Methyl formate
Synonym(s):Formic acid methyl ester;Methyl formate
- CAS NO.:107-31-3
- Empirical Formula: C2H4O2
- Molecular Weight: 60.05
- MDL number: MFCD00003291
- EINECS: 203-481-7
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-04-14 18:24:50
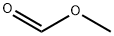
What is Methyl formate?
Description
Methyl Formate, also known as methyl methanoate, is the methyl ester of formic acid. The simplest example of an ester, it is a clear liquid with an ethereal odour, high vapor pressure, and low surface tension. It is an aromatic compound found in apples (Neubeller and Buchloh, 1986), and was identified as a volatile constituent in brewed, roasted, and dried coffee (Lovell et al., 1980); Methyl formate is used primarily to manufacture formamide, dimethylformamide, and formic acid. It is also used as a solvent for quick-drying finishes such as lacquers and in organic synthesis.
Description
Methyl formate, a clear, volatile liquid, is the simplest carboxylate ester. It can be made in the lab by the acid-catalyzed esterification of formic acid and methanol. Industrially, it is produced by the base-catalyzed reaction of methanol and carbon monoxide.
The industrial uses of methyl formate include the manufacture of other formic acid derivatives, as a blowing agent for foams, and as an agricultural fumigant. It was formerly used as a refrigerant as an alternative to sulfur dioxide even though it also poses a toxicity hazard.
Methyl formate has been used as a model ester to study the combustion mechanisms of much more complex biodiesel mixtures. Toward this end, E. A. Carter and co-workers at Princeton University recently performed calculations to predict the hydrogen abstraction kinetics of methyl formate. They found that the predicted rate constants differed significantly from those that were calculated in previous combustion kinetics models.
?
Chemical properties
Methyl formate is a colorless liquid with a pleasant odor. Its solubility in water is 230 g/l at 25 °C (Riddick et al., 1985), but it reacts slowly with water to form formic acid and methyl alcohol (DOT, 1984). It is soluble in ether, chloroform, and is miscible with ethanol (Lide, 2000).
Physical properties
Clear, colorless, mobile liquid with a pleasant, etheral odor. An odor threshold concentration of 130 ppmv was reported by Nagata and Takeuchi (1990).
The Uses of Methyl formate
Methyl formate is used as a fumigant, as alarvicide for food crops, and as a solvent forcellulose acetate.
The Uses of Methyl formate
Methyl formate is used primarily to manufacture formamide, dimethyl formamide, and formic acid. Because of its high vapor pressure, it is used for quick - drying finishes. It is also used as an insecticide and to manufacture certain pharmaceuticals. Foam Supplies, Inc. has trademarked Ecomate, which is used as a blowing agent for foam insulation, as a replacement for CFC, HCFC, or HFCs, with zero ozone depletion potential and < 25 global warming potential.
A historical use of methyl formate, which sometimes brings it attention, was in refrigeration. Before the introduction of less-toxic refrigerants, methyl formate was used as an alternative to sulfur dioxide in domestic refrigerators, such as some models of the famous GE Monitor Top. Owners of methyl formate refrigerators should keep in mind that, even though they operate below atmospheric pressure, if evidence of a leak develops, they should take measures to avoid exposure to the ether-smelling liquid and vapor.
Production Methods
In the laboratory, methyl formate can be produced by the condensation reaction of methanol and formic acid, as follows:
HCOOH + CH3OH → HCOOCH3 + H2O
Industrial methyl formate, however, is usually produced by the combination of methanol and carbon monoxide (carbonylation) in the presence of a strong base, such as sodium methoxide :
CH3OH + CO → HCOOCH3
This process, practiced commercially by BASF among other companies gives 96 % selectivity toward methyl formate, although it can suffer from catalyst sensitivity to water, which can be present in the carbon monoxide feedstock, commonly derived from synthesis gas. Very dry carbon monoxide is, therefore, an essential requirement.
Definition
ChEBI: Methyl formate is a formate ester resulting from the formal condensation of formic acid with methanol. A low-boiling (31.5 ℃) colourless, flammable liquid, it has been used as a fumigant and larvicide for tobacco and food crops. It has a role as a polar aprotic solvent, a fumigant, an insecticide and a refrigerant. It is a formate ester, a methyl ester and a volatile organic compound. It is functionally related to a methanol.
General Description
A clear colorless liquid with an agreeable odor. Flash point -27°F. Less dense than water Vapors heavier than air.
Air & Water Reactions
Highly flammable. Water soluble. Reacts slowly with water to give formic acid, a corrosive material, and methanol, a flammable liquid. Both products are dissolved in the water.
