ISOFENPHOS
Synonym(s):2-(Ethoxy-isopropylamino-thiophosphoryloxy)benzoic acid isopropyl ester
- CAS NO.:25311-71-1
- Empirical Formula: C15H24NO4PS
- Molecular Weight: 345.39
- MDL number: MFCD00041805
- EINECS: 246-814-1
- SAFETY DATA SHEET (SDS)
- Update Date: 2023-05-15 10:43:35
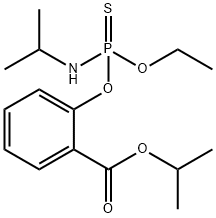
What is ISOFENPHOS?
Chemical properties
Isofenphos is a colorless oil at room temperature. It is sparingly soluble in water, but soluble in cyclohexone, toluene, acetone, and diethyl ether. The US EPA has grouped isofenphos under RUP, which indicates that qualifi ed, certifi ed, and trained workers are required in the safety management of isofenphos. It is used on turf and ornamental trees and shrubs to control white grubs, mole crickets, and other insects, such as soil-dwelling insects, cabbage root fl ies, corn roundworms, and wire worms.
The Uses of ISOFENPHOS
Isofenphos is an organophosphorus insecticide used in soil to control leaf-eating and soil-dwelling pests in vegetables, fruits, turf and field crops.
The Uses of ISOFENPHOS
Insecticide.
The Uses of ISOFENPHOS
Isofenphos is used as a systemic insecticide to control cabbage root flies, carrot flies, onion flies and other soil insects in vegetables, rape, maize, bananas, sugar cane and other crops.
Definition
ChEBI: Isofenphos is an organothiophosphate insecticide, an organic phosphonate, a phosphonic ester, a member of salicylates and an isopropyl ester. It has a role as an EC 3.1.1.7 (acetylcholinesterase) inhibitor and an agrochemical. It is functionally related to an isopropyl salicylate.
General Description
Colorless oil. Non corrosive. Used as an insecticide.
Air & Water Reactions
Hydrolyzed by alkali solution.
Reactivity Profile
Organothiophosphates, such as ISOFENPHOS, are susceptible to formation of highly toxic and flammable phosphine gas in the presence of strong reducing agents such as hydrides. Partial oxidation by oxidizing agents may result in the release of toxic phosphorus oxides.
Health Hazard
Exposures to isofenphos are highly toxic to animals and humans. Acute and prolonged exposures to high concentrations of isofenphos has caused poisoning with symptoms such as increased secretions, diffi culty in breathing, diarrhea, urination, pupil contraction, slowness of the heart, convulsions, and coma. Isofenphos in combination with malathion cause severe poisoning in humans.
Environmental Fate
Soil. Rapidly degraded by microbes via oxidative desulfuration in soils forming isofenphos
oxon (Abou-Assaf et al., 1986; Abou-Assaf and Coats, 1987; Somasundarum et al.,
1989), isopropyl salicylate and carbon dioxide (Somasundaram et al., 1989). The formation
of isofenphos oxon is largely dependent upon the pH, moisture and temperature of the
soil. The degradation rate of isofenphos decreased with a decrease in temperature (35°C
>25°C >15°C), moisture content (22.5% >30% >15%) and in acidic and alkaline soils
(pH 6 and 8 >pH 7). After isofenphos was applied to soil at a rate of 1,12 kg/ai/ha, concentrations
of 8.3, 7.2, 5.1 and 1.0 ppm were found after 5, 21, 43 and 69 days, respectively.
Following a second application, 4.9, 1.55, 0.25 and 0.10 ppm of isofenphos were found
after 5, 21, 43 and 69 days, respectively (Abou-Assaf and Coats, 1987).
A pure culture of Arthrobacter sp. was capable of degrading isofenphos at different
soil concentrations (10, 50 and 100 ppm) in less than 6 hours. In previously treated soils,isofenphos could be mineralized to carbon dioxide by indigenous microorganisms (
Plant. Two weeks following application to thatch, 65% of the applied amount was
present (Sears et al., 1987).
Surface Water. In estuarine water, the half-life of isofenphos ranged from 9.8 to 11.9
days (Lacorte et al., 1995).
Photolytic. Irradiation of an isofenphos (500 mg) in hexane and methanol (100 mL)
using a high pressure mercury lamp (l = 254–360 nm) for 24 hours yielded the following
products: isofenphos oxon and O-ethyl hydrogen-N-isopropylphosphoroamidothioa
Metabolic pathway
14C-Isofenphos is bioactivated by mixed-function oxidases that ultimately give N-desisopropylisofenphos oxon, a product with 2300-fold greater inhibitory potency than isofenphos oxon towards housefly head acetylcholinesterase.271 By housefly and the cuperous chafer, isofenphos is metabolized to give detoxified metabolites without the bioactivated N- desisopropylisofenphos oxon. No difference in kinds of metabolite is found between the two insects. When rats are administered 14C-isofenphos, most of the radioactivity is recovered from the water fraction of the urine and feces. Aminoisofenphos and isofenphos oxon are detected in the benzene fraction, and the other six metabolites are identified as water-soluble metabolites which include O-ethylhydrogen phosphoramidate and O-ethylhydrogen isopropylphosphoramidothioate. Water-soluble metabolites are predominantly formed through cleavage of the P-O-aryl linkage.
