POLYISOPRENE
Synonym(s):CD220 antigen;insulin receptor;IR;kinase InsR;Poly(1-methyl-1-butene-1,4-diyl)
- CAS NO.:9003-31-0
- Empirical Formula: C5H8
- Molecular Weight: 68.12
- MDL number: MFCD00084438
- EINECS: 618-362-9
- SAFETY DATA SHEET (SDS)
- Update Date: 2024-12-18 14:07:02
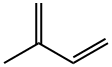
What is POLYISOPRENE?
Description
Polyisoprene is the polymer known as natural rubber, although it can also be manufactured. The natural rubber latex is harvested from the rubber tree, Hevea brasiliensis. This substance has a variety of natural additives, such as proteins and sugars. The polymer from the natural latex is resistant to many solvents and also is easily processed. The synthetic form of this rubber is produced from a pure isoprene solution with a stereospecific isomer to produce the more commonly used cis-l,4 isomer. These rubbers are resistant to abrasion and most solvents and are commercially used in automobile tires, adhesives, and a variety of products that come in close contact with the general public. Their use in baby bottle nipples is a good indication of the extremely low toxicity associated with these elastomers.
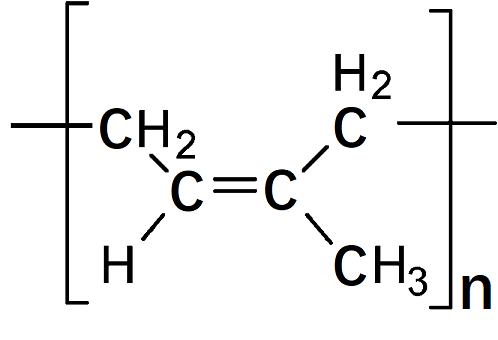
Chemical properties
There are two main solvents for rubber: turpentine and naptha (petroleum). Because rubber does not dissolve easily, the material is finely divided by shredding prior to immersion. Natural rubber has been partially replaced by synthetics, particularly styrene–butadiene, as a generalpurpose rubber. High resilience, low heat buildup, and easy processing are particular advantages of natural rubber when it is often used in blends with synthetic polyisoprene and other elastomers. Natural rubber, alone and in combination with neoprene, has been rated highly for resistance to water, dimethyl sulfoxide, and some alcohols in a comparative test of glove materials; resistance to other solvents varied from good to poor. Polyisoprene supports combustion.
Chemical properties
Natural rubber is the name applied to the polymer cis-1,4-polyisoprene obtained chiefly from the 4590 RUBBER, NATURAL latex of the Hevea brasiliensis tree.
The Uses of POLYISOPRENE
Natural rubber is a vital, strategic, and irreplaceable raw material used in enormous quantities by the commercial, medical, transportation, and defense industries. At least 40,000 different products and over 400 medical devices contain natural rubber.
Definition
The major component of natural rubber, also made synthetically. Forms are stereospecific cis-1,4and trans-1,4-polyisoprene.Both can be produced synthetically by the effect of heat and pressure on isoprene in the presence of stereospecific catalysts. Nat
Production Methods
The latex of natural rubber is obtained from trees (Hevea
brasiliensis); the actual monomer is isopentenyl pyrophosphate
that has been formed by biosynthesis. Natural rubber
contains low-molecular-weight impurities; small amounts of
sugar, fatty acids, proteins, and trace metals all play an
important part in processing.
Depending on the catalyst, rubber may undergo 1,2-; 3,4-;
or 1,4- additional polymerization that leads to several isomeric
structures.
Almost all commercial synthetic polyisoprenes are prepared
from purified isoprene monomer by a solution process.
A stereospecific catalyst, such as an Al–Ti Ziegler type, is
required for direct polymerization to the cis-1,4 isomer.
The production of the finished polymer requires two
separate manufacturing processes: (a) formation of the
rawpolymer and (b) conversion of the polymer to the finished
rubber product. The first step is similar to that of plastic
production. Large-scale operations use bulk materials in an
enclosed system.
General Description
Available as part of Negative Photoresist kit 654892
Industrial uses
Rubber is characterized as being a highly elastic or resilient material, and the natural product is obtained mainly as a latex from cuts in the trunks of the Hevea brasiliensis tree. The latex consists of small particles (averaging about 2500 ? units in diameter) of rubber suspended in an aqueous medium (at about 35% solids content). The system also contains about 6 to 8% nonrubber constituents, some of which are emulsifiers, naturally occurring antioxidants, and proteins.
