. Pyrene mainly exists in the distillate of coal tar pitch. Pyrene is an organic synthetic raw material, which can be oxidized to produce 1,4,5,8-naphthalene tetracarboxylic acid, which is used in dyes, synthetic resins, disperse dyes and engineering plastics; After acylation, the vat dye brilliant orange GR and other dyes can be prepared. It can also make pesticides, plasticizers, etc. On October 27, 2017, the list of carcinogens published by the international agency for research on cancer of the World Health Organization was preliminarily sorted out for reference. Pyrene was included in the list of three types of carcinogens.
Chemical properties
pale yellow to yellow-greenish crystals or chunks
Chemical properties
Pyrene is a colorless crystalline solid when pure or pale yellow plates (impure). Polycyclic aromatic hydrocarbons (PAHs) are compounds containing multiple benzene rings and are also called polynuclear aromatic hydrocarbons. Solids and solutions have a Blue fluores- cence (Merck Index).
Physical properties
Colorless solid (tetracene impurities impart a yellow color) or monoclinic prisms crystallized from alcohol. Solutions have a slight blue fluorescence.
The Uses of Pyrene
Pyrene occurs in coal tar. Also obtained by the destructive hydrogenation of hard coal. Found in wastewater in aquatic environments, and possesses genotoxic characteristics relating to estrogenic/andr ogenic, antiestrogenic and antiandrogenic activity.
The Uses of Pyrene
Pyrene and its derivatives are used commercially to make?dyes?and dye precursors, for example?pyranine?and naphthalene-1,4,5,8-tetracarboxylic acid. It is used as a probe to determine solvent environments and fluorescence spectroscopy.
The Uses of Pyrene
Pyrene may be used as an analytical reference standard for the quantification of the analyte in environmental tobacco smoke samples using high-performance liquid chromatography technique.
Definition
A condensed ring hydro- carbon.
Definition
A solid aromatic compound whose molecules consist of four benzene rings joined together. It is carcinogenic.
Production Methods
There is no commercial production or known use for this compound. Pyrene has been used as starting material for the synthesis of benzo[a]pyrene.
General Description
Colorless solid, solid and solutions have a slight blue fluorescence. Used in biochemical research.
Air & Water Reactions
Insoluble in water.
Reactivity Profile
Pyrene reacts with nitrogen oxides to form nitro derivatives. Pyrene also reacts with 70% nitric acid.
Hazard
A questionable carcinogen, absorbed by skin.
Health Hazard
Pyrene is a carcinogenic agent and is absorbed by the skin. It is a skin irritant, a suspected mutagen, and an equivocal tumor-causing agent. Workers exposed to 3 to 5 mg/m3 of Pyrene exhibited some teratogenic effects. Pyrene is a polycyclic aromatic hydrocarbon (PAH). The acute toxicity of pure PAHs appears low when administered orally or dermally to rats or mice. Human exposure to PAHs is almost exclusively via the gastrointestinal and respiratory tracts, and approximately 99 percent is ingested in the diet. Despite the high concentrations of Pyrene to which humans may be exposed through food, there is currently little information available to implicate diet-derived PAHs as the cause of serious health effects.
Health Hazard
Inhalation of its vapors or ingestion causedirritation of the eyes, excitement, and musclecontraction in rats and mice. An oral LD50value in mice has been reported as 800 mg/kg.Studies on experimental animals do not giveevidence of carcinogenicity. Skin tumors,however, have been reported in mice (NIOSH1986). Pyrene tested negative to a histidinereversion–Ames test and other mutagenictests.
Fire Hazard
When heated to decomposition, Pyrene emits acrid smoke and fumes.
Safety Profile
Poison by inhalation. Moderately toxic by ingestion and intraperitoneal routes. A sh irritant. Questionable carcinogen with experimental tumorigenic data. Human mutation data reported. When heated to decomposition it emits acrid smoke and irritating fumes.
Synthesis
The content in high temperature tar is about 1.2% ~ 1.8%, which is prepared by separation and purification. Using anthracene oil as raw material, vacuum distillation, extraction, recrystallization and purification. The residue of distilled anthracene oil above 360 ℃ is used as raw material, the pyrene fraction is cut by distillation, benzene is used as solvent, washed with concentrated sulfuric acid, the base and unsaturated compounds are removed, and then the pure product is recrystallized with solvent oil. It can also be extracted from tobacco. Pyrene mainly exists in the distillate of coal tar pitch. The medium temperature asphalt is distilled under reduced pressure, and a small amount of direct superheated steam is introduced into the distillation kettle to cut the narrow fraction of pyrene, and then recrystallized with the mixed solution of solvent oil and ethanol or the mixed solution of benzene and solvent oil to obtain industrial pyrene with a purity of 95%.
Potential Exposure
Pyrene is used as an industrial chemi- cal and in biochemical research.