Reactivity Profile
Methyl formate reacts with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated with caustic solutions. Flammable hydrogen is generated by mixing with alkali metals and hydrides.
Hazard
Flammable, dangerous fire and explosionrisk, explosive limits in air 5.9–20%. Eye, upperand lower respiratory tract irritant.
Health Hazard
Inhalation causes irritation of mucous membranes. Prolonged inhalation can produce narcosis and central nervous symptoms, including some temporary visual disturbance. Contact with liquid irritates eyes and may irritate skin if allowed to remain. Ingestion causes irritation of mouth and stomach and central nervous system depression, including visual disturbances.
Health Hazard
Methyl formate is a moderately toxic com pound affecting eyes, respiratory tract, andcentral nervous system. It is an irritant tothe eyes, nose, and lungs. Exposure to highconcentrations of its vapors in air may pro duce visual disturbances, irritations, narcoticeffects, and respiratory distress in humans.Such effects may be manifested at a 1-hourexposure to about 10,000-ppm concentration.Cats died of pulmonary edema from 2-hourexposure to this concentration
The acute oral toxicity of methyl formatewas low in test subjects. The symptoms werenarcosis, visual disturbances, and dyspnea.An oral LD50 value in rabbit is in the range1600 mg/kg..
Fire Hazard
Behavior in Fire: Vapor is heavier than air and may travel considerable distance to a source of ignition and flash back.
Safety Profile
Moderately toxic by ingestion. Inhalation of vapor can cause irritation to nasal passages and conjunctiva, optic neuritis, narcosis, retching, and death from pulmonary irritation. Industrial fatalities have occurred only with exposure to high concentrations. Flammable liquid. Very dangerous fire hazard when exposed to heat or flame; can react vigorously with oxidizing materials. Explosive in the form of vapor when exposed to heat or flame. Reacts with methanol + sodium methoxide to form an explosive product. To fight fire, use alcohol foam, CO2, dry chemical. When heated to decomposition it emits acrid smoke and irritating fumes.
Potential Exposure
Methyl formate is used as a solvent; as an intermediate in pharmaceutical manufacture; and as a fumigant
First aid
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifing upper and lower lids. Seek med-facility. When this chemical has been swallowed, get medi-cal attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit.Medical observation is recommended for 24- -48 h afterbreathing overexposure, as pulmonaryedema may bedelayed. As first aid for pulmonary edema, a doctor orauthorized paramedic may consider administering a cortico-steroid spray.
Environmental Fate
Photolytic. Methyl formate, formed from the irradiation of dimethyl ether in the presence of
chlorine, degraded to carbon dioxide, water, and small amounts of formic acid. Continued
irradiation degraded formic acid to carbon dioxide, water, and hydrogen chloride (Kallos and Tou,
1977; Good et al., 1999).
A rate constant of 2.27 x 10-12 cm3/molecule?sec was reported for the reaction of methyl formate
and OH radicals in the atmosphere (Atkinson, 1989).
Chemical/Physical. Hydrolyzes slowly in water forming methanol and formic acid (NIOSH,
1997). Hydrolysis half-lives reported at 25 °C: 0.91 h at pH 9, 9.1 h at pH 8, 2.19 d at pH 7, and
21.9 d at pH 6 (Mabey and Mill, 1978).
Storage
Color Code—Red: Flammability Hazard: Store ina flammable liquid storage area or approved cabinet awayfrom ignition sources and corrosive and reactive materials.Prior to working with this chemical you should be trainedon its proper handling and storage. Before entering confinedspace where this chemical may be present, check to makesure that an explosive concentration does not exist. Methylformate must be stored to avoid contact with strong oxidizers, such as chlorine, bromine, chlorine dioxide, nitrates,and permanganates, since violent reactions occur. Store intightly closed containers in a cool, well-ventilated areaaway from heat. Sources of ignition, such as smoking andopen flames, are prohibited where methyl formate is handled, used, or stored. Metal containers involving the transferof 5 gallons or more of methyl formate should be groundedand bonded. Drums must be equipped with self-closingvalves, pressure vacuum bungs, and flame arresters. Useonly nonsparking tools and equipment, especially whenopening and closing containers of methyl formate.Wherever methyl formate is used, handled, manufactured,or stored, use explosion-proof electrical equipment andfittings.
Shipping
This compound requires a shipping label of“FLAMMABLE LIQUID.” It falls in Hazard Class 3 andPacking Group I.
Purification Methods
Wash the formate with strong aqueous Na2CO3, dry it with solid Na2CO3 and distil it from P2O5. (Procedure removes free alcohol or acid.) [Beilstein 2 IV 20.]
