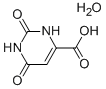
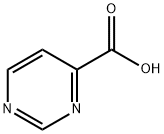
Orotic acid
Synonym(s):2,6-Dihydroxypyrimidine-4-carboxylic acid;6-Carboxy-2,4-dihydroxypyrimidine;6-Carboxyuracil;Uracil-6-carboxylic acid
- CAS NO.:65-86-1
- Empirical Formula: C5H4N2O4
- Molecular Weight: 156.1
- MDL number: MFCD00006027
- EINECS: 200-619-8
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-19 17:28:17

What is Orotic acid?
Chemical properties
white crystalline powder
Originator
Lactinium, Roland
The Uses of Orotic acid
Orotic acid is an intermediate in de novo pyrimidine biosynthesis that may be used to study the specificity and kinetics of orotate phosphoribosyltransferase (OPRT) which catalyzes the reversible phosphoribosyl transfer from 5?-phospho-α-d-ribose 1?-diphosphate (PRPP) to orotic acid (OA), forming pyrophosphate and orotidine 5?-monophosphate (OMP). It is used as a starting material for the potential commercial bioproduction of uridine 5?-monophosphate (UMP) by microbes such as Corynebacterium ammoniagenes (ATCC 6872) or Saccharomyces cerevisiae. It may be used to study the AMPK/SREBP-1 dependent cell signaling pathway and transcription regulation mechanisms that induce hepatic lipogenesis.
The Uses of Orotic acid
hepatoprotectant, uricosuric agent
The Uses of Orotic acid
An intermediate in de novo pyrimidine biosynthesis.
What are the applications of Application
Orotic acid is an intermediate in de novo pyrimidine biosynthesis
Definition
ChEBI: A pyrimidinemonocarboxylic acid that is uracil bearing a carboxy substituent at position C-6.
Manufacturing Process
91.7 g (0.5 mol) of trichloroacetyl chloride was cooled to -35°C in a glass vessel by means of a cooling brine. In the course of 3 h, 27.0 g (0.06 mol) of pure ketene was introduced through a tube. After completion of the reaction, the vessel was immediately put under dry nitrogen to prevent penetration of moisture. So γ,γ,γ-trichloroacetoacetylchloride was produced.
The reaction mixture containing the γ,γ,γ-trichloroacetoacetyl chloride was transfered under nitrogen to a dropping funnel and in the course of 15 min was added with vigorous agitation to a suspension of 69.0 g (1.15 mole) of urea in 90.0 g of anhydrous acetic acid. Water cooling was used so that the reaction temperature would not exceed 40°C. After completion of the addition, the reaction mixture was heated as rapidly as possible to 115°C, and held at
this temperature for 30 min.
Subsequently there was cooling and one more 99.0 g of glacial acetic acid and 180.0 g of water were added. The precipitated 6-trichloromethyluracil was filtered off and dried at 60°C in a vacuum drying cabinet. The yield was 91.0 g or 80%.
In a glass vessel equipped with an agitator, thermometer and pH electrode, 500 ml of water was placed and heated to 80°C. 50 g of 6trichloromethyluracil was then added. By means of the pH electrode, the addition of sodium hydroxide was automatically controlled so that the pH value throughout the whole hydrolysis was 6.5. Into, 165 ml of 5 N NaOH was consumed. Finally, the hydrolysis solution was cooled and the precipitated sodium orotate filtered off.
The crude sodium orotate was again suspended at 80°C in water and brought into solution (pH 10.5) by addition of 30 ml of 5 N NaOH. After treatment with active charcoal, the solution was acidified with 30.0 g of 50% sulfuric acid. The solution was then cooled. The orotic acid was filtered off and carefully washed with water. After drying, 20.5 g of orotic acid, having a purity of 99.3% (titration) was obtained. This corresponds to a 60% yield.
Therapeutic Function
Hepatoprotectant
Synthesis Reference(s)
Journal of the American Chemical Society, 69, p. 1382, 1947 DOI: 10.1021/ja01198a042
General Description
White crystals or crystalline powder.
Air & Water Reactions
Slightly soluble in water.