Carcinogenicity
Pyrene was inactive as a tumor initiator. Based on inadequate animal data and no human data, the IARC classified pyrene as group 3, not classifiable as to its carcinogenicity to humans, and IRIS classified pyrene as a class D compound. Pyrene is present as a major component of the total content of PAHs in the environment. Exposure occurs primarily through tobacco smoke, inhalation of polluted air, and by ingestion of food and water.
Source
Detected in groundwater beneath a former coal gasification plant in Seattle, WA at a concentration of 180 μg/L (ASTR, 1995). Detected in 8 diesel fuels at concentrations ranging
from 0.16 to 24 mg/L with a mean value of 5.54 mg/L (Westerholm and Li, 1994). Identified in
Kuwait and South Louisiana crude oils at concentrations of 4.5 and 3.5 ppm, respectively
(Pancirov and Brown, 1975).
Based on laboratory analysis of 7 coal tar samples, pyrene concentrations ranged from 900 to
18,000 ppm (EPRI, 1990). Detected in 1-yr aged coal tar film and bulk coal tar at concentrations
of 2,700 and 2,900 mg/kg, respectively (Nelson et al., 1996). Lehmann et al. (1984) reported a
pyrene concentration of 125 mg/g in a commercial anthracene oil. Identified in high-temperature
coal tar pitches used in electrodes at concentrations ranging from 4,500 to 34,900 mg/kg
(Arrendale and Rogers, 1981). Lee et al. (1992a) equilibrated eight coal tars with distilled water at
25 °C. The maximum concentration pyrene observed in the aqueous phase is 0.1 mg/L.
Nine commercially available creosote samples contained pyrene at concentrations ranging from
31,000 to 100,000 mg/kg (Kohler et al., 2000). Also detected in asphalt fumes at an average
concentration of 140.21 ng/m3 (Wang et al., 2001).
Schauer et al. (1999) reported pyrene in diesel fuel at a concentration of 64 μg/g and in a dieselpowered
medium-duty truck exhaust at an emission rate of 71.9 μg/km.
California Phase II reformulated gasoline contained pyrene at a concentration of 3.38 g/kg. Gasphase
tailpipe emission rates from gasoline-powered automobiles with and without catalytic
converters were approximately 4.28 and 160 μg/km, respectively (Schauer et al., 2002).
Schauer et al. (2001) measured organic compound emission rates for volatile organic
compounds, gas-phase semi-volatile organic compounds, and particle-phase organic compounds
from the residential (fireplace) combustion of pine, oak, and eucalyptus. The respective gas-phase
and particle-phase emission rates of pyrene were 1.87 and 3.78 mg/kg of pine burned, 2.40 and
1.23 mg/kg of oak burned, and 2.70 and 0.585 mg/kg of eucalyptus burned.
Under atmospheric conditions, a low rank coal (0.5–1 mm particle size) from Spain was burned
in a fluidized bed reactor at seven different temperatures (50 °C increments) beginning at 650 °C.
The combustion experiment was also conducted at different amounts of excess oxygen (5 to 40%)
and different flow rates (700 to 1,100 L/h). At 20% excess oxygen and a flow rate of 860 L/h, the
amount of pyrene emitted ranged from 29.9 ng/kg at 700 °C to 402.9 ng/kg at 750 °C. The greatest
amount of PAHs emitted were observed at 750 °C (Mastral et al., 1999).
Drinking water standard: No MCLGs or MCLs have been proposed by the U.S. EPA (2000).
Environmental Fate
Biological. When pyrene was statically incubated in the dark at 25 °C with yeast extract and
settled domestic wastewater inoculum, complete degradation was demonstrated at the 5 mg/L
substrate concentration after 2 wk. At a substrate concentration of 10 mg/L, however, only 11 and
2% losses were observed after 7 and 14 d, respectively (Tabak et al., 1981).
Soil. The reported half-lives for pyrene in a Kidman sandy loam and McLaurin sandy loam are
260 and 199 d, respectively (Park et al., 1990).
Plant. Hückelhoven et al. (1997) studied the metabolism of pyrene by suspended plant cell
cultures of soybean, wheat, jimsonweed, and purple foxglove. Soluble metabolites were only
detected in foxglove and wheat. Approximately 90% of pyrene was transformed in wheat. In
foxglove, 1-hydroxypyrene methyl ether was identified as the main metabolite but in wheat, the
metabolites were identified as conjugates of 1-hydroxypyrene.
Photolytic. Adsorption onto garden soil for 10 d at 32 °C and irradiated with UV light produced
1,1′- bipyrene, 1,6-pyrenedione, 1,8-pyrenedione, and three unidentified compounds (Fatiadi,
1967). Microbial degradation by Mycobacterium sp. yielded the following ring-fission products:
4-phenanthroic acid, 4-hydroxyperinaphthenone, cinnamic acid, and phthalic acid. The
compounds pyrenol and the cis- and trans-4,5-dihydrodiols of pyrene were identified as
ring-oxidation products (Heitkamp et al., 1988).