Incompatibilities
May form explosive mixture with air. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases, strong acids, oxoacids, epoxides. Reacts slowly with water to form methanol and formic acid. Contact with water, steam releases formic acid. Compounds of the carboxyl group react with all bases, both inorganic and organic (i.e., amines) releasing substantial heat, water and a salt that may be harmful. Incompatible with arsenic compounds (releases hydrogen cyanide gas), diazo compounds, dithiocarbamates isocyanates, mercaptans, nitrides, and sulfides (releasing heat, toxic, and possibly flammable gases), thiosulfates and dithionites (releasing hydrogen sulfate and oxides of sulfur)
Waste Disposal
Incineration; atomizing in a suitable combustion chamber.
References
Lee, Jae S., J. C. Kim, and Y. G. Kim. "Methyl formate as a new building block in C1 chemistry." Applied Catalysis 57.1 (1990): 1-30.
Handa, Yash Paul, et al. "Insulating Thermoplastic Foams Made With Methyl Formate-Based Blowing Agents." (2006).
Properties of Methyl formate
| Melting point: | -100 °C (lit.) |
| Boiling point: | 32-34 °C (lit.) |
| Density | 0.974 g/mL at 20 °C (lit.) |
| vapor density | 2.1 (vs air) |
| vapor pressure | 32.91 psi ( 55 °C) |
| refractive index | n |
| Flash point: | −16 °F |
| storage temp. | Store below +30°C. |
| solubility | 300g/l |
| form | Liquid |
| color | Clear colorless |
| Odor | Pleasant; agreeable. |
| PH | 4-5 (200g/l, H2O, 20℃) |
| explosive limit | 5-23%(V) |
| Odor Threshold | 130ppm |
| Water Solubility | 300 G/L (20 ºC) |
| λmax | λ: 259 nm Amax: 1.00 λ: 260 nm Amax: 0.70 λ: 265 nm Amax: 0.20 λ: 270 nm Amax: 0.04 λ: 310-400 nm Amax: 0.01 |
| Merck | 14,6077 |
| BRN | 1734623 |
| Henry's Law Constant | 0.90 at 5.00 °C, 1.18 at 10.00 °C, 1.51 at 15.00 °C, 1.91 at 20.00 °C, 2.36 at 25.00 °C (column
stripping-UV, Kutsuna et al., 2005) |
| Exposure limits | TLV-TWA 100 ppm (~250 mg/m3) (ACGIH,
MSHA, and OSHA); TLV-STEL 150 ppm
(~375 mg/m3) (ACGIH); IDLH 5000 ppm
(NIOSH). |
| Dielectric constant | 8.5(20℃) |
| Stability: | Stable. Extremely flammable. Readily forms explosive mixtures with air. Note low flash point and very wide explosion limits. Incompatible with oxidizing agents. |
| CAS DataBase Reference | 107-31-3(CAS DataBase Reference) |
| NIST Chemistry Reference | Methyl formate(107-31-3) |
| EPA Substance Registry System | Methyl formate (107-31-3) |
Safety information for Methyl formate
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Skull and Crossbones Acute Toxicity GHS06  Health Hazard GHS08 |
| GHS Hazard Statements |
H224:Flammable liquids H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation H370:Specific target organ toxicity, single exposure |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P280:Wear protective gloves/protective clothing/eye protection/face protection. P403+P233:Store in a well-ventilated place. Keep container tightly closed. |
Computed Descriptors for Methyl formate
Methyl formate manufacturer
JSK Chemicals
RAIGAD CHEMICALS PVT LTD
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran

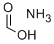
You may like
-
 107-31-3 98%View Details
107-31-3 98%View Details
107-31-3 -
 Methyl formate 107-31-3 98%View Details
Methyl formate 107-31-3 98%View Details
107-31-3 -
 Methyl formate CAS 107-31-3View Details
Methyl formate CAS 107-31-3View Details
107-31-3 -
 Methyl Formate pure CAS 107-31-3View Details
Methyl Formate pure CAS 107-31-3View Details
107-31-3 -
 Methyl formate, 97% CAS 107-31-3View Details
Methyl formate, 97% CAS 107-31-3View Details
107-31-3 -
 Methyl formate 97% For Synthesis CAS 107-31-3View Details
Methyl formate 97% For Synthesis CAS 107-31-3View Details
107-31-3 -
 Liquid Methyl Formate CAS Number 107-31-3View Details
Liquid Methyl Formate CAS Number 107-31-3View Details
107-31-3 -
 Liquid Methyl Formate - MF / CAS No.107-31-3, Grade Standard: Technical GradeView Details
Liquid Methyl Formate - MF / CAS No.107-31-3, Grade Standard: Technical GradeView Details
107-31-3
