L-Tetrahydrofolic Acid
Synonym(s):5,6,7,8-Tetrahydropteroyl-L -glutamic acid;THF
- CAS NO.:135-16-0
- Empirical Formula: C19H23N7O6
- Molecular Weight: 445.43
- MDL number: MFCD00135583
- EINECS: 205-181-1
- SAFETY DATA SHEET (SDS)
- Update Date: 2025-12-23 13:58:55
What is L-Tetrahydrofolic Acid?
Chemical properties
Light Tan Solid
The Uses of L-Tetrahydrofolic Acid
Asa folic acid derivative and coenzyme, L-Tetrahydrofolic Acid can be involved in the metabolism of amino and nucleic acids.
The Uses of L-Tetrahydrofolic Acid
L-Tetrahydrofolic Acid is a folic acid derivative and coenzyme involved in the metabolism of amino and nucleic acids. We have observed that this material decomposes steadily over time. Use immediately upon receipt.
The Uses of L-Tetrahydrofolic Acid
Tetrahydrofolic acid has been used to determine the inhibitory effect of tetrahydrofolate on polymorphonuclear myeloid-derived suppressor cells (PMN-MDSCs). A recent use of tetrahydrofolate (THF) is for studying the activation of riboswitches.
Definition
ChEBI: A derivative of folic acid in which the pteridine ring is fully reduced; it is the parent compound of a variety of coenzymes that serve as carriers of one-carbon groups in metabolic reactions.
Flammability and Explosibility
Not classified
Biochem/physiol Actions
Tetrahydrofolic acid is the parent molecule of the folate derivatives that donate one-carbon units to amino acids, nucleic acids, and lipids. Tetrahydrofolate metabolites participate in the synthesis of purine and pyrimidine and in synthesis and conversion of amino acids
Purification Methods
Very high quality material is now available commercially, and it should be a white powder. It can be dried over P2O5 in a vacuum desiccator and stored in weighed aliquots in sealed ampoules. It is stable at room temperature in sealed ampoules for many months and for much more extended periods at -10o. When moist, it is extremely sensitive to air whereby it oxidises to the yellow 7,8-dihydro derivative. In solution it turns yellow in colour as it oxidises, and then particularly in the presence of acids it turns dark reddish brown in colour. Hence aqueous solutions should be frozen immediately when not in use. It is always advisable to add 2-mercaptoethanol (if it does not interfere with the procedure for which it is used) which stabilises it by depleting the solution of O2. The sulfate salt is more stable but is much less soluble. The best way to prepare standard solutions of this acid is to dissolve it in the desired buffer and estimate the concentration by UV absorption in pH 7 buffer at 297nm ( 22,000 M-1cm-1). If a sample is suspect, it is not advisable to purify it because it is likely to deteriorate further as “dry box” conditions are necessary. Either a new sample is purchased or one is freshly prepared from folic acid. It has the above pKa values. [Hafeti et al. Biochemical Preparations 7 89 1960, UV: Mathews & Huennekens J Biol Chem 235 3304 1960, Osborn & Huennekens J Biol Chem 233 969 1958, O'Dell et al. J Am Chem Soc 69 250 1947, Blakley Biochem J 6 5 331 1957, Asahi Yakugaku Zasshi (J Pharm Soc Jpn) 79 1548 1959, Beilstein 26 III/IV 3879.]