Chemical/Physical. At room temperature, concentrated sulfuric acid will react with pyrene to
form a mixture of disulfonic acids. In addition, an atmosphere containing 10% sulfur dioxide
transformed pyrene into many sulfur compounds, including pyrene-1-sulfonic acid and
pyrenedisulfonic acid (Nielsen et al., 1983).
Shipping
UN3077 Environmentally hazardous substances, solid, n.o.s., Hazard class: 9; Labels: 9-Miscellaneous haz- ardous material, Technical Name Required.
Purification Methods
Crystallise pyrene from EtOH, glacial acetic acid, *benzene or toluene. Purify it also by chromatography of CCl4 solutions on alumina, with *benzene or n-hexane as eluent. [Backer & Whitten J Phys Chem 91 865 1987.] It can also be zone refined and purified by sublimation. Marvel and Anderson [J Am Chem Soc 76 5434 1954] refluxed pyrene (35g) in toluene (400mL) with maleic anhydride (5g) for 4days, then added 150mL of aqueous 5% KOH and refluxed for 5hours with occasional shaking. The toluene layer was separated, washed thoroughly with H2O, concentrated to about 100mL and allowed to cool. Crystalline pyrene was filtered off and recrystallised three times from EtOH or acetonitrile. [Chu & Thomas J Am Chem Soc 108 6270 1986, Russell et al. Anal Chem 50 2961 1986.] The material is free from anthracene derivatives. Another purification step involves passage of pyrene in cyclohexane through a column of silica gel. It can be sublimed in a vacuum and zone refined. The picrate has m 224o. [Kano et al. J Phys Chem 89 3748 1985, Beilstein 5 IV 2467.]
Incompatibilities
Pyrene Dust may form explosive mixture with air. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. Pyrene reacts with nitrogen oxides to form nitro derivatives. It also reacts with 70% nitric acid
Waste Disposal
Dissolve or mix the material with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber. All federal, state, and local environmental regulations must be observed .
Properties of Pyrene
| Melting point: | 148 °C |
| Boiling point: | 393 °C |
| Density | 1.271 |
| vapor pressure | 180 at 25 °C (extrapolated from vapor pressures determined at higher temperatures, Tesconi andYalkowsky, 1998) |
| refractive index | 1.8520 (estimate) |
| Flash point: | 210 °C |
| storage temp. | room temp |
| solubility | ethanol: soluble |
| form | crystalline |
| pka | >15 (Christensen et al., 1975) |
| color | Pale yellow to yellow-greenish |
| Specific Gravity | 1.271 |
| Water Solubility | almost insoluble |
| λmax | 330nm(EtOH)(lit.) |
| Merck | 14,7963 |
| BRN | 1307225 |
| Henry's Law Constant | 0.490 at 25 °C (thermodynamic method-GC/UV spectrophotometry, Altschuh et al., 1999) |
| Exposure limits | OSHA: TWA 0.2 mg/m3 |
| CAS DataBase Reference | 129-00-0(CAS DataBase Reference) |
| NIST Chemistry Reference | Pyrene(129-00-0) |
| IARC | 3 (Vol. Sup 7, 92) 2010 |
| EPA Substance Registry System | Pyrene (129-00-0) |
Safety information for Pyrene
| Signal word | Warning |
| Pictogram(s) |
 Environment GHS09 |
| GHS Hazard Statements |
H410:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P273:Avoid release to the environment. P391:Collect spillage. Hazardous to the aquatic environment P501:Dispose of contents/container to..… |
Computed Descriptors for Pyrene
| InChIKey | BBEAQIROQSPTKN-UHFFFAOYSA-N |
Pyrene manufacturer
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran
![Benzo[b]thien-2-ylboronic acid](https://img.chemicalbook.in/CAS/GIF/98437-23-1.gif)

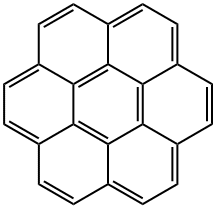
![Benzo[a]pyrene](https://img.chemicalbook.in/CAS/GIF/50-32-8.gif)
You may like
-
 Pyrene 95%, for synthesisView Details
Pyrene 95%, for synthesisView Details
129-00-0 -
 Pyrene CAS 129-00-0View Details
Pyrene CAS 129-00-0View Details
129-00-0 -
 Pyrene CAS 129-00-0View Details
Pyrene CAS 129-00-0View Details
129-00-0 -
 Pyrene, 98% CAS 129-00-0View Details
Pyrene, 98% CAS 129-00-0View Details
129-00-0 -
 Pyrene (purified by sublimation) CAS 129-00-0View Details
Pyrene (purified by sublimation) CAS 129-00-0View Details
129-00-0 -
 Pyrene 96% CAS 129-00-0View Details
Pyrene 96% CAS 129-00-0View Details
129-00-0 -
 Pyrene CAS 129-00-0View Details
Pyrene CAS 129-00-0View Details
129-00-0 -
 Pyrene CAS 129-00-0View Details
Pyrene CAS 129-00-0View Details
129-00-0