Metabolism
Main degradation route is cleavage of the P?O-aryl ester linkage through oxidative desulfuration to isofenphos oxon followed by hydrolysis and oxidative dearylation from isofenphos. In plant, the major metabolites are salicylic acid and dihydroxybenzoic acid.
Degradation
Isofenphos is stable to hydrolysis with a DT50 of >1 year at pH values from 4 to 9. Photodegradation on soil surfaces in the laboratory was extremely rapid but under natural light conditions photolysis was slower (PM).
Toxicity evaluation
The acute oral LD50 for rats is about 20 mg/kg. Inhalation LC50 (4 h) for rats is 0.3–0.5 mg/L air. NOEL (2 yr) for rats 1 mg/kg diet (0.05 mg/kg/d). ADI is 1 μg/kg. In mammals, administered isofenphos is rapidly metabolized and eliminated; almost 95% is excreted within 24 h in the urine and feces. The active metabolite is des-N-isopropylisofenphos oxon.
Properties of ISOFENPHOS
| Melting point: | -12℃ |
| Boiling point: | bp0.01 120° |
| Density | 1.131 g/cm3 (20 ºC) |
| vapor pressure | 2.2 x 10-4 Pa (20 °C) |
| storage temp. | 0-6°C |
| solubility | Chloroform (Slightly), DMSO (Slightly) |
| form | Oil |
| pka | -0.30±0.70(Predicted) |
| Water Solubility | 18 mg l-1 (20 °C) |
| color | Colourless |
| CAS DataBase Reference | 25311-71-1(CAS DataBase Reference) |
| EPA Substance Registry System | Isofenphos (25311-71-1) |
Safety information for ISOFENPHOS
| Signal word | Danger |
| Pictogram(s) |
 Flame Flammables GHS02  Exclamation Mark Irritant GHS07 |
| GHS Hazard Statements |
H225:Flammable liquids H319:Serious eye damage/eye irritation H412:Hazardous to the aquatic environment, long-term hazard |
| Precautionary Statement Codes |
P210:Keep away from heat/sparks/open flames/hot surfaces. — No smoking. P273:Avoid release to the environment. P280:Wear protective gloves/protective clothing/eye protection/face protection. P305+P351+P338:IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses, if present and easy to do. Continuerinsing. |
Computed Descriptors for ISOFENPHOS
New Products
Bromine 99.5% AR (4 x 500ml) Fehling's Solution No. B Amino Acid Kit of 23 items set Ammonium Molybdate Reagent Solution Beam's Reagent Solution Ehrlich's Reagent For detection of urobillinogen 4-((2-isopropoxyethoxy)methyl)phenol 4-Hydroxy Carbazole Amino Salicylic Acid. U.S.P. 1,2,3,4-Tetrahydrocarbazol-4-one 2 – Methoxy – 5- Sulfamoyl Benzoic acid Acetone Isobutryl oxime ester Curcuma aromatica Oil Curry leaf Extract Terminalia bellirica Extract Aloe vera extract 200x Withania somnifera (Ashwagandha Extract) Citrus bioflavonoids Extract 5-Nitrosalicylaldehyde 5-(Difluoromethoxy)-2-Mercapto-1H-Benzimidazole- IP/BP/ Ethyl 3-(Pyridin-2-Ylamino)Propanoate Bilastine -IP/BP/ Cypermethric Acid Chloride Methyl Di Chloride (Mdc)Related products of tetrahydrofuran




You may like
-
 7726-95-6 Bromine 99.5% AR (4 x 500ml) 99%View Details
7726-95-6 Bromine 99.5% AR (4 x 500ml) 99%View Details
7726-95-6 -
 Formamide 99%View Details
Formamide 99%View Details
75-12-7 -
 376608-65-0 2-[[(3Ar,4S,6R,6As)-6-Aminotetrahydro-2,2-Dimethyl-4H-Cy 99%View Details
376608-65-0 2-[[(3Ar,4S,6R,6As)-6-Aminotetrahydro-2,2-Dimethyl-4H-Cy 99%View Details
376608-65-0 -
 2, 4-Pyrimidinediamine 3-Oxide 99%View Details
2, 4-Pyrimidinediamine 3-Oxide 99%View Details
74638-76-9 -
 111974-72-2 99%View Details
111974-72-2 99%View Details
111974-72-2 -
 85-81-4 6-Methoxy-8-Nitroquinoline 99%View Details
85-81-4 6-Methoxy-8-Nitroquinoline 99%View Details
85-81-4 -
![3-Bromo-4,5-Dihydro-1H-Benzo[B]Azepin-2(3H)-One 99%](https://img.chemicalbook.in//Content/image/CP5.jpg) 3-Bromo-4,5-Dihydro-1H-Benzo[B]Azepin-2(3H)-One 99%View Details
3-Bromo-4,5-Dihydro-1H-Benzo[B]Azepin-2(3H)-One 99%View Details
86499-96-9 -
 (−)-Dip-Chloride 85116-37-6 99%View Details
(−)-Dip-Chloride 85116-37-6 99%View Details
85116-37-6