Natural rubber is used for making many types of articles. Because of its abrasion-resistant quality and low hysteresis in reinforced compounds, it is used in truck-tire tread stocks and in conveyor belts that which are employed in conveying abrasive material such as coal, crushed rock, ore, and cinders. In large tires, it has found application in carcass compounds because of the tack and building qualities of the raw polymer. It has also been used in carcass compounds because of the low heat buildup (low hysteresis) of the carcass compound vulcanizate during severe service conditions in tire usage.
Properties of POLYISOPRENE
| Melting point: | 64 °C |
| Boiling point: | 122-142 °C(lit.) |
| Density | 0.92 g/mL at 25 °C |
| refractive index | n |
| Flash point: | >230 °F |
| storage temp. | 2-8°C |
| form | slab/chunk |
| CAS DataBase Reference | 9003-31-0 |
| EPA Substance Registry System | 1,3-Butadiene, 2-methyl-, homopolymer (9003-31-0) |
Safety information for POLYISOPRENE
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Exclamation Mark Irritant GHS07  Health Hazard GHS08 |
| GHS Hazard Statements |
H226:Flammable liquids H304:Aspiration hazard H315:Skin corrosion/irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation H412:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P331:Do NOT induce vomiting. P301+P310:IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician. P303+P361+P353:IF ON SKIN (or hair): Remove/Take off Immediately all contaminated clothing. Rinse SKIN with water/shower. |
Computed Descriptors for POLYISOPRENE
New Products
Tert-butyl bis(2-chloroethyl)carbamate (S)-3-Aminobutanenitrile hydrochloride N-Boc-D-alaninol N-BOC-D/L-ALANINOL N-octanoyl benzotriazole 4-Hydrazinobenzoic acid 3,4-Dibenzyloxybenzaldehyde 1,1’-CARBONYLDIIMIDAZOLE R-2-BENZYLOXY PROPIONIC ACID 1,1’-CARBONYLDI (1,2-4 TRIAZOLE) 4-HYDROXY BENZYL ALCOHOL 3-NITRO-2-METHYL ANILINE (2-Hydroxyphenyl)acetonitrile 4-Bromopyrazole 5-BROMO-2CYANO PYRIDINE 5,6-Dimethoxyindanone 5-broMo-2-chloro-N-cyclopentylpyriMidin-4-aMine 4-methoxy-3,5-dinitropyridine 2-(Cyanocyclohexyl)acetic acid 2-aminopropyl benzoate hydrochloride 1-(4-(aminomethyl)benzyl)urea hydrochloride tert-butyl 4- (ureidomethyl)benzylcarbamate diethyl 2-(2-((tertbutoxycarbonyl)amino) ethyl)malonate Ethyl-2-chloro((4-methoxyphenyl)hydrazono)acetateRelated products of tetrahydrofuran






You may like
-
 Negative photoresist I CAS 9003-31-0View Details
Negative photoresist I CAS 9003-31-0View Details
9003-31-0 -
 55441-95-7 99%View Details
55441-95-7 99%View Details
55441-95-7 -
 N-Vinylformamide 99%View Details
N-Vinylformamide 99%View Details
13162-05-5 -
 Chloro Uracil 1820-81-1 99%View Details
Chloro Uracil 1820-81-1 99%View Details
1820-81-1 -
 207557-35-5 99%View Details
207557-35-5 99%View Details
207557-35-5 -
 2-ethyl-6-methyl-3-hydroxypyridine succinate 99%View Details
2-ethyl-6-methyl-3-hydroxypyridine succinate 99%View Details
127464-43-1 -
 2-ETHYLPYRIDINE 100-71-0 99%View Details
2-ETHYLPYRIDINE 100-71-0 99%View Details
100-71-0 -
 181228-33-1 (S)-Methyl 3-amino-2-((tert-butoxycarbonyl)amino)propanote Hydrochloride (DAP-OMe. HCl) 99%View Details
181228-33-1 (S)-Methyl 3-amino-2-((tert-butoxycarbonyl)amino)propanote Hydrochloride (DAP-OMe. HCl) 99%View Details
181228-33-1