Reactivity Profile
Carboxylic acids, such as Orotic acid, donate hydrogen ions if a base is present to accept them. They react in this way with all bases, both organic (for example, the amines) and inorganic. Their reactions with bases, called "neutralizations", are accompanied by the evolution of substantial amounts of heat. Neutralization between an acid and a base produces water plus a salt. Carboxylic acids with six or fewer carbon atoms are freely or moderately soluble in water; those with more than six carbons are slightly soluble in water. Soluble carboxylic acid dissociate to an extent in water to yield hydrogen ions. The pH of solutions of carboxylic acids is therefore less than 7.0. Many insoluble carboxylic acids react rapidly with aqueous solutions containing a chemical base and dissolve as the neutralization generates a soluble salt. Carboxylic acids in aqueous solution and liquid or molten carboxylic acids can react with active metals to form gaseous hydrogen and a metal salt. Such reactions occur in principle for solid carboxylic acids as well, but are slow if the solid acid remains dry. Even "insoluble" carboxylic acids may absorb enough water from the air and dissolve sufficiently in Orotic acid to corrode or dissolve iron, steel, and aluminum parts and containers. Carboxylic acids, like other acids, react with cyanide salts to generate gaseous hydrogen cyanide. The reaction is slower for dry, solid carboxylic acids. Insoluble carboxylic acids react with solutions of cyanides to cause the release of gaseous hydrogen cyanide. Flammable and/or toxic gases and heat are generated by the reaction of carboxylic acids with diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides. Carboxylic acids, especially in aqueous solution, also react with sulfites, nitrites, thiosulfates (to give H2S and SO3), dithionites (SO2), to generate flammable and/or toxic gases and heat. Their reaction with carbonates and bicarbonates generates a harmless gas (carbon dioxide) but still heat. Like other organic compounds, carboxylic acids can be oxidized by strong oxidizing agents and reduced by strong reducing agents. These reactions generate heat. A wide variety of products is possible. Like other acids, carboxylic acids may initiate polymerization reactions; like other acids, they often catalyze (increase the rate of) chemical reactions.
Fire Hazard
Flash point data for Orotic acid are not available; however, Orotic acid is probably combustible.
Safety Profile
Moderately toxic by ingestion, intraperitoneal, and intravenous routes. Mutation data reported. Whenheated to decomposition it emits toxic fumes of NOx.
Properties of Orotic acid
| Melting point: | ≥300 °C |
| Boiling point: | 280.29°C (rough estimate) |
| Density | 1.6814 (rough estimate) |
| refractive index | 1.4800 (estimate) |
| storage temp. | Sealed in dry,Room Temperature |
| solubility | DMSO : 32 mg/mL (205.00 mM; Need ultrasonic and warming) |
| form | Crystalline Powder |
| pka | pK1:1.8(+1);pK2:9.55(0) (25°C) |
| color | White |
| Water Solubility | Slightly soluble |
| Merck | 13,6942 |
| BRN | 383901 |
| Stability: | Stable. Incompatible with strong oxidizing agents. |
| CAS DataBase Reference | 65-86-1(CAS DataBase Reference) |
| NIST Chemistry Reference | Orotic acid(65-86-1) |
| EPA Substance Registry System | Orotic acid (65-86-1) |
Safety information for Orotic acid
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H302:Acute toxicity,oral H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P302+P352:IF ON SKIN: wash with plenty of soap and water. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for Orotic acid
| InChIKey | PXQPEWDEAKTCGB-UHFFFAOYSA-N |
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran

![FLUORO OROTIC ACID, 5-, [2-14C]](https://img.chemicalbook.in/StructureFile/ChemBookStructure2/GIF/CB5763355.gif)
You may like
-
 65-86-1 Orotic acid 98%View Details
65-86-1 Orotic acid 98%View Details
65-86-1 -
 Orotic acid, ≥98% CAS 65-86-1View Details
Orotic acid, ≥98% CAS 65-86-1View Details
65-86-1 -
 Orotic acid CAS 65-86-1View Details
Orotic acid CAS 65-86-1View Details
65-86-1 -
 Orotic acid CAS 65-86-1View Details
Orotic acid CAS 65-86-1View Details
65-86-1 -
 3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details
3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details -
 20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 -
 3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details
3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details -
 57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4