Properties of L-Tetrahydrofolic Acid
| Melting point: | >162°C (dec.) |
| Boiling point: | 555.12°C (rough estimate) |
| Density | 1.4216 (rough estimate) |
| vapor pressure | 0Pa at 25℃ |
| refractive index | 1.6800 (estimate) |
| storage temp. | -20°C |
| solubility | Aqueous Base (Slightly), DMSO (Slightly), Methanol (Slightly, Heated, Sonicated) |
| form | powder |
| pka | 3.51±0.10(Predicted) |
| color | off-white to tan |
| BRN | 9238187 |
| Stability: | We have observed that this material decomposes steadily over time. Use immediately upon receipt. |
Safety information for L-Tetrahydrofolic Acid
| Signal word | Warning |
| Pictogram(s) |
 Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H315:Skin corrosion/irritation H319:Serious eye damage/eye irritation H335:Specific target organ toxicity, single exposure;Respiratory tract irritation |
| Precautionary Statement Codes |
P261:Avoid breathing dust/fume/gas/mist/vapours/spray. P271:Use only outdoors or in a well-ventilated area. P280:Wear protective gloves/protective clothing/eye protection/face protection. |
Computed Descriptors for L-Tetrahydrofolic Acid
New Products
4,4-Difluoropiperidine hydrochloride tert-butyl 9-methoxy-3-azaspiro[5.5]undecane-3-carboxylate Indole Methyl Resin N-Isopropylurea N,N-Dicyclohexylcarbodiimide(DCC) MELDRUMS ACID 5-METHYLISOXAZOLE-4-CARBOXYLIC ACID Magnessium Bis glycinate Zinc ascorbate 1-bromo-2-butyne 2-acetamidophenol 9(10H)-anthracenone Erythrosin B, 4-Piperidinopiperidine 2-((4-morpholinophenylamino) (methylthio) methylene) malononitrile 2,4-dihydroxybenzaldehyde 3-(4-morpholinophenylamino)-5-amino-1H-pyrazole-4-carbonitrile Methyl 2-methylquinoline-6-carboxylate 2,6-dichloro-4-nitropyridine 4-Bromo-2-chlorobenzonitrile 2-(benzylamino)acetic acid hydrochloride 4-(tert-Butoxycarbonylamino)but- 2-ynoic acid 3,4-dihydro-2H-benzo[b][1,4]dioxepine 1-Phenyl-1-cycloprppanecarboxylicacidRelated products of tetrahydrofuran
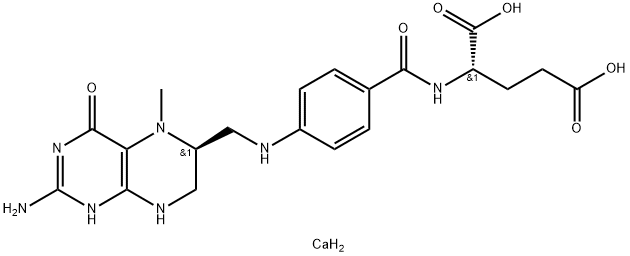
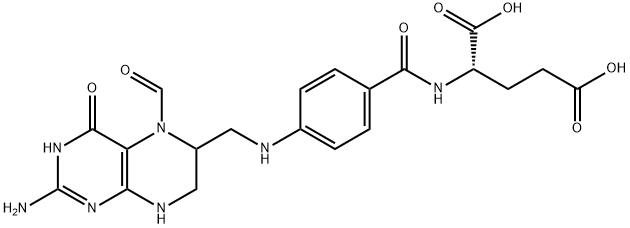
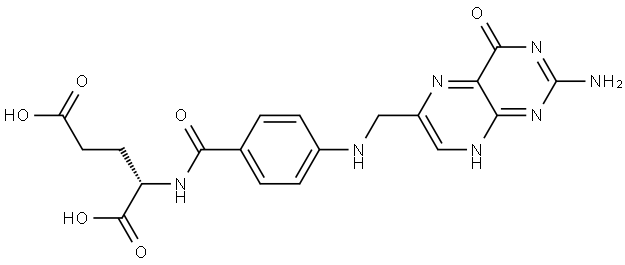
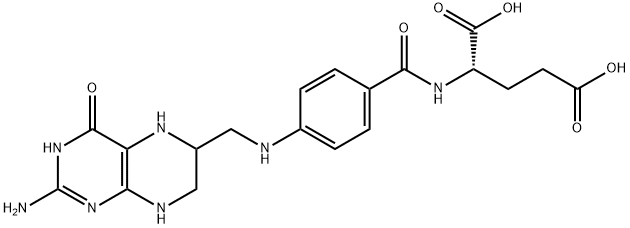
![(2S)-2-[[4-[Bis[(2-aMino-4-oxo-1,4-dihydropteridin-6-yl)Methyl]aMino]benzoyl]aMino]pentanedioic Acid](https://img.chemicalbook.in/CAS/20130318/GIF/1391068-26-0.gif)
You may like
-
 Tetrahydrofolic acid 95% CAS 135-16-0View Details
Tetrahydrofolic acid 95% CAS 135-16-0View Details
135-16-0 -
 Tetrahydrofolic acid CAS 135-16-0View Details
Tetrahydrofolic acid CAS 135-16-0View Details
135-16-0 -
 3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details
3-(4-amino-1-oxoisoindolin-2-yl)-1-methylpiperidine-2,6-dione 98%View Details -
 614-19-7 98%View Details
614-19-7 98%View Details
614-19-7 -
 20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 (2,2-diethoxyethyl)methylamine 98%View Details
20677-73-0 -
 3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details
3-(4-(hydroxyamino)-1-oxoisoindolin-2-yl)piperidine-2,6-dione 98%View Details -
 57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 2-bromo-4-chlorobenzonitrile 98%View Details
57381-49-4 -
 4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
4,6-dichloropyrimidine-5-carbaldehyde 98%View Details
5305-40-8
